Advanced Synthesis of 2-Hydroxy-5-Nonylacetophenone for High-Efficiency Copper Extraction
Introduction to Patented Synthesis Technology
The global demand for efficient hydrometallurgical reagents continues to drive innovation in the synthesis of key intermediates like 2-hydroxy-5-nonylacetophenone (HNA). As detailed in patent CN102206148A, a groundbreaking synthesis process has been developed that fundamentally alters the production landscape for this critical copper extractant precursor. This technology replaces cumbersome multi-step procedures with a streamlined, high-yield pathway that leverages the reactivity of nitriles under Lewis acid catalysis. By shifting away from traditional acylation methods, manufacturers can now achieve total yields exceeding 80 percent while maintaining product purity levels at or above 85 percent. This technical breakthrough not only addresses long-standing efficiency bottlenecks but also aligns with modern green chemistry principles by reducing waste and simplifying downstream processing. For industry leaders seeking a reliable mining chemical supplier, understanding the mechanistic advantages of this patented route is essential for optimizing supply chains and reducing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-hydroxy-5-nonylacetophenone has been plagued by significant technical hurdles inherent to older synthetic methodologies. One prevalent traditional route involves the use of magnesium methylate reacting with paraldehyde, a process notorious for generating gelatinous byproducts that severely complicate stirring and heat transfer in large-scale reactors. 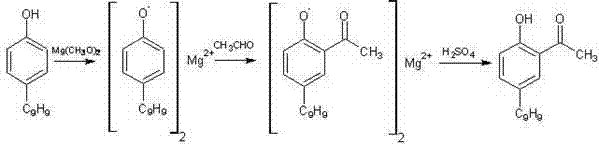 Furthermore, alternative pathways utilizing Friedel-Crafts acylation with excess acetyl chloride often result in complex mixtures of regioisomers and poly-acylated byproducts, making purification arduous and yield inconsistent. Another common approach, the Fries rearrangement of esters, introduces severe safety hazards due to the handling of corrosive acetyl chloride and requires rigorous temperature control to prevent decomposition. These legacy methods collectively suffer from poor atom economy, difficult workup procedures, and an inability to consistently meet the high purity standards required for premium copper extraction applications.
Furthermore, alternative pathways utilizing Friedel-Crafts acylation with excess acetyl chloride often result in complex mixtures of regioisomers and poly-acylated byproducts, making purification arduous and yield inconsistent. Another common approach, the Fries rearrangement of esters, introduces severe safety hazards due to the handling of corrosive acetyl chloride and requires rigorous temperature control to prevent decomposition. These legacy methods collectively suffer from poor atom economy, difficult workup procedures, and an inability to consistently meet the high purity standards required for premium copper extraction applications.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a direct addition reaction between 4-nonylphenol and anhydrous acetonitrile, catalyzed by Lewis acids such as aluminum chloride or zinc chloride in the presence of dry hydrogen chloride gas. 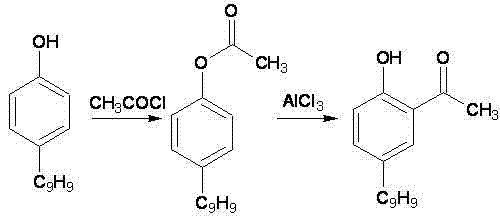 This innovative strategy bypasses the formation of unstable esters or reactive organometallic species, instead proceeding through a stable ketoimine intermediate that precipitates out of the reaction mixture. This physical separation step is a game-changer, as it allows for the removal of soluble impurities prior to the final hydrolysis step, drastically simplifying the purification process. The reaction conditions are milder and more controllable, typically operating between 30 to 100 degrees Celsius, which enhances operational safety and equipment longevity. By condensing what was previously a multi-step sequence into a more direct transformation, this method significantly shortens the synthetic timeline and improves the overall mass balance of the production facility.
This innovative strategy bypasses the formation of unstable esters or reactive organometallic species, instead proceeding through a stable ketoimine intermediate that precipitates out of the reaction mixture. This physical separation step is a game-changer, as it allows for the removal of soluble impurities prior to the final hydrolysis step, drastically simplifying the purification process. The reaction conditions are milder and more controllable, typically operating between 30 to 100 degrees Celsius, which enhances operational safety and equipment longevity. By condensing what was previously a multi-step sequence into a more direct transformation, this method significantly shortens the synthetic timeline and improves the overall mass balance of the production facility.
Mechanistic Insights into Lewis Acid Catalyzed Nitrile Addition
The core of this technological advancement lies in the precise activation of the nitrile group by strong Lewis acids and proton sources. In the presence of dry hydrogen chloride and a catalyst like zinc chloride or aluminum chloride, the nitrogen atom of the acetonitrile becomes highly electrophilic, facilitating a nucleophilic attack by the electron-rich aromatic ring of the 4-nonylphenol. This electrophilic aromatic substitution proceeds ortho to the hydroxyl group due to the directing effects of the phenolic oxygen, forming a stable imine salt intermediate known as nonyl phenol ketoimine. The stability of this intermediate is crucial; unlike transient species in other reactions, this imine salt has low solubility in the reaction medium, causing it to crystallize or precipitate upon cooling. This phenomenon effectively drives the equilibrium forward according to Le Chatelier's principle, ensuring high conversion rates of the starting materials. The subsequent hydrolysis step, performed by refluxing the isolated solid in hot water, cleanly converts the imine functionality into the desired ketone while regenerating the phenolic hydroxyl group, yielding the target 2-hydroxy-5-nonylacetophenone with minimal structural degradation.
