Scalable Synthesis of Furo[3,2-b]pyridine Derivatives for High-Purity API Manufacturing
Scalable Synthesis of Furo[3,2-b]pyridine Derivatives for High-Purity API Manufacturing
The pharmaceutical industry continuously seeks robust and cost-effective pathways for constructing complex heterocyclic scaffolds essential for modern drug discovery. Patent CN114213424B introduces a groundbreaking synthesis method for furo[3,2-b]pyridine derivatives, a critical structural motif found in highly selective JAK1 inhibitors used for treating rheumatoid arthritis. This technology represents a significant departure from legacy methodologies by replacing expensive transition metal catalysis with a streamlined three-step sequence involving phosphonium salt formation, Wittig olefination, and acidic cyclization. For R&D directors and procurement specialists, this patent offers a compelling solution to the challenges of supply chain volatility and high manufacturing costs associated with precious metal-dependent processes. By leveraging common reagents such as triphenylphosphine and formate esters, this route ensures consistent quality and accessibility, positioning it as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
![Chemical structure of the final furo[3,2-b]pyridine derivative (Formula IV)](/insights/img/furo-3-2-b-pyridine-synthesis-supplier-20260303030246-04.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the furo[3,2-b]pyridine core has relied heavily on palladium-catalyzed Sonogashira coupling reactions, as documented in prior art such as WO200551304. These conventional approaches necessitate the use of 2-bromo-3-acetoxypyridine and substituted alkynes, requiring substantial quantities of expensive palladium catalysts to drive the reaction forward. Beyond the direct financial burden of precious metals, these methods introduce significant downstream processing challenges, including the rigorous removal of trace metal residues to meet stringent pharmaceutical purity standards. Furthermore, alternative historical routes reported in journals like J. Heterocycl. Chem. involve multi-step sequences exceeding five distinct transformations, leading to cumulative yield losses and increased operational complexity. Such inefficiencies render traditional methods economically unviable for large-scale industrial production, creating bottlenecks in the supply of high-purity API intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN114213424B simplifies the synthetic landscape into a concise three-step protocol that eliminates the need for transition metals entirely. The process initiates with a straightforward nucleophilic substitution to generate a phosphonium salt, followed by a Wittig reaction with a formate ester to install the necessary carbon framework. The final step involves an elegant acid-mediated cyclization that simultaneously constructs the furan ring and reveals the hydroxyl functionality. This approach not only drastically reduces the number of unit operations but also utilizes inexpensive, commodity-grade chemicals that are readily available in the global market. By avoiding the complexities of air-sensitive catalytic systems and multi-step purifications, this novel route offers a direct path to cost reduction in pharmaceutical intermediate manufacturing while maintaining high throughput and operational safety.
Mechanistic Insights into Wittig Olefination and Acidic Cyclization
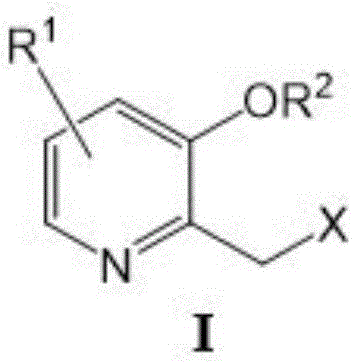
The core of this synthetic strategy lies in the efficient generation of the exocyclic double bond via a Wittig reaction, which serves as the precursor for the subsequent ring closure. Initially, the starting pyridine derivative, represented by Formula I, undergoes quaternization with triphenylphosphine under reflux conditions to form the stable phosphonium salt (Formula II). This intermediate is then treated with a strong base, such as potassium tert-butoxide, to generate the reactive ylide species in situ. Upon addition of ethyl formate, the ylide attacks the carbonyl carbon, resulting in the formation of the vinyl ether intermediate (Formula III) with high stereochemical control. This step is critical as it establishes the two-carbon bridge required for furan ring formation, and the use of formate esters ensures that no extraneous carbon atoms are introduced, thereby simplifying the molecular architecture.
Following the construction of the side chain, the final transformation involves a sophisticated acid-promoted cyclization that converts the linear vinyl ether into the fused bicyclic system. The patent details the use of potent Lewis acids like boron tribromide or Bronsted acids like concentrated sulfuric acid to facilitate this cascade. Under acidic conditions, the ether linkage is cleaved, generating a reactive electrophilic center that undergoes intramolecular attack by the adjacent oxygen or carbon nucleophile, depending on the specific substitution pattern. This cyclization is accompanied by demethylation if methoxy groups are present, directly yielding the hydroxy-substituted furo[3,2-b]pyridine (Formula IV). The robustness of this mechanism allows for flexible reaction conditions, tolerating various solvents and temperatures, which is essential for controlling impurity profiles and ensuring the production of high-purity OLED material or pharmaceutical grades.
