Scalable Manufacturing of Novel Naphthoquinone Derivatives for Oncology Applications
Scalable Manufacturing of Novel Naphthoquinone Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds that offer superior efficacy in oncology treatment, particularly those capable of inhibiting critical enzymes like Topoisomerase II. Patent CN102924429B introduces a groundbreaking synthetic route for a specific class of 1,2-naphthoquinone derivatives, specifically 2-amino-4,5-dioxo-4,5-dihydro-naphtho[1,2-b]thiophene-3-ethyl formate. This compound represents a significant advancement in medicinal chemistry due to its unique fusion of a thiophene ring with a naphthoquinone core, coupled with an amino substituent that drastically improves water solubility compared to traditional analogues. The structural integrity and functional group arrangement of this molecule are critical for its biological activity, as evidenced by its potent inhibition of cancer cell proliferation. For R&D directors and procurement specialists, understanding the precise architecture of this molecule is the first step toward evaluating its potential as a lead compound or a key intermediate in complex drug synthesis pipelines. The visual representation of this core structure highlights the strategic placement of the ethyl ester and amino groups, which are essential for both metabolic stability and target binding affinity.
![Chemical structure of 2-amino-4,5-dioxo-4,5-dihydro-naphtho[1,2-b]thiophene-3-ethyl formate showing the fused ring system](/insights/img/naphthoquinone-derivative-synthesis-anticancer-supplier-20260315114736-01.png)
Beyond its biological promise, the true value of this patent lies in its manufacturability. Traditional methods for synthesizing substituted naphthoquinones often suffer from low yields, harsh conditions that degrade sensitive functional groups, or the use of prohibitively expensive reagents. The methodology outlined in CN102924429B circumvents these historical bottlenecks by employing a robust three-step sequence that balances chemical efficiency with operational simplicity. By utilizing a protection-oxidation-deprotection strategy, the process ensures that the sensitive amino moiety survives the oxidative conditions required to generate the quinone system. This approach not only safeguards the molecular integrity but also streamlines the purification process, resulting in a high-quality orange solid that meets stringent purity specifications required for preclinical and clinical development. For supply chain leaders, this translates to a reliable source of high-value intermediates without the volatility associated with fragile synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino-substituted naphthoquinones has been plagued by significant chemical challenges that hinder commercial viability. Conventional direct oxidation methods often fail to distinguish between the electron-rich aromatic systems and the nucleophilic amino groups, leading to extensive over-oxidation, polymerization, or the formation of intractable tar-like byproducts. Furthermore, many existing protocols rely on stoichiometric amounts of hazardous oxidants under extreme thermal conditions, which not only pose safety risks in a plant setting but also result in poor atom economy and difficult waste management. The lack of selectivity in these older methods frequently necessitates complex chromatographic separations that are impossible to scale beyond gram quantities, effectively rendering them useless for industrial API production. Additionally, the poor water solubility of many traditional naphthoquinone derivatives limits their bioavailability, requiring extensive formulation work that drives up development costs and extends time-to-market for new oncology therapies.
The Novel Approach
The innovative strategy presented in this patent fundamentally alters the synthetic landscape by introducing a temporary masking group prior to the critical oxidation step. By converting the free amine into a tert-butoxycarbonyl (Boc) protected derivative, the synthesis effectively neutralizes the nucleophilicity of the nitrogen atom, rendering it inert to the subsequent oxidative assault. This allows for the use of powerful oxidizing agents like Chromium trioxide to selectively transform the adjacent hydroquinone precursor into the desired 1,2-quinone system without compromising the amine functionality. Once the quinone core is established, the protecting group is cleanly removed under mild acidic conditions using trifluoroacetic acid at low temperatures, preserving the delicate quinone structure. This modular approach decouples the conflicting reactivity requirements of oxidation and amine preservation, resulting in a cleaner reaction profile, higher overall yields, and a product with enhanced physicochemical properties suitable for rigorous biological testing and eventual therapeutic application.
