Advanced Palladium-Catalyzed Synthesis of 2,4-Difluorobenzonitrile for Commercial Scale-Up
The global demand for fluorinated aromatic compounds continues to surge, driven by their critical role in the development of next-generation pharmaceuticals and environmentally friendly agrochemicals. A pivotal intermediate in this sector is 2,4-difluorobenzonitrile, a versatile building block utilized in the synthesis of selective herbicides and various active pharmaceutical ingredients. Recent advancements in synthetic methodology have addressed long-standing challenges associated with its production, specifically focusing on safety and efficiency. Patent CN100457721C discloses a groundbreaking production process that utilizes a palladium-catalyzed cyanation strategy. This innovative approach replaces hazardous traditional reagents with benign alkali metal ferrocyanides, operating under mild conditions to deliver high-purity products. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this technology is essential for optimizing supply chains and ensuring regulatory compliance in the manufacture of complex fluorinated molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4-difluorobenzonitrile has been plagued by significant technical and safety hurdles that hinder large-scale commercial viability. Traditional routes often relied on the nucleophilic fluorination of 2,4-dichlorobenzonitrile, a process depicted in earlier literature such as JP 60-72850. While conceptually straightforward, this halogen exchange frequently requires extremely harsh reaction conditions, including high temperatures and the use of aggressive fluorinating agents that pose severe corrosion risks to reactor equipment. Furthermore, alternative pathways involving the diazotization of m-phenylenediamine, as reported in documents like JP 1990-113037, introduce complex multi-step sequences that are difficult to control. These older methodologies often suffer from prolonged reaction cycles, low overall yields due to side reactions, and the generation of substantial quantities of hazardous waste. The reliance on toxic cyanide sources in some variations further exacerbates environmental concerns, necessitating expensive waste treatment protocols and rigorous safety monitoring that drive up operational costs.
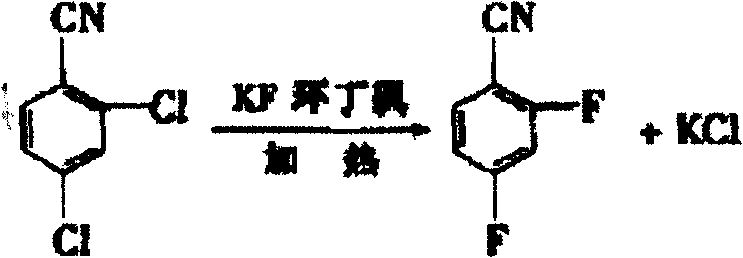
The Novel Approach
In stark contrast to these legacy methods, the novel process described in CN100457721C offers a streamlined and inherently safer alternative through direct palladium-catalyzed cyanation. This method employs 2,4-difluorobromobenzene as the starting substrate, reacting it with alkali metal ferrocyanide in the presence of a palladium catalyst and a base. The use of ferrocyanide is a game-changer; unlike free cyanide salts, ferrocyanides are stable, non-toxic, and easy to handle, drastically reducing the risk profile of the operation. The reaction proceeds smoothly in N,N-dimethylacetamide (DMAC) at moderate temperatures ranging from 100°C to 150°C. This shift not only simplifies the engineering requirements for the reactor but also significantly shortens the process flow. By eliminating the need for dangerous fluorination steps or complex diazotization sequences, this new route achieves superior atom economy and facilitates a much cleaner workup procedure, making it an ideal candidate for cost reduction in fine chemical manufacturing.
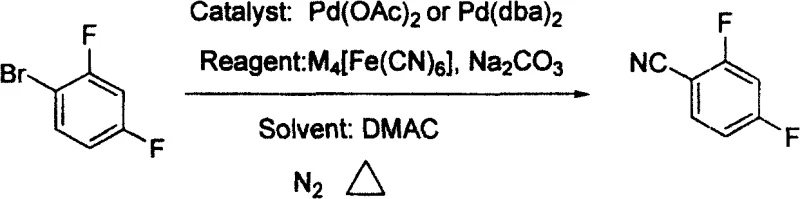
Mechanistic Insights into Pd-Catalyzed Cyanation
The core of this technological breakthrough lies in the efficient catalytic cycle mediated by palladium complexes, such as palladium acetate or bis(dibenzylideneacetone)palladium. The mechanism initiates with the oxidative addition of the aryl bromide bond of 2,4-difluorobromobenzene to the zero-valent palladium species, forming an aryl-palladium(II) intermediate. This step is crucial as it activates the relatively inert carbon-bromine bond for subsequent functionalization. Following this, a transmetallation event occurs where the cyanide group is transferred from the ferrocyanide complex to the palladium center. The presence of the alkali metal carbonate base plays a vital role here, likely facilitating the release of cyanide ions from the stable ferrocyanide lattice without generating free, toxic HCN gas. Finally, reductive elimination releases the desired 2,4-difluorobenzonitrile product and regenerates the active palladium catalyst, allowing the cycle to continue. This mechanistic pathway is highly selective, minimizing the formation of defluorinated byproducts or hydrolysis derivatives that often plague nucleophilic substitutions on electron-deficient aromatic rings.
