Advanced Photocatalytic Synthesis of High-Potency Trifluoromethylated Aniline Intermediates for Oncology Applications
Advanced Photocatalytic Synthesis of High-Potency Trifluoromethylated Aniline Intermediates for Oncology Applications
The pharmaceutical industry is constantly seeking novel chemical entities that offer improved therapeutic indices, particularly in the challenging field of oncology. A significant breakthrough in this domain is detailed in Chinese Patent CN112125838B, which discloses a series of trifluoromethylated aniline compounds and their application in preparing antitumor medicaments. This technology addresses a critical need in modern drug discovery: the efficient modification of existing pharmacophores to enhance potency and metabolic stability. By leveraging visible-light photocatalysis, this invention provides a direct route to introduce the trifluoromethyl moiety—a privileged structure in medicinal chemistry—onto biologically active aniline scaffolds such as aminoglutethimide and sulfonamides. The resulting compounds, particularly the derivative designated as Compound (Ia), exhibit superior antitumor activity, specifically against cervical cancer cell lines, representing a substantial advancement over prior art methodologies.
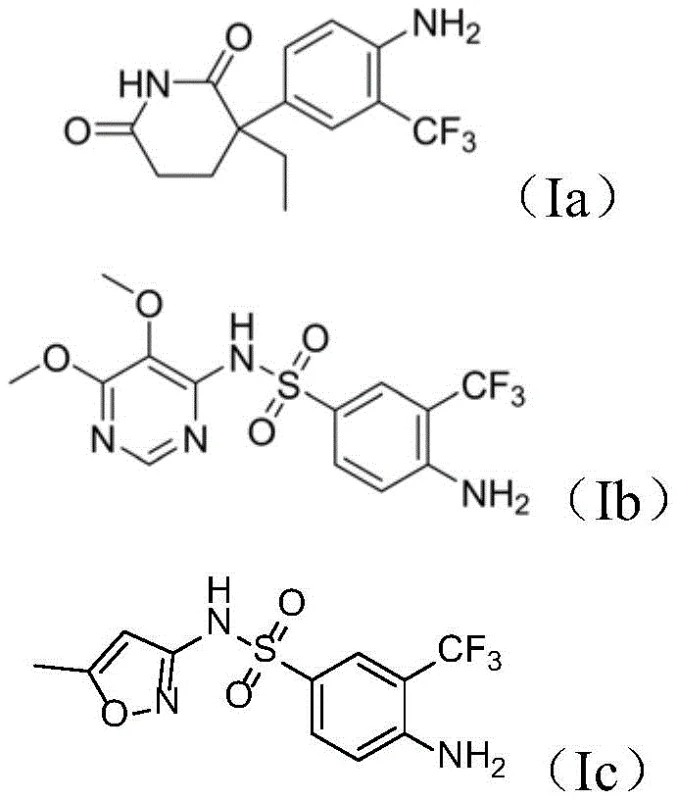
For R&D directors and process chemists, the structural versatility offered by this patent is compelling. The general formula (I) encompasses a range of substituents (R1), allowing for the rapid generation of diverse analogues for structure-activity relationship (SAR) studies. The ability to functionalize complex drug molecules in a single step without protecting group manipulation is a rare and valuable capability. As a reliable pharmaceutical intermediate supplier, understanding these structural nuances is key to supporting clients who are looking to optimize their oncology pipelines with high-purity building blocks that possess enhanced lipophilicity and membrane permeability characteristics inherent to fluorinated compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of trifluoromethyl groups into aromatic systems has been fraught with synthetic challenges. Traditional methods often rely on harsh reaction conditions, including high temperatures, strong acids, or the use of stoichiometric amounts of toxic heavy metal catalysts. These conventional approaches frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. Furthermore, many established protocols require multiple synthetic steps, involving tedious protection and deprotection sequences that drastically reduce overall yield and increase the environmental footprint of the manufacturing process. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, while for supply chain heads, the reliance on specialized, hazardous reagents introduces significant logistical and safety risks.
