Scalable Synthesis of Lapatinib Intermediates Using Recyclable Heterogeneous Palladium Catalysts
The pharmaceutical industry continuously seeks robust synthetic routes for oncology therapeutics, particularly for tyrosine kinase inhibitors like lapatinib. Patent CN102321076B discloses a groundbreaking preparation method for lapatinib intermediates and their analogues, addressing critical bottlenecks in traditional manufacturing. This technology introduces a novel heterogeneous palladium catalyst system that fundamentally alters the efficiency and environmental profile of the synthesis. By shifting away from conventional homogeneous catalysis, this method offers a pathway to high-purity intermediates with significantly reduced metal contamination. For R&D teams and procurement strategists, understanding this shift is vital for optimizing supply chains and ensuring regulatory compliance in API production. The core innovation lies in the specific formulation of the catalyst, which combines diatomite, tin dichloride, and polyvinylpyrrolidone to create a stable, recyclable active site.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lapatinib bases and their derivatives has relied heavily on homogeneous noble metal catalysts, typically palladium complexes dissolved directly in the reaction medium. While effective for small-scale laboratory synthesis, these homogeneous systems present severe disadvantages for industrial application. The primary issue is the difficulty in separating the catalyst from the reaction mixture post-synthesis. This necessitates extensive downstream processing, including the use of expensive scavengers to remove trace heavy metals to meet strict pharmacopeial limits. Furthermore, the inability to recover the precious metal catalyst leads to substantial raw material waste and increased operational costs. Environmental regulations regarding heavy metal discharge also impose significant burdens on facilities utilizing non-recyclable homogeneous systems, creating a complex compliance landscape for manufacturers.
The Novel Approach
The methodology described in the patent overcomes these limitations by employing a specifically engineered heterogeneous Pd catalyst. This catalyst is prepared by heating a mixture of diatomite, tin dichloride, trifluoroacetic acid, H2PdCl4, and polyvinylpyrrolidone in water at temperatures between 100°C and 150°C. The resulting solid catalyst facilitates the crucial Suzuki coupling reaction between the quinazoline intermediate and 5-formylfuran boronic acid. Unlike its homogeneous counterparts, this solid catalyst can be easily separated via simple filtration, allowing for potential recycling and drastically reducing palladium leaching into the product stream. This approach not only protects the environment by minimizing waste but also simplifies the purification workflow, leading to a more cost-effective and sustainable manufacturing process for high-value pharmaceutical intermediates.
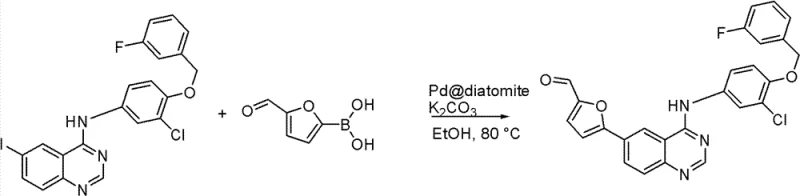
Mechanistic Insights into Heterogeneous Pd-Catalyzed Suzuki Coupling
The mechanistic superiority of this route centers on the stability and activity of the diatomite-supported palladium species. In the Suzuki coupling step, the reaction is conducted in a first organic solvent, preferably absolute ethanol, with potassium carbonate as the base at temperatures ranging from 70°C to 90°C. The diatomite support provides a high surface area that disperses the palladium active sites effectively, preventing agglomeration and maintaining catalytic activity over extended periods. The presence of polyvinylpyrrolidone acts as a stabilizer, further anchoring the metal particles to the support matrix. This structural integrity ensures that the catalytic cycle proceeds efficiently without significant loss of metal into the solution phase. Consequently, the reaction achieves high conversion rates while maintaining a clean impurity profile, which is critical for the subsequent reductive amination step.
Impurity control is inherently enhanced by the heterogeneous nature of the catalyst. In traditional homogeneous processes, palladium residues can catalyze unwanted side reactions or form difficult-to-remove organometallic complexes. By confining the palladium to the solid support, the likelihood of such side reactions is minimized. Additionally, the physical separation of the catalyst prevents the carryover of tin and other promoter residues into the final product, provided the washing steps are optimized. This results in a crude product that requires less rigorous purification to meet the stringent purity specifications demanded by regulatory bodies for oncology drugs. The robustness of this catalytic system also allows for flexibility in substrate scope, enabling the synthesis of various lapatinib analogues by modifying the starting aniline derivatives without compromising reaction efficiency.