Mechanistic Insights into Boc-Protection and Chromium-Mediated Oxidation
The success of this synthetic route hinges on the precise orchestration of electronic effects during the oxidation phase. In the absence of the Boc protecting group, the lone pair electrons on the amino nitrogen would activate the aromatic ring towards electrophilic attack by the chromium species, likely leading to ring destruction or the formation of azo-compounds. However, the electron-withdrawing nature of the carbamate group reduces the electron density of the nitrogen, effectively shielding it while leaving the hydroquinone portion of the molecule available for oxidation. The mechanism involves the formation of a chromate ester intermediate with the phenolic hydroxyl groups, followed by an elimination step that establishes the carbonyl double bonds characteristic of the quinone. This transformation is thermodynamically driven and proceeds efficiently at moderate temperatures of 50-60°C, avoiding the thermal degradation pathways that often plague high-temperature oxidations. The careful control of stoichiometry, specifically maintaining a molar ratio of oxidant to substrate between 3:1 and 3.5:1, ensures complete conversion while minimizing the presence of residual heavy metals in the final crude mixture.
Following the oxidation, the deprotection step serves as a critical quality control checkpoint for impurity management. The use of trifluoroacetic acid (TFA) at 0°C is a deliberate choice to prevent acid-catalyzed decomposition of the newly formed quinone system, which can be sensitive to prolonged exposure to strong acids at elevated temperatures. The cleavage of the Boc group generates volatile byproducts (isobutylene and carbon dioxide) that are easily removed during the solvent evaporation stage, simplifying the downstream processing. This mechanism ensures that the final product, 2-amino-4,5-dioxo-4,5-dihydro-naphtho[1,2-b]thiophene-3-ethyl formate, is obtained with a high degree of chemical purity, free from Boc-related contaminants that could interfere with biological assays. For R&D teams, this level of mechanistic control provides confidence in the reproducibility of the synthesis, allowing for reliable structure-activity relationship (SAR) studies without the noise introduced by variable impurity profiles.
How to Synthesize 2-amino-4,5-dioxo-4,5-dihydro-naphtho[1,2-b]thiophene-3-ethyl formate Efficiently
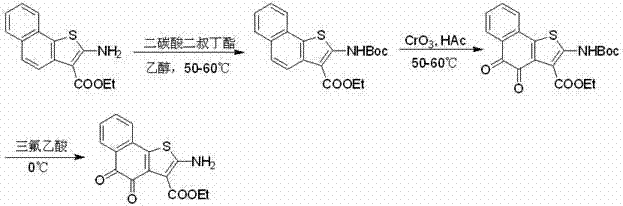
The practical execution of this synthesis is designed for scalability, utilizing standard reactor equipment and commonly available reagents found in most fine chemical facilities. The process begins with the dissolution of the amino-naphthothiophene precursor in absolute ethanol, followed by the addition of di-tert-butyl dicarbonate. This mixture is heated to a gentle reflux range of 50-60°C and stirred for approximately 20 to 24 hours to ensure complete conversion to the N-Boc protected intermediate. Upon completion, the solvent is removed, and the residue is subjected to oxidation using a solution of Chromium trioxide in 60% acetic acid, maintaining the temperature strictly between 50-60°C for 1.5 to 2 hours. The reaction mixture is then quenched in water and extracted with chloroform to isolate the oxidized intermediate. The final step involves treating the dried intermediate with trifluoroacetic acid at 0°C for 5 to 7 hours, followed by solvent removal and column chromatography purification to yield the target orange solid. The visual roadmap below illustrates this sequential transformation, highlighting the key intermediates and reagents involved in this robust protocol.
- Protect the amino group of the starting naphthothiophene ester using di-tert-butyl dicarbonate in ethanol at 50-60°C.
- Oxidize the protected intermediate using Chromium trioxide (CrO3) in acetic acid solution at controlled temperatures to form the quinone core.
