Advanced Green Synthesis of 2,4,5-Trichloropyrimidine for Scalable Commercial Production
Advanced Green Synthesis of 2,4,5-Trichloropyrimidine for Scalable Commercial Production
The global demand for high-performance heterocyclic compounds continues to surge, particularly within the sectors of agrochemicals and pharmaceutical intermediates where structural precision is paramount. A pivotal advancement in this domain is detailed in patent CN113912550A, which outlines a novel, environmentally benign method for preparing 2,4,5-trichloropyrimidine. This specific compound serves as a critical building block for novel active dyes and antibacterial agents, necessitating a supply chain that prioritizes both safety and efficiency. The disclosed technology represents a significant departure from traditional halogenation methods by substituting hazardous gaseous reagents with safer, liquid-phase alternatives. By leveraging sodium hypochlorite for the initial functionalization and bis(trichloromethyl) carbonate for subsequent activation, the process mitigates the severe safety risks associated with chlorine gas and phosgene. For R&D directors and procurement specialists, understanding this shift is essential for securing a reliable agrochemical intermediate supplier capable of meeting stringent regulatory standards while maintaining cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of polychlorinated pyrimidines has relied heavily on aggressive chlorinating agents such as elemental chlorine, thionyl chloride, phosphorus oxychloride, and phosgene. These conventional pathways, while chemically effective, impose substantial burdens on manufacturing infrastructure due to the extreme toxicity and corrosivity of the reagents involved. The handling of chlorine gas requires specialized containment systems and rigorous safety protocols to prevent accidental releases that could endanger personnel and the surrounding environment. Furthermore, the use of thionyl chloride and phosphorus oxychloride generates significant quantities of acidic waste gases like sulfur dioxide and hydrogen chloride, complicating waste treatment and increasing the overall environmental footprint of the production facility. These factors collectively drive up capital expenditure for safety equipment and operational costs for waste neutralization, making the traditional routes less attractive for modern, sustainability-focused chemical enterprises seeking cost reduction in agrochemical intermediate manufacturing.
The Novel Approach
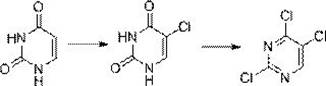
In stark contrast to these legacy methods, the innovative process described in the patent utilizes a two-step sequence that dramatically simplifies the operational landscape while enhancing safety profiles. The first stage employs sodium hypochlorite under acidic conditions to selectively chlorinate uracil at the 5-position, effectively replacing the need for direct chlorine gas injection. This is followed by a second stage where bis(trichloromethyl) carbonate, a solid and manageable substitute for phosgene, facilitates the conversion of hydroxyl groups to chlorides. This strategic substitution of reagents not only lowers the barrier to entry for safe production but also streamlines the workflow by utilizing reagents that are easier to store, transport, and dose with high precision. The result is a robust synthetic pathway that maintains high yields while aligning with green chemistry principles, thereby offering a compelling value proposition for supply chain heads focused on continuity and risk mitigation.
Mechanistic Insights into Electrophilic Chlorination and Activation
The core of this synthetic strategy lies in the controlled electrophilic substitution at the C5 position of the uracil ring, driven by the in situ generation of hypochlorous acid or related chlorinating species from sodium hypochlorite in an acidic medium. In the presence of sulfuric acid, the sodium hypochlorite creates a highly reactive chlorinating environment that targets the electron-rich double bond of the pyrimidine ring without causing excessive degradation of the heterocyclic core. This regioselectivity is crucial for ensuring that the resulting 5-chlorouracil intermediate is formed with minimal formation of poly-chlorinated byproducts or ring-opened impurities. The subsequent heating step to 95-100°C ensures the completion of the reaction and facilitates the crystallization of the intermediate, allowing for easy isolation via filtration. This level of control over the reaction trajectory is vital for R&D teams aiming to establish a reproducible process that consistently delivers high-purity 2,4,5-trichloropyrimidine suitable for downstream coupling reactions.
Following the isolation of the mono-chlorinated intermediate, the second mechanistic phase involves the activation of the carbonyl groups at positions 2 and 4 using bis(trichloromethyl) carbonate in the presence of a catalytic amount of N,N-dimethylformamide (DMF). This reaction likely proceeds through a Vilsmeier-Haack type mechanism where DMF reacts with the carbonate to generate a highly electrophilic iminium salt species. This activated complex then attacks the oxygen atoms of the uracil derivative, converting the carbonyl functionalities into good leaving groups that are subsequently displaced by chloride ions. The use of polar aprotic solvents such as chloroform or tetrahydrofuran is critical here, as they stabilize the transition states and ensure homogeneous mixing of the solid carbonate with the organic substrate. By maintaining the temperature between 60-65°C, the process optimizes the kinetic energy required for chlorination while preventing thermal decomposition, ultimately leading to a clean conversion with an HPLC purity often exceeding 98%.
