Advanced Dual-Basic Ionic Liquid Catalysts for High-Yield Chalcone Manufacturing
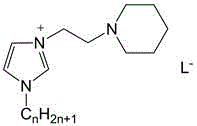
The landscape of fine chemical synthesis is undergoing a transformative shift towards greener, more efficient catalytic systems, a trend exemplified by the innovations detailed in patent CN110590742B. This pivotal intellectual property introduces a novel application of N1-long-chain alkyl-N3-piperidine ethyl imidazolium salts, a specialized class of ionic liquids that function as dual-basic catalysts for the synthesis of chalcone compounds. Unlike traditional methods that rely on harsh inorganic bases or expensive metal complexes, this technology leverages the unique amphiphilic nature of these ionic liquids to facilitate aldol condensation reactions in aqueous media. The breakthrough lies in the catalyst's ability to combine surface activity with dual basicity, effectively overcoming the limitations of previous ionic liquid systems which often suffered from high dosage requirements or low yields. For R&D directors and process chemists, this represents a significant opportunity to streamline the production of high-purity chalcone compounds, which serve as critical scaffolds in pharmaceutical intermediates and agrochemical applications. By enabling reactions to proceed with yields exceeding 85% under mild conditions, this patent outlines a pathway to substantial operational efficiency and environmental compliance.
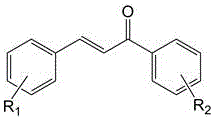
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of chalcone derivatives has been plagued by significant technical and economic bottlenecks inherent to conventional catalytic strategies. Traditional approaches often utilize inorganic acids or bases, which, while inexpensive, impose severe corrosion risks on reactor equipment and generate large volumes of saline wastewater that require costly treatment protocols. Alternatively, the use of metal-organic compounds or transition metal catalysts introduces the risk of heavy metal contamination in the final product, necessitating complex and expensive purification steps to meet stringent pharmaceutical purity standards. Furthermore, methods employing microwave or ultrasonic assistance, while potentially faster, demand high capital expenditure for specialized equipment and often rely on volatile organic solvents like tetrafluoroborate, which pose safety and environmental hazards. Even earlier generations of ionic liquid catalysts faced challenges; composite systems involving ionic liquids paired with strong acids or bases retained corrosive properties, while single-component basic ionic liquids often required excessive loading amounts to achieve acceptable conversion rates, thereby inflating raw material costs and complicating downstream separation processes.
The Novel Approach
The methodology disclosed in patent CN110590742B fundamentally redefines the catalytic landscape by introducing a surface-active, dual-basic ionic liquid system that operates efficiently in water. This novel approach utilizes N1-long-chain alkyl-N3-piperidine ethyl imidazolium salts, which possess a unique molecular architecture featuring a hydrophobic long-chain alkyl group and hydrophilic ionic headgroups. This amphiphilic structure imparts surfactant-like properties, enhancing the interfacial contact between the organic reactants and the aqueous reaction medium without the need for additional phase transfer agents. The reaction proceeds by mixing substituted benzaldehyde and substituted acetophenone with water and the ionic liquid catalyst, followed by heating to moderate temperatures between 30-80°C. As illustrated in the reaction scheme below, this process not only simplifies the operational workflow but also ensures that the catalyst remains in the aqueous phase post-reaction, allowing for straightforward product isolation via ethyl acetate extraction and enabling the direct recycling of the catalytic solution.
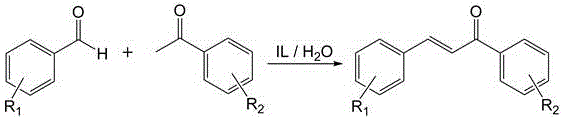
Mechanistic Insights into Dual-Basic Ionic Liquid Catalysis
The exceptional performance of this catalytic system is rooted in its sophisticated dual-basic mechanism, which synergistically activates the reactants through distinct electronic pathways. The ionic liquid cation, specifically the piperidine ethyl imidazolium moiety, exhibits Lewis basicity, while the accompanying anion (such as carboxylates, phenoxides, or carbonates) provides Bronsted basicity. This dual functionality allows the catalyst to simultaneously activate the carbonyl group of the acetophenone via Lewis acid-base interactions and deprotonate the alpha-carbon via Bronsted base mechanisms, significantly lowering the activation energy for the aldol condensation. Moreover, the long alkyl chain (where n=8-18) creates a micro-environment that concentrates the organic substrates near the catalytic sites, mimicking enzymatic pockets and driving the reaction equilibrium towards product formation. This structural design effectively addresses the issue of low mass transfer rates often observed in biphasic aqueous-organic systems, ensuring high turnover frequencies even at relatively low catalyst loadings ranging from 1:20 to 1:4 molar ratios relative to the aldehyde.
From an impurity control perspective, the mild reaction conditions afforded by this ionic liquid system are paramount for maintaining high product quality. Traditional strong base catalysis often promotes side reactions such as polymerization or over-condensation, leading to complex impurity profiles that are difficult to separate. In contrast, the tunable basicity of the ionic liquid allows for precise control over the reaction kinetics, minimizing the formation of by-products and ensuring a clean crude product profile. The use of water as the primary solvent further suppresses thermal runaway risks and eliminates the introduction of solvent-derived impurities. Consequently, the resulting chalcone compounds exhibit high purity levels suitable for direct use in sensitive applications like API synthesis or optical material fabrication, reducing the burden on downstream purification units and aligning with the rigorous quality expectations of a reliable pharmaceutical intermediates supplier.
