Scalable Aqueous Synthesis of Tripropargyl Isocyanurate for Advanced Polymer Applications
The development of high-performance thermosetting resins has long been constrained by the complexity and environmental burden of synthesizing their precursor monomers. A pivotal advancement in this domain is detailed in patent CN102030717B, which discloses a robust synthetic method for 1,3,5-tripropargyl-1,3,5-triazine-2,4,6-trione, a critical trialkynyl monomer. This innovation shifts the paradigm from hazardous organic solvent systems to a benign aqueous phase transfer catalysis (PTC) protocol. For R&D directors and supply chain strategists, this transition represents not merely a chemical modification but a fundamental restructuring of the production value chain. By leveraging inexpensive commodity chemicals like isocyanuric acid and propargyl bromide in a water-based medium, the process mitigates the risks associated with volatile organic compound (VOC) emissions while enhancing operational safety. The technical breakthrough lies in the ability to achieve efficient triple N-alkylation under mild thermal conditions, specifically between 75-80°C, which dramatically lowers energy consumption compared to high-temperature fusion methods. This report analyzes the mechanistic depth and commercial viability of this route, positioning it as a cornerstone for next-generation polymer additive manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkynyl-functionalized triazine derivatives relied heavily on polar aprotic solvents such as dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) in the presence of strong alkaline salts. While chemically feasible, these conventional pathways suffer from severe industrial drawbacks that hinder large-scale adoption. The primary limitation is the formidable challenge of solvent recovery; DMF and DMSO possess high boiling points and strong affinity for water, making distillation energy-intensive and economically inefficient. Furthermore, prior art often necessitated the use of toxic co-solvents like benzene during the post-treatment purification stages to precipitate the product, introducing significant occupational health hazards and regulatory compliance burdens. From a supply chain perspective, the reliance on these specialized solvents creates vulnerability to price volatility and availability fluctuations. Additionally, the reaction conditions in these organic media often require rigorous anhydrous environments to prevent side reactions, increasing the capital expenditure for reactor drying and maintenance. The cumulative effect is a process with a bloated cost structure, complex waste streams requiring incineration, and a safety profile that is increasingly untenable in modern green chemistry frameworks.
The Novel Approach
In stark contrast, the methodology outlined in CN102030717B introduces a streamlined aqueous system that fundamentally resolves the solvent bottleneck. By employing water as the sole reaction medium, the process eliminates the need for expensive, hard-to-recycle organic solvents entirely. The core of this innovation is the utilization of phase transfer catalysts (PTC), such as tetrabutylammonium chloride (TBAC), tetrabutylphosphonium chloride, or 18-crown-6-ether, which facilitate the transport of reactive anionic species across the interface between the aqueous base and the organic halide. This allows the reaction to proceed efficiently at moderate temperatures of 75-80°C over a duration of 15-20 hours. The simplicity of the workup procedure is equally transformative; upon completion, the product precipitates or can be easily separated, and residual inorganic salts like sodium bromide are removed through simple water washing. This approach not only reduces the environmental footprint by avoiding toxic benzene derivatives but also simplifies the equipment requirements, allowing for the use of standard stainless steel reactors without the need for exotic linings resistant to aggressive organic solvents. The result is a synthesis route that is inherently safer, more cost-effective, and readily adaptable to multi-ton scale production.