From an impurity control perspective, this catalytic system offers distinct advantages over stoichiometric methods. The mild reaction conditions prevent the thermal degradation of the nitrile group, which can occur under the extreme temperatures required for older fluorination routes. Additionally, the use of ferrocyanide ensures that cyanide is delivered in a controlled manner, preventing local excesses that could lead to polymerization or other side reactions. The choice of DMAC as a solvent is also strategic; it effectively solubilizes both the organic substrate and the inorganic ferrocyanide salt, creating a homogeneous reaction environment that enhances mass transfer and reaction kinetics. For quality assurance teams, this translates to a consistent impurity profile that is easier to manage during downstream purification, ensuring the final high-purity 2,4-difluorobenzonitrile meets stringent specifications required for pharmaceutical applications.
How to Synthesize 2,4-Difluorobenzonitrile Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and atmospheric conditions to maximize yield and catalyst turnover. The process is designed to be robust, tolerating slight variations in temperature while maintaining high conversion rates. Operators should note that the molar ratio of the ferrocyanide source to the substrate is optimized to be slightly sub-stoichiometric or near-equivalent, leveraging the efficiency of the catalyst to drive the reaction to completion. Detailed standard operating procedures regarding the specific addition order of reagents and the precise control of the nitrogen blanket are critical for reproducibility. For a comprehensive guide on the exact experimental parameters and step-by-step execution, please refer to the standardized synthesis protocol outlined below.
- Charge a reactor with N,N-dimethylacetamide (DMAC) solvent, 2,4-difluorobromobenzene, alkali metal ferrocyanide (such as potassium ferrocyanide trihydrate), a palladium catalyst (like palladium acetate), and an alkali metal carbonate base under nitrogen protection.
- Heat the reaction mixture to a temperature range of 100°C to 150°C and maintain stirring for a duration of 1 to 6 hours to ensure complete conversion of the starting material.
- Upon completion, filter the reaction mixture to remove solid salts and catalyst residues, then subject the filtrate to fractional distillation under reduced pressure to isolate high-purity 2,4-difluorobenzonitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing process represents a significant opportunity to de-risk the supply of critical fluorinated intermediates. The primary advantage lies in the drastic simplification of the safety infrastructure required for production. By substituting highly toxic cyanide salts with benign ferrocyanides, facilities can reduce the regulatory burden and insurance costs associated with handling hazardous materials. This shift also mitigates the risk of production stoppages due to safety incidents or regulatory inspections, thereby enhancing the overall reliability of the supply chain. Furthermore, the simplified post-treatment process, which involves merely filtration followed by distillation, reduces the consumption of auxiliary chemicals and solvents, leading to substantial operational savings.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from multiple factors inherent to its design. Firstly, the elimination of expensive and hazardous fluorinating agents lowers the raw material costs significantly. Secondly, the low loading of the palladium catalyst, which can be as low as 0.1 mol%, minimizes the expense associated with precious metal usage, especially when coupled with efficient recovery systems. Thirdly, the shortened reaction time and single-step transformation reduce energy consumption and labor hours per batch. These factors combine to create a leaner manufacturing process that delivers substantial cost savings without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of sourcing specialized reagents or managing dangerous goods logistics. This new method utilizes readily available and stable starting materials like 2,4-difluorobromobenzene and common inorganic salts. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in utility supplies or environmental conditions. Consequently, manufacturers can maintain higher inventory levels and shorter lead times, ensuring that downstream customers in the pharmaceutical and agrochemical sectors receive their materials consistently and without delay.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial tonnage often reveals hidden bottlenecks, particularly regarding heat management and waste disposal. This palladium-catalyzed route is inherently scalable because it operates at moderate temperatures and pressures, avoiding the need for specialized high-pressure reactors. Moreover, the environmental footprint is markedly reduced due to the non-toxic nature of the cyanide source and the minimal generation of halogenated waste streams. This alignment with green chemistry principles facilitates easier permitting and compliance with increasingly strict environmental regulations, future-proofing the production capability against legislative changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,4-difluorobenzonitrile using this advanced methodology. These insights are derived directly from the patent data and practical implementation experiences, aiming to clarify the operational benefits and technical feasibility for potential partners. Understanding these details is crucial for making informed decisions about integrating this intermediate into your broader chemical supply network.
Q: Why is alkali metal ferrocyanide preferred over traditional cyanide sources?
A: Alkali metal ferrocyanides are significantly less toxic and safer to handle compared to traditional cyanide sources like sodium cyanide or potassium cyanide. They offer a non-toxic alternative that simplifies safety protocols and waste treatment in industrial settings.
Q: What are the typical reaction conditions for this palladium-catalyzed cyanation?
A: The reaction typically proceeds in N,N-dimethylacetamide (DMAC) solvent at temperatures between 100°C and 150°C under a nitrogen atmosphere. The catalyst loading is low, ranging from 0.1% to 5% molar equivalent relative to the substrate.
Q: How does this method improve product purity compared to older routes?
A: This method avoids harsh fluorination conditions and complex diazotization steps found in older routes, resulting in fewer side reactions. The simple workup involving filtration and distillation allows for the isolation of product with purity levels reaching up to 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Difluorobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex fluorinated intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,4-difluorobenzonitrile meets the exacting standards required for API synthesis and agrochemical formulation. Our facility is equipped to handle the specific safety requirements of palladium-catalyzed reactions, providing a secure and compliant environment for your production needs.
We invite you to collaborate with us to optimize your supply chain for fluorinated building blocks. By leveraging our technical capabilities, you can achieve significant efficiencies and secure a stable source of high-quality materials. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing processes can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
