Scalable Synthesis of Thienamycin Derivatives for Global Antibiotic Production
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, with carbapenems remaining a critical line of defense against resistant bacterial strains. A pivotal advancement in this domain is detailed in patent CN101367830B, which discloses a highly efficient synthetic method for thienamycin derivatives. This technology addresses longstanding bottlenecks in the production of carbapenem intermediates by streamlining the construction of the characteristic bicyclic nucleus. By leveraging a novel application of the intramolecular Wittig reaction using commercially available reagents, this route offers a compelling alternative to traditional, more cumbersome methodologies. For global stakeholders in the fine chemical sector, understanding this process is essential for securing reliable supply chains of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the carbapenem skeleton has relied heavily on two primary strategies: intramolecular carbene insertion and intramolecular Wittig reactions using custom-synthesized reagents. While carbene insertion offers mild conditions, it is often plagued by extended synthetic routes that result in diminished overall yields, making it less attractive for cost-sensitive commercial manufacturing. Alternatively, traditional Wittig approaches frequently necessitate the prior preparation of specific phosphorus ylides, such as diethyl methylphosphonate or ethyl diethyl phosphite. These reagents are not typically available as bulk commodities and must be synthesized in dedicated steps involving toxic raw materials and harsh reaction conditions. This dependency introduces significant supply chain vulnerabilities, increases the environmental footprint due to additional waste generation, and escalates the overall cost of goods sold for the final antibiotic active pharmaceutical ingredient.
The Novel Approach
The methodology presented in the referenced patent fundamentally shifts this paradigm by utilizing commercial triethyl phosphite directly as the Wittig reagent. This strategic substitution eliminates the need for the independent, multi-step synthesis of specialized phosphorus compounds. The process initiates with the acylation of a readily available azetidinone precursor, followed immediately by the cyclization step. This reduction in step count not only accelerates the production timeline but also minimizes the accumulation of impurities that often occur during intermediate isolation and purification. By simplifying the chemical architecture of the synthesis, manufacturers can achieve a more robust and economically viable process for generating thienamycin derivatives, positioning this method as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
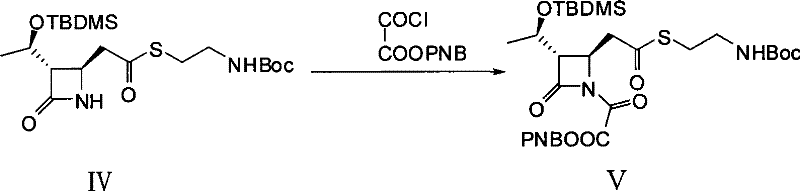
Mechanistic Insights into Intramolecular Wittig Cyclization
The core of this synthetic innovation lies in the mechanistic efficiency of the intramolecular Wittig reaction facilitated by triethyl phosphite. In the initial activation phase, the nitrogen atom of the azetidinone ring is acylated using oxalyl chloride mono-p-nitrobenzyl ester. This transformation generates a highly reactive imide-like intermediate (Compound V), which primes the molecule for the subsequent cyclization. The use of the p-nitrobenzyl group is particularly strategic, as it serves as a robust protecting group that can withstand the reaction conditions while facilitating the necessary electronic activation for the nucleophilic attack by the phosphite species.
Upon introduction of triethyl phosphite in the presence of a hydroquinone catalyst, the phosphorus center attacks the activated carbonyl carbon, initiating a cascade that results in the formation of the carbon-carbon double bond within the newly formed five-membered ring. This intramolecular closure constructs the critical 1-azabicyclo[3.2.0]hept-2-ene skeleton characteristic of carbapenems. The reaction proceeds under reflux conditions in solvents such as xylene or toluene, ensuring sufficient thermal energy to drive the elimination of the phosphate byproduct. This mechanism is notable for its high stereoselectivity and ability to preserve the sensitive beta-lactam ring integrity, which is often a point of failure in less optimized synthetic routes.
