Advanced Benzenesulfonamide Carbapenem Derivatives: Scalable Synthesis and Commercial Potential
The pharmaceutical landscape is constantly evolving to combat the rising tide of bacterial resistance, necessitating the development of next-generation antibiotics with improved pharmacokinetic profiles. Patent CN101711250B introduces a novel class of benzenesulfonamide methylene substituted mercaptopyrrolidine carbapenem derivatives that address critical limitations of existing therapies. These compounds, represented by general formula (I), exhibit potent antibacterial activity against both Gram-positive and Gram-negative pathogens, including resistant strains, while demonstrating a remarkably extended half-life in vivo. 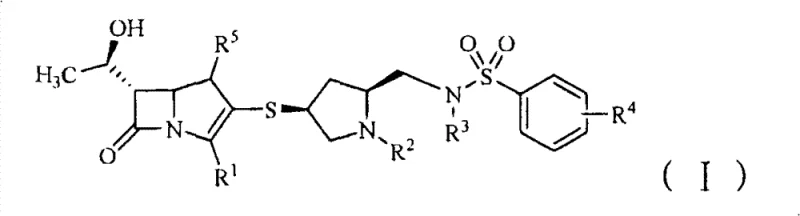 . For procurement managers and supply chain heads seeking a reliable pharmaceutical intermediates supplier, understanding the structural nuances and synthetic accessibility of these molecules is paramount. The innovation lies not just in the biological efficacy but in the robust chemical architecture that allows for scalable manufacturing and potential cost reduction in antibiotic manufacturing.
. For procurement managers and supply chain heads seeking a reliable pharmaceutical intermediates supplier, understanding the structural nuances and synthetic accessibility of these molecules is paramount. The innovation lies not just in the biological efficacy but in the robust chemical architecture that allows for scalable manufacturing and potential cost reduction in antibiotic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional carbapenem antibiotics such as Meropenem and Doripenem have served as the backbone for treating severe infections, yet their clinical utility is increasingly compromised by short plasma half-lives, typically around one hour in humans. This pharmacokinetic constraint necessitates frequent intravenous administrations, which burdens healthcare systems and reduces patient compliance. Furthermore, the synthesis of conventional carbapenems often involves complex protection and deprotection strategies that can lower overall yields and increase production costs. The reliance on unstable intermediates and harsh reaction conditions in older methodologies poses significant challenges for commercial scale-up of complex pharmaceutical intermediates. Additionally, the emergence of bacteria producing beta-lactamases and exhibiting resistance to renal dehydropeptidase I (DHP-I) requires constant structural modification of the core scaffold, which traditional methods struggle to accommodate efficiently without sacrificing purity or stability.
The Novel Approach
The approach detailed in the patent data leverages a strategic modification of the C-2 side chain, introducing a benzenesulfonamide methylene substituted mercaptopyrrolidine moiety. This structural alteration is designed to enhance affinity for penicillin-binding proteins (PBPs) while simultaneously improving metabolic stability. By utilizing a nucleophilic substitution strategy to attach this sophisticated side chain to the carbapenem core, the synthesis avoids some of the more cumbersome steps associated with earlier generations. The use of specific leaving groups, such as diphenyl phosphate, facilitates a cleaner coupling reaction, thereby reducing impurity profiles. This novel pathway supports the production of high-purity carbapenem derivatives that maintain stability against DHP-I, allowing for monotherapy applications. The result is a compound class that offers a superior therapeutic index, potentially reducing the total dosage required and extending the post-antibiotic effect, which is a critical value proposition for healthcare providers.