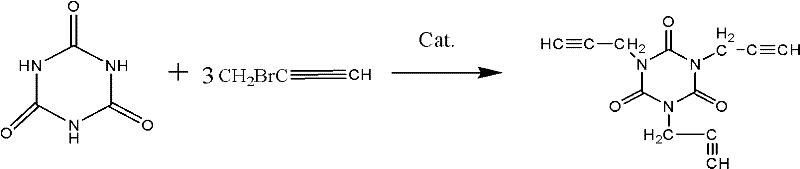
Mechanistic Insights into Phase Transfer Catalyzed N-Alkylation
The chemical elegance of this synthesis lies in the precise orchestration of nucleophilic substitution within a biphasic or pseudo-homogeneous aqueous environment. Isocyanuric acid, possessing three acidic imide protons, acts as the nucleophile upon deprotonation by the alkali metal hydroxide (NaOH or KOH). In a purely aqueous phase, the resulting tri-anion is heavily solvated and lacks the lipophilicity required to react effectively with the organic propargyl bromide. The phase transfer catalyst serves as a molecular shuttle; its lipophilic cation (e.g., tetrabutylammonium) pairs with the deprotonated isocyanurate anion, extracting it into the organic micro-environment or interface where the concentration of propargyl bromide is highest. This dramatically increases the local concentration of the reactive nucleophile, accelerating the SN2 alkylation kinetics. The reaction proceeds sequentially, alkylating the three nitrogen positions on the triazine ring. Crucially, the use of a molar excess of propargyl bromide (ratios ranging from 1:6 to 1:10 relative to isocyanuric acid) ensures that the reaction drives to the fully substituted tri-propargyl product, minimizing the formation of mono- or di-substituted intermediates which would act as impurities detrimental to the final resin's crosslinking density.
Impurity control in this system is managed through both kinetic optimization and physical separation. The mild reaction temperature of 75-80°C is sufficient to drive the alkylation but low enough to suppress the polymerization of the sensitive alkyne groups, which can occur at elevated temperatures or in the presence of trace transition metals. The choice of PTC is also critical; quaternary ammonium salts like TBAC offer a balance of stability and activity, whereas crown ethers provide high selectivity but at a higher cost. Post-reaction, the purification strategy leverages the differential solubility of the components. The target monomer is insoluble in cold water, while the byproduct salts (NaBr) and excess base remain in the aqueous phase. A subsequent wash with absolute ethanol removes any adsorbed organic impurities or unreacted propargyl bromide, yielding a high-purity white solid. This rigorous control over the impurity profile is essential for R&D teams, as residual halides or partially alkylated species can compromise the thermal curing profile of the final poly-triazine resin, leading to inconsistent mechanical properties in the cured matrix.
How to Synthesize 1,3,5-Tripropargyl-1,3,5-triazine-2,4,6-trione Efficiently
Implementing this synthesis requires careful attention to the addition rate of the alkylating agent and the maintenance of the thermal window to ensure safety and yield optimization. The process begins with the dissolution of isocyanuric acid and the base in water, followed by the introduction of the catalyst. The dropwise addition of propargyl bromide is exothermic and must be controlled to prevent runaway temperatures that could trigger alkyne polymerization. Detailed standardized operating procedures regarding stirring speeds, specific addition rates, and filtration parameters are critical for reproducibility. For a comprehensive breakdown of the exact experimental conditions and step-by-step execution protocols derived from the patent examples, please refer to the technical guide below.
- Charge a reactor with isocyanuric acid, alkali metal hydroxide (NaOH/KOH), phase transfer catalyst (e.g., TBAC), and water, then heat to 75-80°C with stirring.
- Slowly add propargyl bromide dropwise to the reaction mixture while maintaining the temperature between 75-80°C for 15-20 hours.