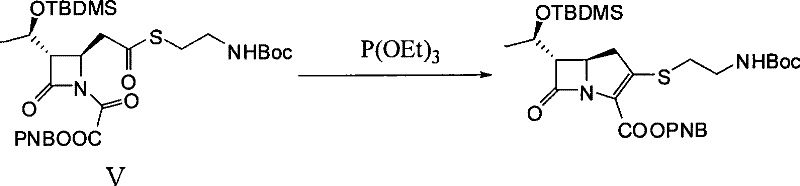
How to Synthesize Thienamycin Derivative Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the key intermediate, Compound IV, which itself is derived from commercially available 4-acetoxy-2-azetidinone through allylation, oxidation, and esterification. Once Compound IV is secured, the novel two-step sequence described in the patent can be implemented. The acylation step must be conducted under strictly anhydrous conditions to prevent hydrolysis of the acid chloride species, while the subsequent Wittig cyclization demands an oxygen-free environment to protect the phosphite reagent from oxidation. Detailed standardized operating procedures for these transformations are critical for reproducibility.
- Perform acylation of the azetidinone starting material (Compound IV) with oxalyl chloride mono-p-nitrobenzyl ester in anhydrous toluene at 25-30°C to generate the activated intermediate (Compound V).
- Conduct an intramolecular Wittig cyclization by reacting Compound V with commercial triethyl phosphite and hydroquinone catalyst in xylene or toluene under reflux conditions.
- Purify the resulting crude thienamycin derivative using flash chromatography with a dichloromethane and acetone solvent system to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents tangible strategic benefits that extend beyond simple chemical yield. The primary driver of value is the drastic simplification of the raw material portfolio. By relying on commodity chemicals like triethyl phosphite rather than custom-synthesized specialty reagents, organizations can mitigate the risk of supply disruptions and leverage established vendor networks for better pricing. Furthermore, the reduction in synthetic steps directly correlates to a reduction in processing time and resource consumption, including solvents, energy, and labor hours required for intermediate workups.
- Cost Reduction in Manufacturing: The elimination of the Wittig reagent synthesis step removes a significant cost center associated with toxic precursors and specialized equipment. This qualitative improvement in process efficiency translates to substantial cost savings in pharmaceutical intermediate manufacturing without compromising product quality. Additionally, the use of common solvents like toluene and xylene allows for easier solvent recovery and recycling, further enhancing the economic profile of the production cycle.
- Enhanced Supply Chain Reliability: Dependence on niche reagents often creates single points of failure in the supply chain. By shifting to commercially available triethyl phosphite, the process becomes more resilient to market fluctuations. The starting materials for the precursor synthesis, such as allyl bromide and zinc powder, are also widely produced industrial chemicals, ensuring that the entire workflow remains robust and capable of sustaining continuous production schedules even during periods of raw material scarcity.
- Scalability and Environmental Compliance: The reaction conditions described, specifically the moderate temperatures and the absence of highly hazardous reagents in the key cyclization step, facilitate safer scale-up operations. This aligns with modern environmental, health, and safety (EHS) standards, reducing the burden of waste treatment and regulatory compliance. The streamlined nature of the route minimizes the generation of complex waste streams, making it an environmentally preferable option for large-scale commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thienamycin derivative synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear framework for evaluating the technology's fit within existing manufacturing portfolios. Understanding these nuances is vital for technical teams assessing feasibility.
Q: What is the primary advantage of using triethyl phosphite in this synthesis?
A: The primary advantage is the elimination of the need to synthesize specialized Wittig reagents like diethyl methylphosphonate in-house. Triethyl phosphite is a commercially available commodity chemical, which significantly simplifies the supply chain, reduces raw material costs, and avoids the handling of toxic precursors required for custom reagent synthesis.
Q: How does this method improve upon traditional carbene insertion routes?
A: Traditional intramolecular carbene insertion routes often suffer from longer synthetic sequences and lower overall yields. This patented method utilizes a concise 2-step sequence from the key azetidinone precursor, offering milder reaction conditions and a more direct path to the bicyclic core, thereby enhancing process efficiency and scalability.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly suitable for scale-up. It employs robust solvents like toluene and xylene, operates at manageable temperatures (reflux for cyclization, ambient for acylation), and utilizes stable, commercially sourced reagents. These factors minimize operational risks and facilitate the transition from laboratory benchtop to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thienamycin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of antibiotic production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN101367830B can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of carbapenem intermediate meets the exacting standards required for downstream API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific economic benefits tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and secure a reliable source of high-purity pharmaceutical intermediates for your global operations.