Mechanistic Insights into Nucleophilic Substitution and Side Chain Coupling
The core chemical transformation enabling this technology is the nucleophilic substitution reaction between the thiol-containing pyrrolidine intermediate (Formula II) and the activated carbapenem core (Formula III). 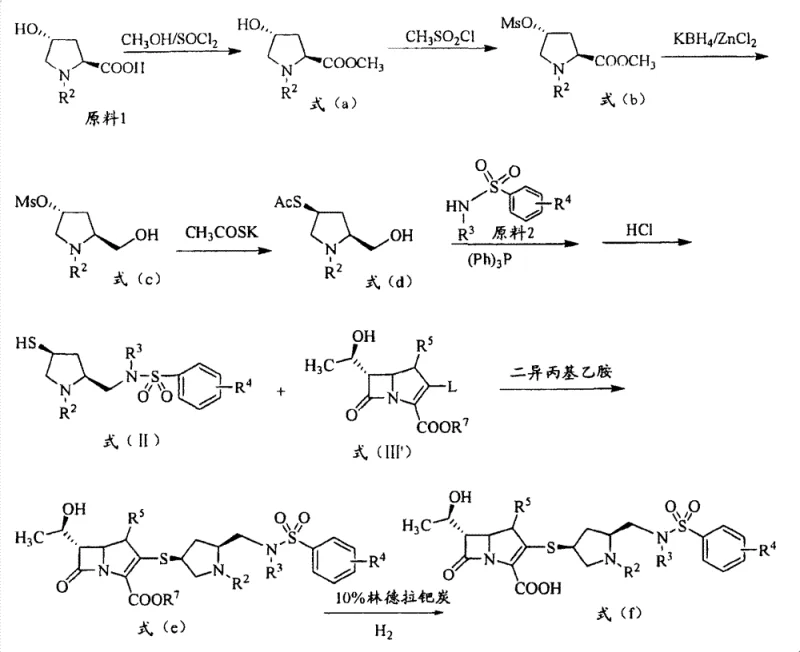 . In this mechanism, the sulfur atom of the mercaptopyrrolidine acts as a potent nucleophile, attacking the C-3 position of the carbapenem ring system where a leaving group (L) is positioned. The patent specifies that preferred leaving groups include diphenyl phosphate, which provides an optimal balance of reactivity and stability during the reaction. The process is typically conducted in polar aprotic solvents like acetonitrile or DMF, often in the presence of a base such as diisopropylethylamine to scavenge the acid generated during the substitution. This step is crucial for establishing the stereochemical integrity of the final product, as the chiral centers in the pyrrolidine ring must be preserved to ensure biological activity. The reaction conditions are carefully controlled, often at low temperatures initially to prevent degradation of the sensitive beta-lactam ring, followed by warming to drive the reaction to completion.
. In this mechanism, the sulfur atom of the mercaptopyrrolidine acts as a potent nucleophile, attacking the C-3 position of the carbapenem ring system where a leaving group (L) is positioned. The patent specifies that preferred leaving groups include diphenyl phosphate, which provides an optimal balance of reactivity and stability during the reaction. The process is typically conducted in polar aprotic solvents like acetonitrile or DMF, often in the presence of a base such as diisopropylethylamine to scavenge the acid generated during the substitution. This step is crucial for establishing the stereochemical integrity of the final product, as the chiral centers in the pyrrolidine ring must be preserved to ensure biological activity. The reaction conditions are carefully controlled, often at low temperatures initially to prevent degradation of the sensitive beta-lactam ring, followed by warming to drive the reaction to completion.
Impurity control is managed through the strategic use of protecting groups on the amino and carboxyl functionalities throughout the synthesis. For instance, the use of tert-butoxycarbonyl (Boc) groups on the pyrrolidine nitrogen and p-nitrobenzyl esters on the carbapenem carboxylate ensures that reactive sites are masked until the final stages. The deprotection sequence, involving catalytic hydrogenation over palladium-carbon and acid treatment, is designed to be orthogonal, removing the protecting groups without affecting the sensitive beta-lactam moiety. This meticulous control over the reaction pathway minimizes the formation of diastereomers and hydrolysis byproducts, which are common pitfalls in carbapenem synthesis. The final purification steps, often involving recrystallization from water-isopropanol mixtures, further enhance the purity profile, ensuring that the resulting active pharmaceutical ingredient meets stringent regulatory standards for parenteral administration.
How to Synthesize Benzenesulfonamide Carbapenem Derivatives Efficiently
The synthesis of these advanced intermediates follows a logical, multi-step sequence that begins with readily available chiral pool materials. The process initiates with the modification of trans-4-hydroxy-L-proline, undergoing esterification, mesylation, reduction, and thioacetylation to generate the key mercaptopyrrolidine building block. This intermediate is then coupled with various benzenesulfonamide derivatives via a Mitsunobu reaction or similar nucleophilic displacement to install the diverse R4 substituents. The final assembly involves the coupling of this functionalized side chain with the carbapenem core, followed by global deprotection. The detailed standardized synthesis steps see the guide below for specific reaction conditions and stoichiometry optimized for industrial throughput.