- Cool the mixture to room temperature, separate the solid product, wash thoroughly with water and absolute ethanol, and vacuum dry at 55°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this aqueous PTC methodology offers profound strategic advantages that extend beyond simple unit cost calculations. The elimination of high-boiling polar aprotic solvents removes a significant line item from the operating budget, as there is no longer a need for complex distillation columns or solvent recovery units. This simplification of the downstream processing infrastructure directly translates to reduced capital expenditure (CAPEX) for new production lines and lower maintenance costs for existing facilities. Furthermore, the use of water as a solvent inherently improves the safety profile of the plant, potentially lowering insurance premiums and reducing the regulatory burden associated with VOC emissions and hazardous waste disposal. The raw materials—isocyanuric acid and propargyl bromide—are commodity chemicals available from multiple global suppliers, ensuring supply continuity and mitigating the risk of single-source bottlenecks. This robustness in the supply chain is crucial for maintaining consistent production schedules for high-value polymer additives.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the substitution of expensive, difficult-to-recycle organic solvents with water, which is virtually free and non-toxic. By removing the energy-intensive steps of solvent distillation and recovery, the overall utility consumption per kilogram of product is drastically reduced. Additionally, the simplified workup procedure, which relies on filtration and washing rather than chromatography or complex extraction, reduces labor hours and processing time. The use of inexpensive phase transfer catalysts in catalytic quantities further minimizes raw material costs. This lean manufacturing approach allows for substantial margin improvement, making the final high-performance resin monomer more competitive against traditional epoxy or bismaleimide systems in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity feedstocks ensures that production is not held hostage by the scarcity of specialized reagents. Isocyanuric acid is a bulk chemical produced in massive quantities for the cyanuric chloride and swimming pool sanitizer industries, guaranteeing a stable and diversified supply base. Similarly, propargyl bromide is a standard intermediate in fine chemical synthesis. This abundance means that procurement teams can negotiate favorable long-term contracts and maintain safety stocks without fear of obsolescence or extreme price spikes. The mild reaction conditions also mean that the manufacturing can be performed in general-purpose chemical reactors, providing flexibility to shift production between different sites or campaigns without requiring dedicated, specialized equipment that might sit idle during downtime.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is straightforward due to the absence of hazardous solvents and the moderate thermal requirements. The reaction can be safely conducted in standard glass-lined or stainless steel vessels without the need for high-pressure ratings. From an environmental standpoint, the aqueous effluent primarily contains inorganic salts which are easier to treat than organic solvent waste streams, facilitating compliance with increasingly stringent environmental regulations like REACH or TSCA. The "green" nature of this synthesis also aligns with the sustainability goals of downstream customers in the automotive and aerospace sectors, who are under pressure to reduce the carbon footprint of their supply chains, thereby adding intangible value to the product portfolio.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this trialkynyl monomer. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these nuances is vital for integrating this monomer into your existing resin formulation workflows.
Q: Why is the aqueous PTC method superior to traditional DMF/DMSO routes?
A: Traditional methods utilize toxic polar aprotic solvents like DMF or DMSO which are difficult to recover and pose significant environmental hazards. The aqueous PTC method eliminates these volatile organic compounds, drastically simplifying waste treatment and reducing raw material costs.
Q: What represents the optimal molar ratio for high yield in this synthesis?
A: According to patent data, utilizing a molar excess of propargyl bromide relative to isocyanuric acid (ratios of 1:6 to 1:10) significantly drives the triple alkylation to completion, achieving yields up to 64.20% compared to lower stoichiometric ratios.
Q: How does the thermal stability of the cured resin compare to standard epoxies?
A: The resulting poly-1,3,5-tripropargyl-1,3,5-triazine-2,4,6-trione exhibits exceptional thermal stability with decomposition temperatures exceeding 380°C, far surpassing standard epoxy systems, making it ideal for aerospace and high-temperature electronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,5-Tripropargyl-1,3,5-triazine-2,4,6-trione Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN102030717B are realized in full-scale operations. We understand that high-performance resin monomers demand stringent purity specifications to function correctly in curing cycles; therefore, our rigorous QC labs employ advanced analytical techniques to verify the absence of partial alkylation byproducts and residual halides. Whether you require custom synthesis modifications or bulk supply of this critical triazine derivative, our infrastructure is designed to deliver consistency and quality that meets the exacting standards of the global polymer industry.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your product lineup. By leveraging our manufacturing efficiencies, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to request specific COA data and route feasibility assessments to validate the compatibility of our material with your current processing parameters. Let us collaborate to bring next-generation heat-resistant materials to market faster and more economically.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