- Remove the Boc protecting group using trifluoroacetic acid (TFA) at 0°C to reveal the final active amino-quinone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The reliance on commodity chemicals such as ethanol, acetic acid, and chromium trioxide eliminates the dependency on scarce or proprietary catalysts that often create supply bottlenecks and price volatility. The operational simplicity of the process, characterized by ambient pressure and moderate temperature requirements, reduces the energy consumption and capital expenditure associated with specialized high-pressure or cryogenic reactor setups. Furthermore, the high selectivity of the reaction minimizes the generation of complex byproduct mixtures, thereby reducing the load on waste treatment facilities and lowering the environmental compliance costs associated with hazardous waste disposal. These factors combine to create a manufacturing process that is not only cost-effective but also environmentally more sustainable than traditional alternatives, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive, bulk-grade solvents significantly lowers the raw material cost per kilogram of the final product. By avoiding complex multi-step sequences that require isolation of unstable intermediates, the process reduces labor hours and equipment occupancy time, leading to substantial operational savings. The high atom economy of the oxidation step ensures that a maximum proportion of the starting material is converted into the desired product, minimizing waste and maximizing yield efficiency. Additionally, the straightforward workup procedures involving simple extraction and evaporation reduce the need for costly preparative HPLC purification on a large scale, further driving down the cost of goods sold (COGS) for this high-value intermediate.
- Enhanced Supply Chain Reliability: The use of widely available reagents ensures that production schedules are not disrupted by the long lead times often associated with specialty chemicals. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, resulting in consistent batch-to-batch performance. This reliability is crucial for maintaining continuous supply lines to downstream drug developers who require steady volumes of intermediates for clinical trial material production. Moreover, the scalability of the protocol from gram to multi-kilogram scales allows suppliers to rapidly ramp up production in response to surging demand without the need for extensive process re-engineering or technology transfer delays.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently in larger reaction vessels without loss of yield or purity. The ability to perform the oxidation in acetic acid, a solvent that can be recovered and recycled, contributes to a reduced environmental footprint. While chromium reagents require careful handling, the defined stoichiometry and contained reaction environment facilitate effective waste management and adherence to strict regulatory standards. The final product's enhanced water solubility also offers downstream benefits in formulation, potentially reducing the need for toxic co-solvents in the final drug product, which is an increasingly important consideration for regulatory approval and market acceptance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel naphthoquinone derivative. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of incorporating this intermediate into their drug discovery pipelines or manufacturing portfolios. The answers reflect the balance between chemical innovation and practical industrial application that defines this synthetic approach.
Q: Why is the Boc-protection step critical in this naphthoquinone synthesis?
A: The amino group is highly susceptible to oxidation. Protecting it with a Boc group prevents side reactions and degradation during the harsh Chromium(VI) oxidation step, ensuring the integrity of the final quinone structure.
Q: What are the biological advantages of this specific naphthoquinone derivative?
A: This compound exhibits strong inhibition against Topoisomerase II and demonstrates significant cytotoxicity against cancer cell lines like MCF-7 and MT-4, while the amino group enhances water solubility for better bioavailability.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial solvents like ethanol and acetic acid, operates at moderate temperatures (50-60°C), and avoids exotic catalysts, making it highly adaptable for multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-amino-4,5-dioxo-4,5-dihydro-naphtho[1,2-b]thiophene-3-ethyl formate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the acceleration of oncology drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2-amino-4,5-dioxo-4,5-dihydro-naphtho[1,2-b]thiophene-3-ethyl formate meets the exacting standards required for preclinical and clinical research. Our commitment to technical excellence allows us to navigate the complexities of quinone chemistry, delivering products that empower your research teams to focus on breakthrough discoveries rather than supply chain uncertainties.
We invite you to collaborate with us to optimize your supply chain for this and other complex pharmaceutical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that validate the commercial viability of your projects. Contact us today to discuss how our advanced manufacturing capabilities can support your next generation of anticancer therapies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