- Preparation of the mercaptopyrrolidine intermediate via esterification, mesylation, reduction, and thioacetylation of trans-4-hydroxy-L-proline.
- Coupling of the thiol intermediate with a benzenesulfonamide derivative using Mitsunobu reaction conditions to form the sulfonamide linkage.
- Final nucleophilic substitution with a carbapenem core bearing a leaving group, followed by deprotection and hydrogenation to yield the target acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the transition to this new class of intermediates offers tangible logistical and economic benefits derived from the underlying chemistry. The synthetic route utilizes starting materials that are commercially available in bulk quantities, such as trans-4-hydroxy-L-proline and common benzenesulfonamides, which mitigates the risk of raw material shortages. The reaction conditions, while requiring precise control, do not rely on exotic or prohibitively expensive catalysts, which contributes to cost reduction in antibiotic manufacturing. Furthermore, the isolation of intermediates as crystalline solids at various stages allows for effective purification and quality control checkpoints, reducing the likelihood of batch failures and ensuring consistent supply continuity. The robustness of the synthesis supports scaling from laboratory to commercial production without significant re-engineering of the process.
- Cost Reduction in Manufacturing: The elimination of certain complex enzymatic resolution steps found in older carbapenem processes, replaced by efficient chemical transformations using standard reagents like thionyl chloride and potassium thioacetate, streamlines the production workflow. This simplification reduces the consumption of specialized enzymes and lowers the overall cost of goods sold. Additionally, the high yields reported in the patent examples for key steps, such as the esterification and reduction phases, minimize waste generation and maximize material throughput. By optimizing the stoichiometry and reaction times, manufacturers can achieve significant operational efficiencies that translate directly into margin improvements.
- Enhanced Supply Chain Reliability: The reliance on stable, non-perishable reagents and the ability to store key intermediates as protected esters enhances the resilience of the supply chain. Unlike some biological fermentation processes that are susceptible to contamination and variability, this chemical synthesis offers predictable timelines and output volumes. The modular nature of the synthesis, where different benzenesulfonamide variants can be introduced late in the sequence, allows for flexible manufacturing schedules to meet fluctuating market demands. This adaptability is crucial for reducing lead time for high-purity antibiotics, ensuring that patients have timely access to life-saving medications.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that can be recovered and recycled, aligning with modern green chemistry principles. The waste streams generated are primarily organic and can be treated using standard industrial wastewater protocols, reducing the environmental footprint of production. The avoidance of heavy metal catalysts in the main coupling steps, reserving palladium only for the final hydrogenation where it can be efficiently filtered and recovered, further supports environmental compliance. This sustainable approach not only meets regulatory requirements but also appeals to environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbapenem derivatives. The answers are derived from the detailed experimental data and structural analysis provided in the patent documentation, offering clarity on the feasibility and performance of the technology. Understanding these aspects is essential for stakeholders evaluating the integration of these intermediates into their existing portfolios.
Q: What is the primary advantage of these new carbapenem derivatives over Meropenem?
A: The primary advantage is a significantly extended plasma half-life, approximately 2 to 3 times longer than Meropenem or Doripenem, allowing for reduced dosing frequency while maintaining broad-spectrum antibacterial activity.
Q: Are these intermediates stable for large-scale storage?
A: Yes, the intermediates, particularly the protected esters and salts described in the patent, demonstrate stability suitable for industrial handling, with specific crystallization protocols outlined to ensure purity and shelf-life.
Q: What is the key reaction mechanism used in the final coupling step?
A: The final coupling utilizes a nucleophilic substitution reaction where the thiol group of the pyrrolidine side chain attacks the carbapenem core, typically displacing a diphenyl phosphate leaving group under basic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenem Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of complex antibiotic intermediates. Our facility is equipped with state-of-the-art reactors and purification systems capable of handling the sensitive chemistry required for carbapenem synthesis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch of benzenesulfonamide carbapenem derivatives meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce costs through our advanced manufacturing capabilities. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your next project. Together, we can accelerate the delivery of next-generation antibiotics to the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
