Revolutionizing 1-Oxacephalosporin Synthesis: A Scalable One-Pot Chemical Route for Global Supply Chains
The pharmaceutical industry constantly seeks robust manufacturing pathways that balance high purity with economic efficiency, particularly for complex beta-lactam antibiotics. Patent CN1980939B introduces a transformative production method for 1-oxacephalosporin-7α-methoxy-3-chloromethyl derivatives, which serve as critical precursors for potent antimicrobial agents like latamoxef and flomoxef. This technology addresses a longstanding bottleneck in cephalosporin chemistry by replacing hazardous and capital-intensive photo-irradiation steps with a streamlined, chemically driven one-pot reaction sequence. By leveraging a tandem halogenation-methoxylation-reduction strategy, this innovation allows manufacturers to bypass the isolation of unstable intermediates, thereby enhancing overall process safety and throughput. For global supply chain leaders, this represents a pivotal shift towards more reliable and cost-effective sourcing of high-value pharmaceutical intermediates, ensuring continuity in the production of life-saving antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 7α-methoxy-3-chloromethyl oxacephems relied heavily on photochemical reactions, which introduced significant operational friction and cost burdens. As illustrated in prior art, the traditional route required the exposure of 3-exomethylene compounds to chlorine gas followed by irradiation with light to induce 7α-methoxylation. This dependency on optical reaction equipment necessitated substantial capital expenditure and specialized maintenance, creating a barrier to entry for many manufacturers. Furthermore, these photochemical processes were plagued by inconsistent reaction kinetics and poor scalability, often resulting in suboptimal yields; for instance, earlier attempts using quinoline as a base reported yields as low as 9.6%, rendering the process economically unviable for large-scale production. The requirement to isolate intermediate compounds between steps further exacerbated material loss and increased solvent consumption, complicating waste management and extending production lead times significantly.
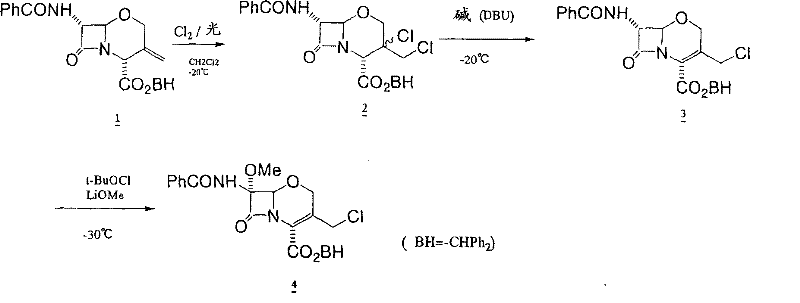
The Novel Approach
In stark contrast, the methodology disclosed in CN1980939B utilizes a sophisticated one-pot chemical cascade that completely eliminates the need for light irradiation, marking a significant leap in process engineering. The new route initiates with the chlorination of the 3-exomethylene starting material using chlorine gas in the presence of an aromatic amine base, followed immediately by methoxylation using lithium methoxide without isolating the chlorinated intermediate. This seamless transition is capped by a final reduction step using agents like sodium sulfite to secure the target 7α-methoxy-3-chloromethyl structure. By maintaining the reaction mixture throughout the sequence, the process avoids the thermal and mechanical stress associated with intermediate isolation, preserving the integrity of the sensitive beta-lactam ring. This approach not only simplifies the operational workflow but also dramatically improves mass balance, enabling yields that consistently exceed 75% and often reach above 90%, providing a robust foundation for commercial scale-up of complex pharmaceutical intermediates.
