Advanced Catalytic Synthesis of 4-Chloro-4'-Hydroxybenzophenone for Scalable Pharmaceutical Manufacturing
Advanced Catalytic Synthesis of 4-Chloro-4'-Hydroxybenzophenone for Scalable Pharmaceutical Manufacturing
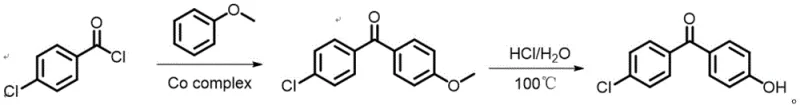
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high yield with environmental sustainability, particularly for critical intermediates like 4-chloro-4'-hydroxybenzophenone (4-CBP). As detailed in the recent patent CN114920637A, a groundbreaking preparation process has been developed that fundamentally shifts the paradigm from traditional stoichiometric Lewis acid catalysis to a reusable, heterogeneous cobalt-based system. This innovation addresses the long-standing challenges of equipment corrosion and hazardous waste generation inherent in the production of hypolipidemic drug precursors. By leveraging a porous microporous material [Co3(HCOO)6], manufacturers can now achieve superior conversion rates while drastically simplifying downstream processing. This report analyzes the technical merits and commercial viability of this novel approach for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4-chloro-4'-hydroxybenzophenone has relied heavily on aluminum chloride (AlCl3) as the primary catalyst for Friedel-Crafts acylation. While chemically effective, this legacy methodology imposes severe operational burdens on manufacturing facilities. The stoichiometric requirement for AlCl3 generates massive quantities of hydrochloric acid waste and aluminum-containing sludge during the quenching phase, creating significant environmental compliance hurdles. Furthermore, the highly corrosive nature of the reaction mixture necessitates expensive glass-lined or Hastelloy reactors, driving up capital expenditure. Traditional protocols often utilize toxic solvents such as nitrobenzene or chlorobenzene, which pose serious occupational health risks and require complex abatement systems. Additionally, the separation of the catalyst from the product matrix is notoriously difficult, often leading to product discoloration and requiring multiple recrystallization steps to meet purity specifications, thereby eroding overall process efficiency and yield.
The Novel Approach
In stark contrast, the innovative process disclosed in patent CN114920637A utilizes a specialized porous microporous cobalt formate framework, [Co3(HCOO)6], to catalyze the acylation of anisole with 4-chlorobenzoyl chloride. This heterogeneous catalyst functions effectively under mild conditions, typically between 25°C and 60°C, and can be easily recovered via simple filtration for repeated cycles without significant loss of activity. The reaction can proceed under solvent-free conditions or with minimal amounts of benign solvents like acetone or DMF, eliminating the need for large volumes of hazardous organic media. Following the acylation, the intermediate 4-chloro-4'-methoxybenzophenone undergoes a straightforward demethoxylation using dilute hydrochloric acid in water, yielding the final product with exceptional purity. This streamlined workflow not only enhances safety but also significantly reduces the physical footprint and utility consumption of the manufacturing process.
Mechanistic Insights into Co-Catalyzed Friedel-Crafts Acylation
The efficacy of the [Co3(HCOO)6] catalyst stems from its unique structural properties, where cobalt ions act as potent Lewis acid centers within a rigid three-dimensional framework. In this catalytic cycle, the cobalt centers coordinate with the carbonyl oxygen of the 4-chlorobenzoyl chloride, significantly enhancing the electrophilicity of the carbonyl carbon. This activation lowers the energy barrier for the nucleophilic attack by the electron-rich anisole ring, facilitating the formation of the C-C bond with high regioselectivity. Unlike homogeneous catalysts that dissolve into the reaction medium, the microporous structure of the cobalt formate provides defined channels that restrict the orientation of substrate molecules. This spatial confinement effect minimizes side reactions such as polyacylation or isomerization, ensuring that the reaction proceeds cleanly towards the desired para-substituted intermediate. The stability of the coordination bonds within the framework ensures that the catalyst maintains its integrity throughout the reaction, preventing metal leaching into the product stream.
Impurity control is inherently built into this mechanistic design, offering a distinct advantage over traditional methods where metal salts often contaminate the final API intermediate. Because the catalyst is a solid heterogeneous material, it can be physically separated from the liquid reaction mixture by filtration immediately upon completion. This physical barrier prevents the formation of stable complexes between the catalyst and the product ketone, which is a common issue with AlCl3 that requires destructive hydrolysis to break. Consequently, the crude product obtained after solvent removal is already of high purity, often exceeding 99% as confirmed by liquid chromatography analysis. The subsequent hydrolysis step to remove the methoxy group uses aqueous acid, which is easily washed away, leaving behind a product free from heavy metal residues. This clean profile is critical for pharmaceutical applications where strict limits on elemental impurities are enforced by regulatory bodies.
