Advanced Silver-Catalyzed Synthesis of Hydroxyalkyl Quinoline Derivatives for Commercial Scale-Up
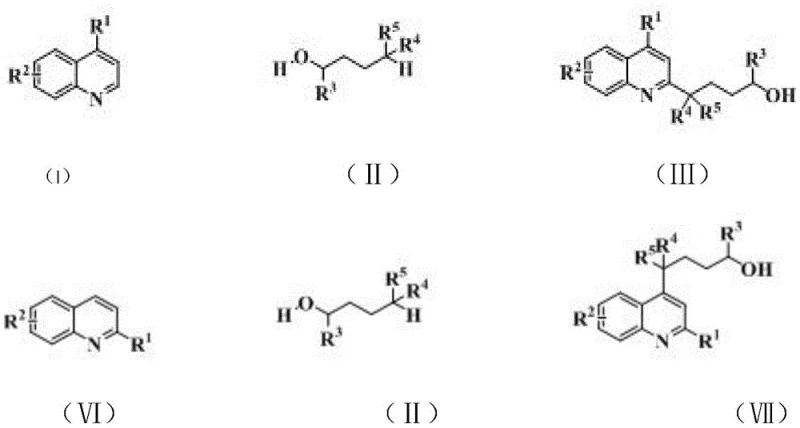
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic compounds, particularly quinoline derivatives, which serve as critical scaffolds for antimalarial, antiviral, and anticancer agents. A significant breakthrough in this domain is documented in Chinese Patent CN111960997B, which discloses a novel method for synthesizing hydroxyalkyl-substituted quinoline derivatives. This technology leverages a silver salt-catalyzed coupling reaction between quinoline compounds and long-chain aliphatic alcohols, operating under remarkably mild conditions. Unlike traditional methods that often rely on expensive transition metal catalysts or harsh reaction environments, this approach utilizes accessible silver salts such as silver nitrate or silver carbonate in conjunction with common oxidants. The result is a versatile platform capable of producing 2-position or 4-position substituted quinolines with high regioselectivity and yield. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more sustainable and cost-efficient manufacturing of complex pharmaceutical intermediates, addressing both the economic and environmental pressures facing modern chemical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of quinoline rings at the 2- or 4-positions has presented significant synthetic challenges, often requiring multi-step sequences or the use of pre-functionalized starting materials. Conventional alkylation strategies frequently depend on precious metal catalysts such as iridium or ruthenium complexes, which are not only prohibitively expensive but also pose supply chain risks due to their scarcity. Furthermore, many existing protocols necessitate the use of strong bases, anhydrous conditions, or elevated temperatures that can degrade sensitive functional groups on the alcohol substrates. The reliance on specialized ligands to control regioselectivity adds another layer of complexity and cost, making the scale-up of these processes economically unviable for bulk production. Additionally, the generation of heavy metal waste from these catalysts creates substantial environmental compliance burdens, requiring sophisticated waste treatment protocols that further inflate the operational expenditure of manufacturing facilities.
The Novel Approach
The methodology outlined in patent CN111960997B introduces a paradigm shift by employing earth-abundant silver salts as the primary catalytic species, effectively bypassing the need for rare precious metals. This innovative route facilitates the direct coupling of unactivated aliphatic alcohols with quinoline cores through a radical-mediated mechanism initiated by persulfate oxidants. The reaction proceeds efficiently in mixed solvent systems containing water, which not only reduces the consumption of volatile organic compounds but also simplifies the solubility profile of the inorganic salts involved. By operating at moderate temperatures ranging from 25°C to 70°C, the process preserves the integrity of thermally labile substituents, thereby expanding the scope of compatible substrates. This approach dramatically simplifies the operational workflow, allowing for the direct conversion of cheap, commercially available alcohols into high-value hydroxyalkyl quinoline intermediates with minimal purification steps required.
Mechanistic Insights into Silver-Catalyzed C-H Functionalization
The core of this synthetic advancement lies in the unique reactivity of the silver catalyst in conjunction with the oxidant system. The proposed mechanism involves the generation of carbon-centered radicals from the aliphatic alcohol substrate via hydrogen atom transfer facilitated by the silver-oxidant complex. These radicals subsequently attack the electron-deficient quinoline ring, preferentially at the C2 or C4 positions due to the stabilization of the resulting intermediate radical cation. The presence of a protonic acid additive plays a crucial dual role: it enhances the solubility of slightly soluble silver salts like silver carbonate, ensuring a homogeneous catalytic environment, and it activates the quinoline nitrogen, increasing the electrophilicity of the ring towards radical attack. This synergistic effect ensures high conversion rates and excellent regioselectivity, minimizing the formation of undesired isomers that typically plague free-radical alkylation reactions. The careful balance of oxidant strength and acid concentration allows for precise control over the reaction kinetics, preventing over-oxidation of the alcohol or degradation of the quinoline scaffold.
From an impurity control perspective, the choice of oxidant, such as potassium persulfate or sodium persulfate, is critical for maintaining a clean reaction profile. These oxidants decompose into benign sulfate byproducts, which are easily removed during the aqueous workup, unlike heavy metal residues that require complex chelation strategies. The reaction conditions are tuned to favor the formation of the desired hydroxyalkyl chain without affecting the terminal hydroxyl group, preserving the functionality needed for downstream derivatization into halides or esters. This chemoselectivity is paramount for pharmaceutical applications where strict impurity thresholds must be met. The robustness of the catalytic cycle against various substituents on the quinoline ring, including halogens and electron-donating groups like methoxy, demonstrates the versatility of this mechanism. Such tolerance allows medicinal chemists to rapidly generate diverse libraries of analogues for structure-activity relationship studies without redesigning the synthetic route for each new derivative.