The Novel Approach
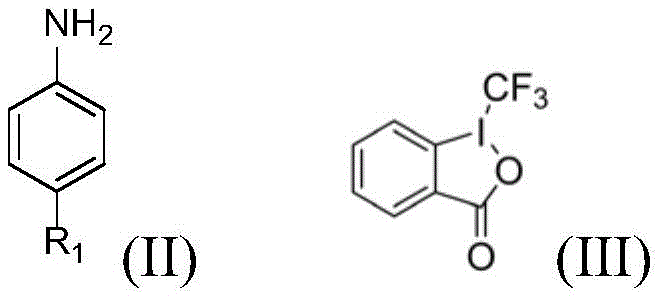
In stark contrast, the methodology described in patent CN112125838B utilizes a visible-light-driven organocatalytic system that operates under exceptionally mild conditions. The core innovation lies in the use of fluorescein as a photocatalyst in conjunction with a hypervalent iodine reagent, specifically 1-(trifluoromethyl)-1,2-benziodoxol-3(1H)-one. This combination enables the direct trifluoromethylation of the aniline ring at room temperature, bypassing the need for thermal activation or expensive transition metals. The reaction proceeds efficiently in a green solvent system comprising a mixture of DMF and water, highlighting a commitment to sustainability. This streamlined approach not only simplifies the operational workflow but also ensures high site selectivity, delivering the desired trifluoromethylated product with minimal byproduct formation.
Mechanistic Insights into Fluorescein-Catalyzed Radical Trifluoromethylation
To fully appreciate the technical robustness of this synthesis, one must examine the underlying mechanistic pathway. The reaction is initiated by the absorption of blue light by the fluorescein catalyst, which promotes it to an excited state capable of engaging in single-electron transfer (SET) processes. In this excited state, the catalyst interacts with the hypervalent iodine reagent, facilitating the homolytic cleavage of the carbon-iodine bond to generate a trifluoromethyl radical. This highly reactive species then selectively attacks the electron-rich aromatic ring of the aniline substrate. The presence of the amino group directs the substitution, typically favoring the para-position due to electronic activation, although steric factors from the R1 group can influence the outcome. The subsequent oxidation and deprotonation steps restore aromaticity and regenerate the ground-state catalyst, completing the catalytic cycle.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways. Because the reaction occurs under neutral, mild conditions, there is a significantly reduced risk of acid-catalyzed degradation of sensitive functional groups often present in complex drug molecules like aminoglutethimide. The use of water as a co-solvent further aids in suppressing side reactions that might occur in purely organic media. For quality assurance teams, this translates to a cleaner crude reaction profile, which simplifies downstream purification. The high selectivity minimizes the formation of regioisomers, ensuring that the final high-purity pharmaceutical intermediate meets stringent specifications required for clinical trial material production without the need for extensive recrystallization or preparative HPLC.
How to Synthesize Trifluoromethylated Aniline Efficiently
The practical implementation of this chemistry is straightforward, making it accessible for both laboratory-scale optimization and pilot plant operations. The standard protocol involves dissolving the aniline substrate and the trifluoromethylating agent in a 1:1 mixture of DMF and water, followed by the addition of a catalytic amount of fluorescein. The mixture is then irradiated with blue LEDs at ambient temperature for a period of 6 to 8 hours. Upon completion, the product is isolated through a simple liquid-liquid extraction using ethyl acetate and brine, followed by drying and concentration. The crude material is typically pure enough to be polished via standard silica gel chromatography. For a detailed breakdown of the specific molar ratios, solvent volumes, and purification parameters, please refer to the standardized synthesis guide below.
- Prepare a reaction mixture by dissolving the aniline substrate (e.g., aminoglutethimide) and 1-(trifluoromethyl)-1,2-benziodoxol-3(1H)-one in a 1: 1 volume ratio of DMF and water.
- Add fluorescein as the photocatalyst to the solution and irradiate the mixture with blue light at room temperature for 6 to 8 hours to facilitate the radical trifluoromethylation.
