Advanced Solvent-Free Synthesis of 7-(4-Chlorophenyl)-5,6-dihydro-7aH-benzo[h]1,2,4-triazolo[3,4-b]quinazoline-5,6-diketone for Commercial Scale-up
The pharmaceutical industry is constantly seeking robust synthetic routes for novel heterocyclic scaffolds that demonstrate potent biological activity, particularly in the realm of oncology. Patent CN102911177A, published in early 2013, introduces a significant advancement in the preparation of 1,2-naphthoquinone derivatives, specifically detailing the synthesis of 7-(4-chlorophenyl)-5,6-dihydro-7aH-benzo[h]1,2,4-triazolo[3,4-b]quinazoline-5,6-diketone. This compound represents a strategic fusion of a nitrogen-containing heterocycle with a 1,2-naphthoquinone core, a structural motif known for superior topoisomerase inhibition compared to traditional 1,4-naphthoquinones. The introduction of a chlorine substituent at the 4-position of the phenyl ring is not merely a structural variation but a critical design element intended to enhance lipophilicity and cellular uptake, thereby amplifying the compound's anticancer efficacy. For R&D directors and procurement specialists evaluating new intermediates, this patent offers a compelling case study in green chemistry, demonstrating how a complex polycyclic system can be assembled efficiently without the need for volatile organic solvents during the reaction phase.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of fused heterocyclic systems involving naphthoquinones often relies on multi-step sequences that require harsh reaction conditions and extensive use of organic solvents. Conventional pathways frequently involve the separate preparation of intermediates, purification steps between each stage, and the use of heavy metal catalysts or toxic reagents that complicate downstream processing. These legacy methods pose significant challenges for commercial scale-up, including high operational expenditures related to solvent recovery and waste treatment, as well as safety risks associated with handling large volumes of flammable liquids. Furthermore, the thermal stability of 1,2-naphthoquinone derivatives can be sensitive, and prolonged exposure to aggressive conditions in traditional protocols may lead to decomposition or the formation of difficult-to-remove impurities, ultimately compromising the purity profile required for pharmaceutical applications.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the methodology described in CN102911177A utilizes a streamlined, one-pot condensation strategy that operates under solvent-free conditions. By directly mixing 3-amino-1,2,4-triazole, p-chlorobenzaldehyde, and 2-hydroxy-1,4-naphthoquinone in the presence of a catalytic amount of p-toluenesulfonic acid, the reaction proceeds smoothly at temperatures between 110-125°C. This approach eliminates the need for bulk reaction solvents, drastically reducing the environmental footprint and simplifying the reactor setup. The visual representation of this efficient transformation highlights the direct convergence of three readily available starting materials into the complex target scaffold.
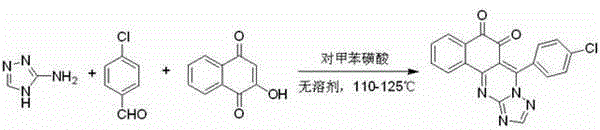
The reaction completes within a concise timeframe of 2.5 to 3 hours, yielding the desired product as a yellow solid after a simple workup involving dichloromethane dissolution and ethanol recrystallization. This drastic simplification of the synthetic workflow translates directly into reduced processing time and lower energy consumption, addressing key pain points for supply chain managers focused on throughput and cost efficiency.
Mechanistic Insights into Acid-Catalyzed Cyclization
From a mechanistic perspective, the transformation relies on the activation of the carbonyl groups by p-toluenesulfonic acid, which acts as a Brønsted acid catalyst to facilitate nucleophilic attacks and subsequent dehydration steps. The initial interaction likely involves the condensation of the amine group of the triazole with the aldehyde to form an imine intermediate, which then undergoes cyclization with the hydroxyl-naphthoquinone moiety. The rigidity of the final structure, as depicted in the molecular diagram, suggests a highly conjugated system that contributes to its stability and specific electronic properties necessary for biological interaction.
![Chemical structure of 7-(4-chlorophenyl)-5,6-dihydro-7aH-benzo[h]1,2,4-triazolo[3,4-b]quinazoline-5,6-diketone showing the chlorophenyl substitution](/insights/img/7-4-chlorophenyl-triazoloquinazoline-synthesis-pharma-supplier-20260309002310-01.png)
The presence of the chlorine atom on the phenyl ring is a crucial feature that differentiates this derivative from non-halogenated analogs. In medicinal chemistry, halogenation is a proven strategy to modulate metabolic stability and binding affinity; in this specific scaffold, the chlorine atom likely enhances the compound's ability to intercalate with DNA or inhibit topoisomerase enzymes more effectively than its unsubstituted counterparts. Understanding this structure-activity relationship is vital for R&D teams aiming to optimize lead compounds, as it validates the selection of p-chlorobenzaldehyde over other aldehydes. The high selectivity of this reaction, evidenced by the clean formation of the product with minimal side reactions, indicates a well-controlled thermodynamic pathway that favors the formation of the fused triazoloquinazoline ring system.