How to Synthesize 2,4,5-Trichloropyrimidine Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the yield of both the intermediate and the final product. The process begins with the preparation of an acidic aqueous slurry where uracil is suspended and treated with a slight excess of sodium hypochlorite solution, typically ranging from 1.05 to 1.5 equivalents to ensure complete consumption of the starting material. Once the 5-chlorouracil is isolated and dried, it is dissolved in an organic solvent system containing a catalytic promoter, after which the solid chlorinating agent is introduced gradually to manage the exotherm. The detailed standardized synthetic steps, including specific workup procedures and purification techniques like vacuum rectification, are outlined in the guide below to assist technical teams in replicating these results.
- React uracil with sodium hypochlorite under acidic conditions (sulfuric acid) at 5-25°C, followed by heating to 95-100°C to isolate 5-chlorouracil.
- Dissolve 5-chlorouracil in a polar aprotic solvent like chloroform or tetrahydrofuran with a catalytic amount of DMF.
- Add bis(trichloromethyl) carbonate at 60-65°C to effect chlorination, then separate layers and concentrate to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical yield, fundamentally altering the cost structure and risk profile of the supply chain. By eliminating the reliance on gaseous chlorine and phosgene, facilities can significantly reduce the capital investment required for specialized corrosion-resistant reactors and gas scrubbing systems. This reduction in infrastructure complexity translates directly into lower overhead costs and a more agile production capability that can respond quickly to market fluctuations without being bottlenecked by hazardous material handling permits. Furthermore, the use of solid and liquid reagents simplifies logistics, as these materials can be transported and stored with far fewer regulatory hurdles compared to compressed toxic gases, thereby enhancing the overall reliability of the raw material supply.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous reagents with commodity chemicals like sodium hypochlorite and triphosgene derivatives leads to a substantial optimization of raw material costs. Since these reagents are widely available and do not require the same level of specialized containment as chlorine gas, the operational expenditure related to safety monitoring and emergency response is drastically simplified. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the load on downstream purification units and lowering solvent consumption during recrystallization or distillation steps. This holistic reduction in processing complexity ensures that the final cost of goods sold is competitive, providing a strong margin buffer for distributors and end-users alike.
- Enhanced Supply Chain Reliability: Sourcing 2,4,5-trichloropyrimidine from manufacturers utilizing this green chemistry approach ensures a more stable supply line that is less susceptible to disruptions caused by safety incidents or regulatory crackdowns on toxic emissions. The ease of handling the reagents means that production can be scaled up or down with greater flexibility, allowing suppliers to meet urgent demand spikes without the lead times associated with procuring restricted hazardous gases. This reliability is critical for pharmaceutical and agrochemical companies that operate on tight production schedules and cannot afford delays caused by raw material shortages or transportation restrictions on dangerous goods. Consequently, partnering with a supplier who has mastered this technology provides a strategic advantage in maintaining continuous manufacturing operations.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale industrial production, as evidenced by the successful demonstration in 500L reactors within the patent examples, indicating a clear path to multi-ton scale-up. The replacement of toxic gases with safer alternatives significantly reduces the generation of hazardous waste streams, making it easier for manufacturing sites to comply with increasingly stringent environmental regulations regarding air and water emissions. This environmental compliance not only protects the manufacturer from potential fines but also enhances the brand reputation of the entire supply chain by aligning with global sustainability goals. For supply chain heads, this means securing a long-term partner who can grow with their needs without facing existential threats from changing environmental laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,4,5-trichloropyrimidine, derived directly from the technical specifications and beneficial effects outlined in the patent literature. These insights are intended to clarify the operational advantages and quality standards associated with this specific synthetic methodology for potential partners and technical evaluators. Understanding these details is key to making informed decisions about integrating this intermediate into broader synthesis campaigns for active pharmaceutical ingredients or crop protection agents.
Q: What are the safety advantages of using sodium hypochlorite over chlorine gas?
A: Using sodium hypochlorite eliminates the need for handling highly toxic and corrosive chlorine gas cylinders, significantly reducing equipment corrosion risks and improving onsite operational safety for workers.
Q: Why is bis(trichloromethyl) carbonate preferred for the second chlorination step?
A: Bis(trichloromethyl) carbonate acts as a solid source of phosgene that is easier to handle and transport than gaseous phosgene, allowing for precise stoichiometric control and reduced environmental release.
Q: What purity levels can be achieved with this synthetic route?
A: The optimized process described in the patent demonstrates the ability to achieve HPLC purity levels exceeding 98% for the final 2,4,5-trichloropyrimidine product through standard workup and distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trichloropyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory requirement but a strategic imperative for modern chemical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN113912550A are fully realized in practical, large-volume outputs. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2,4,5-trichloropyrimidine meets the exacting standards required for sensitive downstream applications in drug discovery and agrochemical formulation. Our commitment to quality assurance means that clients receive a product with a consistent impurity profile, facilitating smoother regulatory filings and faster time-to-market for their final products.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized processes can reduce your total cost of ownership compared to traditional sourcing options. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capacity to serve as a dependable long-term partner in your supply chain for high-value heterocyclic intermediates.