How to Synthesize Hydroxyalkyl Quinoline Derivatives Efficiently
To implement this synthesis effectively, one must adhere to the optimized parameters defined in the patent data to ensure maximum yield and purity. The process begins with the precise stoichiometric mixing of the quinoline substrate and the alcohol compound in a reactor equipped with efficient stirring. The addition of the silver catalyst, oxidant, and acid additive must be managed to maintain the correct molar ratios, typically favoring an excess of the alcohol to drive the equilibrium forward. The reaction is conducted under an inert nitrogen atmosphere to prevent interference from atmospheric oxygen, although the oxidant provides the necessary oxygen equivalents for the transformation. Following the reaction period, a standardized workup procedure involving quenching with sodium bicarbonate and extraction with ethyl acetate ensures the isolation of the crude product. For detailed operational parameters and specific troubleshooting tips regarding solvent ratios and temperature gradients, refer to the structured guide below.
- Prepare the reaction mixture by combining the quinoline substrate, long-chain aliphatic alcohol, silver salt catalyst (e.g., AgNO3 or Ag2CO3), oxidant (e.g., K2S2O8), and protonic acid additive in a mixed organic/aqueous solvent system.
- Stir the reaction mixture under nitrogen protection at a controlled temperature between 25°C and 70°C for a duration of 6 to 24 hours to ensure complete coupling.
- Quench the reaction with sodium bicarbonate, perform liquid-liquid extraction with ethyl acetate, dry the organic layer, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver-catalyzed technology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. By replacing scarce and volatile precious metals like iridium with abundant silver salts, manufacturers can insulate their production costs from the fluctuations of the precious metals market. Furthermore, the use of simple aliphatic alcohols as alkylating agents eliminates the need for pre-activated halides or organometallic reagents, which are often hazardous and expensive to transport and store. This simplification of the bill of materials translates directly into improved margin stability and reduced logistical complexity for global supply chains.
- Cost Reduction in Manufacturing: The elimination of expensive ligand systems and precious metal catalysts results in significant cost savings per kilogram of produced intermediate. The process utilizes inexpensive oxidants and acids that are commodity chemicals, further driving down the variable cost of goods sold. Additionally, the ability to use water as a co-solvent reduces the volume of organic solvents required, lowering both procurement costs and waste disposal fees. The high selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall atom economy and reducing the cost burden associated with raw material consumption.
- Enhanced Supply Chain Reliability: Sourcing silver salts and aliphatic alcohols is far less risky than securing specialized transition metal complexes, which often have limited suppliers and long lead times. The robustness of the reaction conditions means that production is less susceptible to delays caused by stringent handling requirements or equipment failures associated with high-pressure or cryogenic processes. This reliability ensures a consistent flow of high-purity pharmaceutical intermediates to downstream API manufacturers, mitigating the risk of stockouts that can disrupt drug production schedules. The compatibility with standard stainless steel reactors further enhances supply chain resilience by allowing production to be easily transferred between different manufacturing sites without specialized infrastructure.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The use of water-miscible solvents and the generation of non-toxic sulfate byproducts simplify the wastewater treatment process, ensuring compliance with increasingly strict environmental regulations. This green chemistry profile reduces the regulatory burden on manufacturing facilities and lowers the capital expenditure required for pollution control equipment. The straightforward purification via silica gel chromatography or crystallization allows for rapid throughput, enabling manufacturers to respond quickly to market demand surges without compromising on product quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing production workflows.
Q: What are the key advantages of using silver salt catalysts over traditional transition metals for quinoline alkylation?
A: Silver salt catalysts offer a significantly more cost-effective alternative to precious metals like iridium or ruthenium while maintaining high activity under mild conditions. The process eliminates the need for expensive ligand systems and allows for the use of readily available aliphatic alcohols as alkylating agents, simplifying the supply chain and reducing raw material costs.
Q: How does this synthesis method improve product purity compared to conventional routes?
A: The method utilizes a specific combination of oxidants and protonic acid additives that enhance regioselectivity for the 2- or 4-positions on the quinoline ring. This specificity minimizes the formation of side-products and isomers, resulting in crude products with high HPLC purity (often exceeding 96%) prior to final purification, thereby reducing downstream processing burdens.
Q: Is this process scalable for industrial production of antimalarial intermediates?
A: Yes, the process is highly scalable due to its use of inexpensive reagents, moderate reaction temperatures (25-70°C), and water-compatible solvent systems. The absence of air-sensitive reagents or extreme pressure requirements makes it suitable for large-scale batch reactors, ensuring reliable supply continuity for critical pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxyalkyl Quinoline Supplier
The technological advancements described in patent CN111960997B highlight the immense potential of silver-catalyzed C-H functionalization for the efficient production of high-value quinoline derivatives. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the identity and purity of every batch, guaranteeing that our hydroxyalkyl quinoline intermediates are ready for immediate use in sensitive pharmaceutical applications. We understand the critical nature of supply chain continuity in the drug development lifecycle and are committed to delivering excellence in every shipment.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in silver-catalyzed synthesis can drive efficiency and value for your organization.