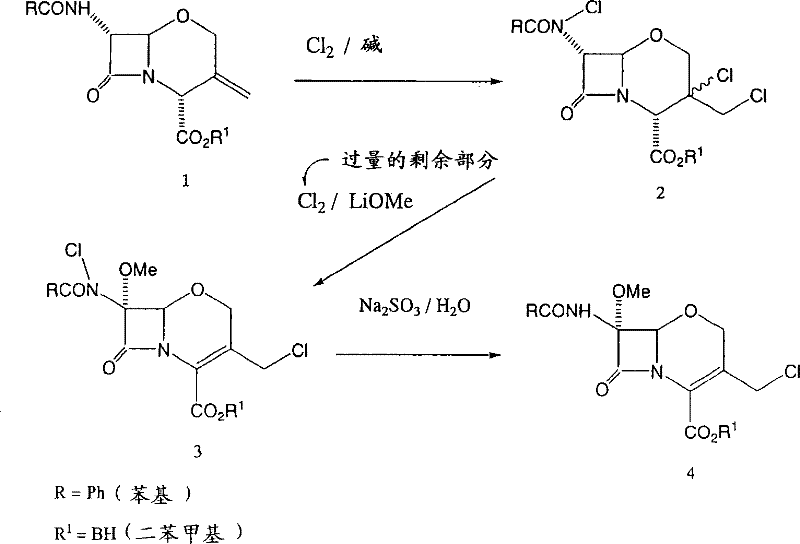
Mechanistic Insights into Tandem Halogenation-Methoxylation-Reduction
The core of this technological breakthrough lies in the precise control of reaction conditions that stabilize key intermediates which were previously prone to degradation. The first step involves the electrophilic addition of chlorine to the exomethylene double bond and the 7-position, facilitated by a base such as pyridine or collidine at controlled temperatures between 0°C and 5°C. Crucially, the process employs an excess of the halogenating agent (3 to 4 molar equivalents), which serves a dual purpose: driving the initial chlorination to completion and remaining in the solution to facilitate the subsequent methoxylation step. This strategic excess eliminates the need for recharging reagents or changing solvents, a common source of inefficiency in multi-step syntheses. The presence of the base also neutralizes generated hydrogen chloride, preventing acid-catalyzed decomposition of the starting material or the newly formed chlorinated species, thus ensuring a clean conversion to the dichloro-intermediate.
Following chlorination, the introduction of lithium methoxide at cryogenic temperatures (-40°C to -60°C) triggers a nucleophilic substitution that installs the critical 7α-methoxy group while retaining the 3-chloromethyl functionality. A key mechanistic advantage identified in this patent is the stability of the resulting N-chloro intermediate (Compound III) under these specific basic conditions; unlike previous methods where beta-lactam ring opening was a prevalent side reaction, this protocol maintains ring integrity. The final addition of a reducing agent, such as sodium sulfite or sodium thiosulfate, selectively reduces the N-Cl bond to an N-H bond without affecting other sensitive functional groups. This careful orchestration of redox and substitution chemistry ensures that the final product is obtained with high stereochemical purity and minimal impurity profiles, meeting the stringent quality standards required for active pharmaceutical ingredient (API) synthesis.
How to Synthesize 1-Oxacephalosporin-7α-methoxy-3-chloromethyl Derivative Efficiently
Implementing this synthesis route requires strict adherence to temperature gradients and reagent stoichiometry to maximize the benefits of the one-pot design. The process begins by dissolving the 3-exomethylene precursor in a chlorinated solvent like dichloromethane and cooling the mixture to 0°C before introducing chlorine gas and the organic base. Once the chlorination is confirmed via HPLC, the temperature is lowered to -40°C to -50°C for the dropwise addition of the lithium methoxide solution, a step that demands precise thermal control to prevent exothermic runaway and by-product formation. After the methoxylation phase, the reaction is quenched and reduced using an aqueous solution of sodium sulfite, followed by standard workup procedures involving extraction and crystallization. For detailed operational parameters, safety guidelines, and specific stoichiometric ratios tailored to your facility's capabilities, please refer to the standardized synthesis protocol provided below.
- React the 3-exomethylene starting compound with a halogenating agent (chlorine) in the presence of an aromatic amine base at 0°C to 5°C to form the chlorinated intermediate.
- Without isolating the intermediate, add lithium methoxide (LiOMe) at -40°C to -60°C in the presence of excess halogenating agent to introduce the 7α-methoxy group.