From an impurity control perspective, this mechanism offers distinct advantages over competing technologies. In traditional Friedel-Crafts reactions, the high reactivity of the acylating agent often leads to over-acylation or polymerization, creating a complex soup of impurities that are difficult to separate via distillation or crystallization. However, in this patented process, the formation of the ketoimine acts as a built-in purification gate. Since many potential side products do not form the same insoluble imine salt structure, they remain in the mother liquor during the filtration step. This means that before the final product is even generated, a significant portion of organic impurities is physically discarded with the filtrate. Furthermore, the use of hot water for hydrolysis serves as a secondary purification step, dissolving inorganic salts and polar byproducts while leaving the organic product amenable to extraction. This dual-stage purification capability ensures that the final distilled product meets stringent purity specifications without the need for expensive chromatographic techniques or multiple recrystallizations.
How to Synthesize 2-Hydroxy-5-Nonylacetophenone Efficiently
The practical implementation of this synthesis route is designed for robustness and scalability, making it ideal for industrial adoption. The process begins by charging a dry reactor with an appropriate organic solvent, such as tetrahydrofuran or toluene, followed by the addition of 4-nonylphenol and anhydrous acetonitrile. A Lewis acid catalyst is introduced, and dry hydrogen chloride gas is bubbled through the mixture to initiate the formation of the ketoimine intermediate. Once the reaction is complete, the mixture is cooled to induce precipitation, allowing the intermediate to be filtered and washed. The solid cake is then transferred to a hydrolysis vessel where it is refluxed with hot water, converting the imine to the final ketone.
- React 4-nonylphenol with anhydrous acetonitrile in an organic solvent (e.g., THF, Toluene) using a Lewis acid catalyst (AlCl3, ZnCl2) and dry HCl gas to form the ketoimine intermediate.
- Cool the reaction mixture to precipitate the nonyl phenol ketoimine, then filter and wash the solid to remove soluble impurities.
- Hydrolyze the filtered intermediate by refluxing in hot water, followed by organic solvent extraction and vacuum distillation to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route represents a strategic opportunity to optimize costs and mitigate supply risks. The elimination of complex organometallic reagents and hazardous acyl chlorides simplifies the raw material portfolio, relying instead on widely available commodity chemicals like acetonitrile and hydrogen chloride. This shift reduces dependency on specialized suppliers and minimizes the logistical challenges associated with transporting dangerous goods. Moreover, the simplified workup procedure, characterized by a straightforward filtration and hydrolysis sequence, significantly reduces the consumption of solvents and energy compared to the extensive extraction and purification cycles required by older methods. These operational efficiencies translate directly into lower manufacturing overheads and a more competitive pricing structure for the final product.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in processing steps and the avoidance of expensive reagents. By eliminating the need for moisture-sensitive Grignard reagents or corrosive acetyl chloride, the process reduces the cost of raw materials and the specialized equipment needed to handle them. The ability to isolate the intermediate via filtration also minimizes solvent usage during purification, as the bulk of impurities are removed mechanically rather than through repeated washing or distillation. Consequently, the overall cost of goods sold is significantly lowered, providing a substantial margin advantage in the competitive market for mining chemicals.
- Enhanced Supply Chain Reliability: Supply continuity is greatly improved due to the use of stable, shelf-stable raw materials that are produced on a massive global scale. Unlike processes that require freshly prepared catalysts or reagents with short shelf lives, the inputs for this synthesis—4-nonylphenol, acetonitrile, and Lewis acids—are standard industrial chemicals with robust supply networks. This reduces the risk of production stoppages due to material shortages or quality variability. Additionally, the robustness of the reaction against minor fluctuations in conditions ensures consistent batch-to-batch quality, further stabilizing the supply chain for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is superior because it avoids the generation of gelatinous wastes that can foul reactors and piping systems. The solid-state isolation of the intermediate allows for easier handling in large vessels, facilitating a smooth scale-up from pilot plants to full commercial production. Furthermore, the reduced use of hazardous chlorinating agents and the simpler aqueous workup result in a cleaner waste stream that is easier and cheaper to treat. This alignment with environmental regulations reduces compliance costs and enhances the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-hydroxy-5-nonylacetophenone using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific operational needs.
Q: Why does the new synthesis route offer better yield than traditional Friedel-Crafts acylation?
A: The novel route avoids the harsh conditions and excessive byproduct formation associated with traditional Friedel-Crafts acylation using acetyl chloride. By forming a stable ketoimine intermediate that can be physically separated via filtration before hydrolysis, the process effectively removes impurities early, leading to a total yield exceeding 80%.
Q: What are the safety advantages of this method over the Fries rearrangement route?
A: Unlike the Fries rearrangement which requires hazardous acetyl chloride and generates significant byproducts during rearrangement, this process utilizes safer commodity reagents like acetonitrile and hydrogen chloride gas. This significantly reduces operational risks and simplifies the purification workflow.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable because it eliminates the gelation issues common in Grignard-type syntheses. The ability to isolate the intermediate as a solid allows for robust quality control and easier handling in large reactors, ensuring consistent purity above 85%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-5-Nonylacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficiency of hydrometallurgical operations. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patented synthesis are fully realized at an industrial level. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-hydroxy-5-nonylacetophenone performs optimally in copper extraction applications. Our state-of-the-art facilities are equipped to handle the specific requirements of Lewis acid catalyzed reactions, maintaining the anhydrous conditions necessary for maximum yield and consistency.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term production goals. Let us be your partner in driving efficiency and reliability in the mining chemicals sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