How to Synthesize 4-Chloro-4'-Hydroxybenzophenone Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters to maximize the benefits of the cobalt catalyst. The process begins with the careful mixing of 4-chlorobenzoyl chloride and anisole, typically in a molar ratio ranging from 1.1:1 to 2.1:1, to ensure complete conversion of the limiting reagent. The addition of the [Co3(HCOO)6] catalyst, usually at 5-40% of the anisole mass, initiates the exothermic acylation, which must be managed to maintain the temperature within the optimal 25-60°C window. Once the intermediate is formed and isolated, the demethoxylation step is conducted in an aqueous environment with 6N hydrochloric acid, providing a green and cost-effective method to reveal the phenolic hydroxyl group. For a comprehensive breakdown of the standardized operating procedures and specific workup techniques, please refer to the detailed guide below.
- Conduct Friedel-Crafts acylation of anisole and 4-chlorobenzoyl chloride using [Co3(HCOO)6] catalyst at 25-60°C.
- Filter and recover the heterogeneous catalyst for reuse, then isolate the 4-chloro-4'-methoxybenzophenone intermediate.
- Perform demethoxylation of the intermediate using 6N hydrochloric acid in water to yield the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this cobalt-catalyzed process represents a strategic opportunity to optimize cost structures and mitigate supply risks. The elimination of stoichiometric aluminum chloride removes the logistical burden of handling and disposing of tons of corrosive waste acid, which translates directly into reduced waste treatment costs and lower regulatory compliance overhead. The ability to recycle the catalyst multiple times significantly lowers the raw material cost per kilogram of product, offering a sustainable economic model that is resilient to fluctuations in metal prices. Furthermore, the simplified post-processing workflow, which avoids complex extraction and purification sequences, shortens the overall production cycle time. This efficiency gain allows manufacturing facilities to increase throughput without expanding infrastructure, ensuring a more reliable supply of this critical pharmaceutical intermediate to meet market demand.
- Cost Reduction in Manufacturing: The shift from a stoichiometric to a catalytic process fundamentally alters the cost equation by reducing the consumption of expensive reagents and minimizing waste disposal fees. By replacing toxic solvents like nitrotoluene with recyclable alternatives or running the reaction solvent-free, the process drastically cuts down on solvent purchase and recovery costs. The high selectivity of the catalyst reduces the formation of by-products, which means less material is lost during purification, thereby improving the overall mass balance and yield. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Relying on a robust, heterogeneous catalyst system reduces the risk of production delays caused by equipment corrosion or maintenance issues associated with harsh acidic conditions. The mild reaction conditions allow for the use of standard stainless steel equipment in some stages, reducing dependency on specialized glass-lined reactors that often have long lead times for repair or replacement. Additionally, the raw materials—anisole and 4-chlorobenzoyl chloride—are commodity chemicals with stable global availability, ensuring that production schedules are not disrupted by raw material shortages. This stability is crucial for maintaining continuous supply lines to downstream pharmaceutical manufacturers who operate on tight just-in-time inventory models.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as atom economy and waste prevention, align perfectly with increasingly stringent environmental regulations worldwide. The absence of heavy metal contamination in the wastewater stream simplifies effluent treatment, allowing facilities to operate within stricter discharge limits without costly upgrades. The scalability of the fixed-bed or slurry reactor systems suitable for this heterogeneous catalyst ensures that production can be ramped up from pilot scale to multi-ton commercial volumes seamlessly. This adaptability supports long-term growth strategies and ensures that the manufacturing process remains viable and compliant as environmental standards evolve over the coming decades.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this novel synthetic pathway. These insights are derived directly from the experimental data and beneficial effects reported in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating technology transfer and for quality assurance teams establishing specification limits. We encourage stakeholders to review these points carefully to fully appreciate the operational improvements offered by this cobalt-catalyzed methodology.
Q: How does the [Co3(HCOO)6] catalyst improve upon traditional AlCl3 methods?
A: The porous cobalt catalyst eliminates the massive waste acid and corrosive smoke associated with aluminum chloride, allows for catalyst recovery and reuse, and operates under milder conditions with higher selectivity.
Q: What purity levels can be achieved with this new synthetic route?
A: The process consistently achieves liquid chromatography purity of 99% or higher, meeting stringent commercial standards for pharmaceutical intermediates without requiring complex purification steps.
Q: Is the solvent system environmentally sustainable?
A: Yes, the process can be conducted under solvent-free conditions or with recyclable green solvents like acetone or DMF, replacing toxic nitrotoluene and chlorobenzene used in legacy methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-4'-Hydroxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced catalytic technologies is key to maintaining a competitive edge in the fine chemical sector. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the [Co3(HCOO)6] catalyzed route are translated into reliable industrial reality. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to process excellence ensures that every batch of 4-chloro-4'-hydroxybenzophenone we supply adheres to the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for fenofibrate intermediates and other critical pharmaceutical building blocks. By leveraging our technical expertise, you can achieve Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive efficiency, sustainability, and profitability in your manufacturing operations through the adoption of next-generation synthetic chemistry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