How to Synthesize Chalcone Compounds Efficiently
The implementation of this green synthesis route offers a streamlined protocol that balances high efficiency with operational simplicity, making it highly attractive for commercial scale-up of complex organic intermediates. The process begins with the preparation of the specific ionic liquid catalyst, followed by the direct addition of stoichiometric amounts of substituted benzaldehyde and acetophenone into an aqueous reaction vessel. Detailed standard operating procedures regarding temperature ramping, stirring speeds, and workup protocols are critical for maximizing the benefits of this technology. For a comprehensive guide on executing this synthesis with optimal parameters, please refer to the standardized protocol outlined below.
- Mix substituted benzaldehyde and substituted acetophenone with water and add the N1-long-chain alkyl-N3-piperidine ethyl imidazolium salt ionic liquid catalyst.
- Heat the reaction mixture to 30-80°C and stir for 1-12 hours to complete the aldol condensation.
- Extract the product with ethyl acetate, dry and concentrate the organic phase, and recycle the aqueous catalyst phase for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-catalyzed process translates into tangible strategic advantages that extend beyond mere technical feasibility. The shift from corrosive inorganic reagents to a reusable, non-corrosive ionic liquid system drastically reduces maintenance costs associated with reactor lining and replacement, while the elimination of volatile organic solvents in favor of water significantly lowers waste disposal fees and regulatory compliance burdens. This technological leap facilitates cost reduction in fine chemical manufacturing by simplifying the supply chain for raw materials and reducing the dependency on expensive metal catalysts that are subject to market volatility. Furthermore, the robustness of the catalyst, which maintains activity over multiple cycles, ensures a stable and predictable production schedule, mitigating the risks of batch failures that can disrupt global supply chains.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the elimination of costly downstream purification steps typically required to remove heavy metal residues from traditional catalysts. By utilizing a metal-free ionic liquid system, manufacturers can bypass expensive scavenging resins and chromatography columns, leading to substantial cost savings in processing time and consumables. Additionally, the ability to recycle the aqueous catalyst phase for at least six consecutive cycles without significant yield degradation means that the effective cost per kilogram of catalyst is amortized over a much larger production volume. This efficiency, combined with the use of water as a cheap and abundant solvent, creates a highly competitive cost structure for producing high-purity chalcone compounds.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and stability of the raw materials involved. The precursors—substituted benzaldehydes and acetophenones—are commodity chemicals with well-established global supply networks, reducing the risk of sourcing bottlenecks. The catalyst itself, being a synthetic organic salt, does not rely on scarce precious metals like palladium or platinum, insulating the production process from geopolitical supply shocks. Moreover, the mild reaction conditions (30-80°C) reduce energy consumption and allow for operation in a wider range of manufacturing facilities, enhancing the flexibility of the supply network and reducing lead time for high-purity chalcone compounds during periods of high demand.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is facilitated by the inherent safety of the aqueous system, which eliminates fire hazards associated with flammable organic solvents. The green chemistry credentials of this method, characterized by atom economy and reduced E-factor (waste-to-product ratio), align perfectly with increasingly stringent environmental regulations globally. This compliance advantage future-proofs the manufacturing asset against tightening emissions standards and carbon taxes. The ease of product isolation via simple extraction and crystallization ensures that scale-up does not introduce complex engineering challenges, allowing for rapid capacity expansion to meet market needs for pharmaceutical and agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid technology, derived directly from the experimental data and beneficial effects reported in the patent literature. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this green chemistry solution into their existing manufacturing portfolios. The answers reflect the specific performance metrics and operational parameters validated in the patent examples.
Q: What are the advantages of using dual-basic ionic liquids over traditional base catalysts?
A: Dual-basic ionic liquids offer surface activity and tunable basicity, allowing for reactions in green solvents like water with yields exceeding 85%, while avoiding the corrosion issues associated with inorganic bases.
Q: Can the ionic liquid catalyst be reused in chalcone synthesis?
A: Yes, the aqueous phase containing the catalyst can be separated after extraction and reused for at least 6 cycles without significant loss in catalytic activity or yield.
Q: What types of substituents are tolerated in this synthesis method?
A: The method is robust against various substituents including nitro, chloro, hydroxyl, alkyl, and alkoxy groups at different positions on the benzene rings of both aldehyde and ketone precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chalcone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the dual-basic ionic liquid system described in CN110590742B to redefine the production standards for fine chemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required by the global pharmaceutical industry. We are uniquely positioned to leverage this green synthesis route to deliver cost-effective, high-quality intermediates that support your drug development pipelines.
We invite you to collaborate with us to optimize your supply chain and capitalize on the efficiencies offered by this next-generation catalytic method. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to reach out to our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your transition to this sustainable manufacturing platform is smooth, compliant, and commercially advantageous.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