How to Synthesize 7-(4-Chlorophenyl)-5,6-dihydro-7aH-benzo[h]1,2,4-triazolo[3,4-b]quinazoline-5,6-diketone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise control over stoichiometry and temperature to maximize yield and minimize impurity formation. The patent specifies a molar ratio of 1:1~1.1:1 for the triazole, aldehyde, and naphthoquinone respectively, ensuring that the limiting reagent is fully consumed while preventing excessive accumulation of unreacted starting materials. The detailed standardized synthesis steps below outline the exact protocol for achieving the reported 85-90% yields, providing a clear roadmap for technical teams to replicate the process reliably.
- Uniformly mix 3-amino-1,2,4-triazole, p-chlorobenzaldehyde, 2-hydroxy-1,4-naphthoquinone, and a catalytic amount of p-toluenesulfonic acid in a reaction vessel.
- Heat the mixture with stirring, strictly controlling the temperature between 110-125°C, and maintain reaction for 2.5 to 3 hours.
- Dissolve the crude mixture in dichloromethane, wash with water, remove solvent, and recrystallize from 95% ethanol to obtain the pure yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free methodology offers tangible benefits that extend beyond mere chemical elegance. The elimination of reaction solvents fundamentally alters the cost structure of the manufacturing process by removing the expenses associated with purchasing, storing, and disposing of large volumes of organic liquids. This shift not only reduces the direct material costs but also mitigates regulatory burdens related to volatile organic compound (VOC) emissions, making the process more sustainable and compliant with increasingly stringent environmental regulations globally.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of solvent usage during the reaction phase. In traditional fine chemical manufacturing, solvent recovery and waste treatment can account for a significant portion of operating expenses. By conducting the reaction in a melt phase or neat conditions, the facility saves substantially on utility costs for distillation and reduces the volume of hazardous waste requiring specialized disposal. Furthermore, the use of p-toluenesulfonic acid as a catalyst is economically favorable compared to expensive transition metal catalysts, eliminating the need for costly metal scavenging steps to meet residual metal specifications in the final API.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis—3-amino-1,2,4-triazole, p-chlorobenzaldehyde, and 2-hydroxy-1,4-naphthoquinone—are commodity chemicals with established global supply chains. Their widespread availability ensures that production schedules are not vulnerable to the bottlenecks often associated with exotic or custom-synthesized reagents. This reliability allows for better inventory planning and reduces the risk of production stoppages due to raw material shortages, ensuring a consistent flow of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The reaction conditions, specifically the temperature range of 110-125°C, are easily achievable using standard industrial heating systems such as steam or thermal oil, facilitating seamless scale-up from kilogram to tonne quantities. The simplicity of the workup procedure, involving a straightforward extraction and recrystallization, minimizes the complexity of the equipment train required. This operational simplicity translates to higher throughput and reduced downtime between batches, while the green nature of the process aligns with corporate sustainability goals, enhancing the brand value of the final product in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific triazoloquinazoline derivative. These answers are derived directly from the experimental data and claims presented in the source patent documentation, providing accurate guidance for potential partners and technical evaluators.
Q: What is the reported yield and purity of this novel synthesis method?
A: According to patent CN102911177A, the method achieves high yields ranging from 85% to 90% depending on the scale, producing a yellow solid with a melting point of 245-246°C.
Q: Does this compound exhibit specific biological activity?
A: Yes, preliminary pharmacological testing indicates strong anticancer activity, specifically against breast cancer cell line MCF-7, with an IC50 value of 5.84 μg/mL.
Q: Why is the solvent-free approach advantageous for supply chain management?
A: Eliminating reaction solvents significantly reduces waste disposal costs and simplifies the workup procedure, leading to faster production cycles and enhanced environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-(4-Chlorophenyl) Triazoloquinazoline Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic and patent literature into commercially viable manufacturing processes. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN102911177A can be realized on an industrial scale without compromising quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 7-(4-chlorophenyl)-5,6-dihydro-7aH-benzo[h]1,2,4-triazolo[3,4-b]quinazoline-5,6-diketone meets the exacting standards required for pharmaceutical development.
We invite global pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthetic route for your anticancer drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your project timelines and help you secure a reliable supply of this high-value intermediate.