How to Synthesize 7-Hydroxyfuro[3,2-b]pyridine Efficiently
The synthesis of 7-hydroxyfuro[3,2-b]pyridine serves as a prime example of the versatility and efficiency of this patented platform. The process begins with the preparation of the phosphonium salt from 2-chloromethyl-3,4-dimethoxypyridine, followed by the Wittig reaction with ethyl formate to generate the key enol ether intermediate. The final cyclization step utilizes boron tribromide for demethylation and ring closure, followed by an acid treatment to finalize the structure. This sequence has been optimized to minimize side reactions and maximize isolation yields through simple filtration and extraction techniques. For detailed operational parameters, stoichiometry, and safety protocols required to replicate this high-yielding pathway in your facility, please refer to the standardized synthesis guide below.
- Reflux the substituted pyridine derivative (Formula I) with triphenylphosphine in a solvent like toluene to generate the phosphonium salt (Formula II).
- React the phosphonium salt with a base and formate ester under controlled temperatures to perform a Wittig reaction, yielding the vinyl ether intermediate (Formula III).
- Subject the vinyl ether intermediate to acidic conditions using Lewis or Bronsted acids to induce cyclization and demethylation, forming the final furo[3,2-b]pyridine (Formula IV).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic advantages regarding cost stability and supply continuity. By eliminating the dependency on volatile precious metal markets and complex catalytic systems, manufacturers can secure a more predictable cost structure for their raw materials. The simplicity of the workup procedures, which primarily involve filtration and liquid-liquid extraction rather than column chromatography or specialized metal scavenging, significantly reduces processing time and waste generation. This operational efficiency allows for faster batch turnover and reduced lead time for high-purity pharmaceutical intermediates, ensuring that downstream API production schedules are met without delay. Furthermore, the use of common solvents like toluene and dichloromethane simplifies solvent recovery and recycling, contributing to a more sustainable and environmentally compliant manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts removes a major cost driver and the associated expense of metal removal technologies. Additionally, the high yields reported in the patent examples indicate minimal material loss, leading to substantial cost savings per kilogram of finished product. The use of commodity reagents further insulates the production cost from fluctuations in the specialty chemical market, providing a stable economic model for long-term contracts.
- Enhanced Supply Chain Reliability: Since the starting materials and reagents are widely produced and stocked globally, the risk of supply disruption is significantly mitigated compared to routes relying on custom-synthesized alkynes or specialized ligands. This availability ensures that production can be scaled up rapidly to meet surging demand without the long lead times typically associated with sourcing exotic precursors. The robustness of the chemistry also means that production is less susceptible to minor variations in raw material quality, enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial application, utilizing standard reactor equipment and avoiding hazardous high-pressure or cryogenic conditions. The simplified purification steps reduce the volume of organic waste and silica gel consumption, aligning with green chemistry principles. This ease of scale-up facilitates the transition from pilot plant to commercial production, allowing suppliers to quickly ramp up capacity from 100 kgs to multi-ton quantities while maintaining strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this furo[3,2-b]pyridine synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, impurity control, and scalability. Understanding these factors is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. We encourage stakeholders to review these answers to gain a comprehensive understanding of the process capabilities and limitations.
Q: Why is this new synthesis method preferred over traditional Palladium-catalyzed routes?
A: Traditional methods often rely on expensive Palladium catalysts and Sonogashira couplings, which introduce heavy metal impurities and high costs. This novel method utilizes common reagents like triphenylphosphine and formates, eliminating the need for precious metals and complex purification steps.
Q: What are the typical yields and purity levels achievable with this process?
A: According to patent data, the process achieves high yields across all three steps, with individual step yields ranging from approximately 74% to over 95%. The final product, such as 7-hydroxyfuro[3,2-b]pyridine, demonstrates HPLC purity greater than 98% after standard workup procedures.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is designed for industrial scalability. It uses readily available raw materials, avoids sensitive catalytic systems, and employs robust reaction conditions (reflux, standard extractions) that are easily transferable from laboratory to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furo[3,2-b]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to marketplace. Our state-of-the-art facilities are equipped to handle the specific requirements of this phosphonium-based chemistry, including rigorous QC labs capable of verifying stringent purity specifications and low residual solvent levels. We are committed to delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical industry, supporting your R&D and commercial goals with reliability and precision.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us help you optimize your supply chain and accelerate the delivery of life-saving medications to patients worldwide through our expert manufacturing capabilities.