- Complete the sequence by adding a reducing agent such as sodium sulfite or sodium thiosulfate to reduce the N-Cl bond and isolate the final 7α-methoxy-3-chloromethyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this non-photochemical route offers profound advantages in terms of total cost of ownership and supply reliability. The elimination of photo-reactors removes a major capital expenditure item and reduces the maintenance burden associated with specialized optical equipment, directly lowering the barrier for manufacturing entry. Furthermore, the one-pot nature of the reaction significantly reduces solvent usage and waste generation, as there is no need for intermediate isolation, drying, or solvent swapping between steps. This consolidation of unit operations translates into shorter cycle times and reduced energy consumption for heating and cooling, contributing to a more sustainable and economically efficient manufacturing footprint. By minimizing the number of handling steps, the risk of cross-contamination and human error is also drastically reduced, ensuring a more consistent supply of high-quality intermediates.
- Cost Reduction in Manufacturing: The transition from a multi-step isolation process to a continuous one-pot reaction fundamentally alters the cost structure of producing oxacephem intermediates. By removing the need to isolate and purify the unstable chlorinated intermediate, manufacturers save significantly on labor, filtration media, and solvent volumes. The high yield profile, often exceeding 90%, means that less raw material is wasted per kilogram of final product, optimizing the utilization of expensive starting materials. Additionally, the avoidance of photo-irradiation equipment eliminates the depreciation costs and energy demands of high-intensity lamps, leading to substantial operational savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Reliance on photochemical processes often introduces bottlenecks due to equipment availability and batch-to-batch variability caused by light intensity fluctuations. The new chemical method relies on standard reactor vessels and well-understood reagents like chlorine and lithium methoxide, which are readily available in the global chemical market. This shift ensures that production schedules are not held hostage by specialized equipment maintenance or failure, providing a more predictable and resilient supply stream. The robustness of the reaction conditions also allows for easier technology transfer between manufacturing sites, securing the supply chain against regional disruptions.
- Scalability and Environmental Compliance: Scaling photochemical reactions from the laboratory to industrial tonnage is notoriously difficult due to the Beer-Lambert law limiting light penetration in large vessels. This chemical alternative scales linearly with reactor volume, making it ideal for commercial production ranging from hundreds of kilograms to multi-ton campaigns. Moreover, the reduction in solvent waste and the use of aqueous quenching steps align with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. This compliance advantage reduces the regulatory burden on manufacturers and future-proofs the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new production method is essential for stakeholders evaluating its integration into existing supply chains. The following questions address common concerns regarding process safety, impurity control, and downstream compatibility. These answers are derived directly from the experimental data and claims within the patent documentation, ensuring that the information provided is accurate and actionable for technical decision-makers. We encourage partners to review these details to fully appreciate the value proposition of this advanced synthetic route.
Q: Why is the one-pot method superior to photo-irradiation for oxacephem production?
A: The one-pot chemical method eliminates the need for expensive optical reaction equipment and avoids the low yields (often below 10%) associated with traditional photo-irradiation processes, significantly improving industrial feasibility.
Q: How does this process ensure high purity for pharmaceutical intermediates?
A: By controlling reaction temperatures strictly (0°C for chlorination, -40°C for methoxylation) and utilizing a novel stable intermediate (Compound III), the process prevents beta-lactam ring cleavage and minimizes by-product formation, achieving yields over 90%.
Q: Can this synthesis route be scaled for commercial antibiotic manufacturing?
A: Yes, the elimination of intermediate isolation steps, solvent exchanges, and photo-reactors makes this route highly scalable. It reduces operational complexity and waste generation, aligning perfectly with green chemistry principles for large-scale API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Oxacephalosporin-7α-methoxy-3-chloromethyl Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot synthesis are realized in practical, high-volume manufacturing. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex beta-lactam impurity profiles, guaranteeing that every batch of 1-oxacephalosporin intermediate meets the highest global pharmacopoeia standards. Our commitment to process excellence means we can deliver this critical antibiotic precursor with the consistency and quality required for regulated pharmaceutical markets.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this non-photochemical route for your specific portfolio. By collaborating with our technical procurement team, you can access specific COA data and route feasibility assessments tailored to your production needs. Let us help you optimize your supply chain for resilience and cost-efficiency, ensuring a steady flow of high-purity intermediates for your antibiotic manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