- Perform post-reaction workup by extracting with ethyl acetate and saturated NaCl, drying the organic layer, and purifying the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the shift to this photocatalytic methodology offers transformative benefits. The elimination of transition metal catalysts is perhaps the most significant cost driver; removing the need for palladium, copper, or rhodium not only lowers the direct cost of goods sold (COGS) but also eradicates the expensive and time-consuming heavy metal scavenging steps that are mandatory in GMP manufacturing. This simplification of the downstream processing directly contributes to cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the use of commercially available, commodity-grade starting materials ensures that the supply chain is not vulnerable to the bottlenecks often associated with exotic, custom-synthesized reagents.
- Cost Reduction in Manufacturing: The economic impact of this process extends beyond just reagent costs. By operating at room temperature, the energy consumption for heating or cooling reactors is drastically minimized compared to traditional thermal methods. The simplified workup procedure, which avoids complex distillation or crystallization steps often required to remove metal residues, reduces labor hours and solvent usage. Consequently, the overall production cost per kilogram is significantly lowered, providing a competitive pricing structure for bulk procurement without compromising on the quality or purity of the final active pharmaceutical ingredient (API) precursor.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance for water in the solvent system and the stability of the fluorescein catalyst mean that the process is less sensitive to minor fluctuations in raw material quality or environmental conditions. This reliability reduces the risk of batch failures and ensures consistent delivery schedules. Additionally, since the key reagents are widely commercialized, sourcing is not dependent on a single niche supplier, thereby mitigating the risk of supply disruptions and enabling more flexible inventory management strategies for long-term projects.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration issues, but modern flow chemistry technologies have largely solved this, making the commercial scale-up of complex pharmaceutical intermediates feasible. The benign nature of the catalyst and the aqueous solvent component align perfectly with increasingly strict environmental regulations regarding waste disposal. By generating less hazardous waste and avoiding toxic heavy metals, this process facilitates easier regulatory approval and reduces the environmental compliance burden on manufacturing facilities, supporting sustainable growth initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these trifluoromethylated compounds. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what partners can expect when integrating this technology into their development pipelines.
Q: What is the primary advantage of using fluorescein for trifluoromethylation compared to traditional metal catalysts?
A: The use of fluorescein eliminates the need for expensive and toxic transition metals like palladium or copper. This organocatalytic approach operates under mild room temperature conditions with visible light, significantly simplifying the purification process by removing the requirement for rigorous heavy metal scavenging steps often mandated in pharmaceutical manufacturing.
Q: How does the introduction of a trifluoromethyl group affect the biological activity of aniline-based drugs?
A: The trifluoromethyl group possesses strong electroabsorbability and lipophilicity while maintaining a stable C-F bond. Its introduction significantly alters the acidity, polarity, and metabolic stability of the parent molecule. In the context of this patent, modifying drugs like aminoglutethimide resulted in a marked improvement in antitumor activity, specifically against cervical cancer cells, demonstrating more than a twofold increase in potency compared to the unmodified parent compound.
Q: Is this photocatalytic method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method is highly amenable to scale-up. The reaction utilizes commercially available raw materials and proceeds at room temperature without the need for high pressure or extreme heating. The simple aqueous/organic solvent system and the use of a non-metal catalyst reduce safety hazards and waste treatment costs, making it a viable candidate for commercial-scale manufacturing of complex oncology intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylated Aniline Supplier
The potential of trifluoromethylated aniline derivatives in oncology is immense, yet realizing this potential requires a manufacturing partner with deep technical expertise and proven scalability. NINGBO INNO PHARMCHEM stands ready to support your drug development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art photocatalytic reactors and rigorous QC labs capable of ensuring stringent purity specifications for every batch. We understand that consistency is critical in the pharmaceutical supply chain, and our dedicated process chemistry team is committed to optimizing this green synthesis route to meet your specific volume and timeline requirements.
We invite you to discuss how this advanced trifluoromethylation technology can accelerate your oncology program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are prepared to provide specific COA data for our reference standards and comprehensive route feasibility assessments to demonstrate how we can become your strategic partner in delivering high-quality, cost-effective intermediates for the next generation of antitumor therapies.