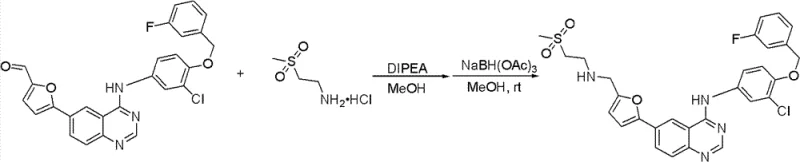
How to Synthesize Lapatinib Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear, four-step sequence that transforms readily available starting materials into the target lapatinib base or its analogues. The process begins with the nucleophilic substitution of 4-chloro-6-iodo-quinazoline with a substituted aniline derivative in isopropanol under reflux conditions. Following this, the heterogeneous catalyst is prepared in a separate aqueous step. The core carbon-carbon bond formation occurs via the Suzuki coupling described previously, linking the quinazoline core with the furan moiety. Finally, the aldehyde group on the furan ring undergoes reductive amination with 2-(methylsulfonyl)ethylamine hydrochloride using an organic base and a reducing agent like sodium borohydride. Detailed standardized synthesis steps see the guide below.
- Reflux compound of Formula I with 4-chloro-6-iodo-quinazoline in isopropanol to form the quinazoline core linkage.
- Prepare the heterogeneous Pd catalyst by mixing diatomite, tin dichloride, trifluoroacetic acid, H2PdCl4, and polyvinylpyrrolidone in water at 100-150°C.
- Perform Suzuki coupling between the quinazoline intermediate, 5-formylfuran boronic acid, and the Pd catalyst in organic solvent with K2CO3 at 70-90°C.
- Conduct reductive amination with 2-(methylsulfonyl)ethylamine hydrochloride and organic base to yield the final lapatinib base or analogue.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heterogeneous catalytic route offers tangible strategic benefits beyond mere technical feasibility. The elimination of complex heavy metal scavenging processes translates directly into reduced processing time and lower consumption of auxiliary chemicals. This streamlining of the manufacturing workflow enhances overall throughput and reduces the burden on waste treatment facilities. Furthermore, the ability to recover and potentially reuse the palladium catalyst mitigates the financial risk associated with the volatility of precious metal prices. These factors collectively contribute to a more resilient and cost-efficient supply chain for critical oncology intermediates.
- Cost Reduction in Manufacturing: The implementation of a recyclable heterogeneous catalyst fundamentally alters the cost structure of the synthesis. By avoiding the loss of palladium in the mother liquor and eliminating the need for expensive metal scavengers, the direct material costs are substantially lowered. Additionally, the simplified workup procedure reduces labor and utility costs associated with extended purification steps. This economic efficiency makes the process highly competitive for large-scale production, offering significant cost savings in pharmaceutical intermediate manufacturing without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent production capabilities, which is paramount for maintaining supply continuity in the pharmaceutical sector. The use of stable, commercially available reagents and mild reaction conditions minimizes the risk of batch failures due to sensitive operational parameters. This reliability allows for better inventory planning and reduces the lead time for high-purity intermediates. Suppliers adopting this technology can offer more dependable delivery schedules, thereby strengthening their partnerships with downstream API manufacturers and mitigating the risk of drug shortages.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles, facilitating easier regulatory approval and long-term sustainability. The reduction in hazardous waste generation simplifies compliance with increasingly strict environmental regulations. Moreover, the process is inherently scalable; the heterogeneous catalyst performs consistently across different batch sizes, from pilot plant to commercial tonnage. This scalability ensures that the technology can meet growing market demand for lapatinib and its analogues while maintaining a minimal environmental footprint, a key consideration for modern corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical implementation and benefits of the technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of the heterogeneous Pd catalyst in this synthesis?
A: The heterogeneous Pd catalyst, supported on diatomite, allows for easy recovery and recycling, significantly reducing palladium residue in the final product and lowering raw material costs compared to homogeneous systems.
Q: How does this method improve impurity control for pharmaceutical grades?
A: By utilizing a solid-supported catalyst, the process eliminates the need for complex heavy metal scavenging steps often required with homogeneous catalysts, thereby simplifying purification and ensuring stringent purity specifications are met.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method employs mild reaction conditions (70-90°C), common solvents like ethanol and isopropanol, and robust catalytic systems, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lapatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies in modern pharmaceutical synthesis. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN102321076B are successfully translated into industrial reality. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of lapatinib intermediate meets the exacting standards required for oncology drug development. Our commitment to technical excellence ensures that complex synthetic challenges are met with precision and reliability.
We invite global pharmaceutical partners to collaborate with us on optimizing their supply chains through advanced process chemistry. By leveraging our expertise in heterogeneous catalysis and scale-up engineering, we can help you achieve substantial cost savings and improved operational efficiency. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your commercial goals and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
