Advanced Manufacturing of Vinorelbine Tartrate: A Breakthrough in Yield and Purity for Oncology APIs
The pharmaceutical industry constantly seeks robust manufacturing routes for complex oncology agents, and the recent disclosure in patent CN114524830B represents a significant leap forward in the production of vinorelbine tartrate. As a semi-synthetic vinca alkaloid, vinorelbine is a critical first-line treatment for non-small cell lung cancer and breast cancer, yet its intricate chemical architecture has historically posed severe challenges for mass production. The traditional reliance on labor-intensive chromatography has created bottlenecks in both yield and supply continuity. This new technical disclosure outlines a refined synthetic pathway that strategically replaces these inefficient purification steps with precise crystallization protocols. By optimizing reaction conditions—specifically temperature control and reagent stoichiometry—the process achieves a total molar yield approaching 60%, a substantial improvement over the roughly 30% yields seen in legacy methods. For global procurement teams, this signals a potential stabilization of supply chains for this high-value pharmaceutical intermediate. The following analysis dissects the chemical innovations that enable this efficiency, providing R&D and supply chain leaders with a clear view of the technology's commercial viability.
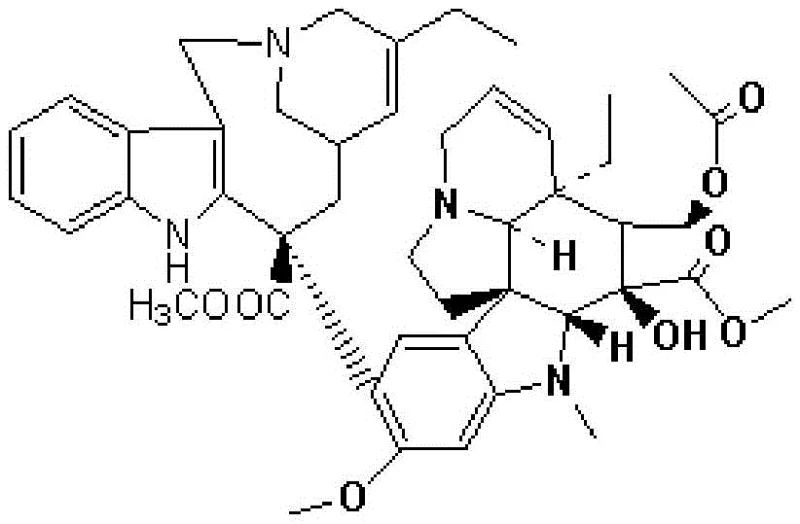
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of vinorelbine tartrate has been plagued by low efficiency and excessive processing times, primarily due to the dependence on column chromatography for purification. Prior art, such as the methods disclosed in CN 102199165A, typically involves converting vinorelbine to an N-oxide, coupling it to form a cationic dimer, and then proceeding through reduction and bromination. However, the critical flaw in these legacy routes is the necessity for multiple silica gel chromatography steps to isolate intermediates and the final product. Silica gel chromatography is inherently difficult to scale; it consumes vast quantities of organic solvents, requires significant manual intervention or complex automated systems, and often results in product loss due to irreversible adsorption or degradation on the stationary phase. Furthermore, the extended production cycle—often exceeding ten days for a single batch due to chromatography run times—severely limits manufacturing capacity. The instability of key intermediates, such as 7'-bromo-dehydrated vinblastine, under acidic or heated conditions during these prolonged processes further exacerbates yield losses, making the conventional approach economically unsustainable for meeting growing global demand.
The Novel Approach
The methodology presented in patent CN114524830B fundamentally reengineers the downstream processing to bypass these chromatographic bottlenecks. Instead of relying on silica gel columns for the final purification, this novel approach leverages a combination of alumina decolorization and sequential crystallization steps to achieve pharmaceutical-grade purity. The innovation lies not just in the purification but in the upstream reaction control that makes such purification possible. By meticulously controlling the synthesis of the cationic dimer and the subsequent reduction to dehydrated vinblastine, the process minimizes the formation of hard-to-remove impurities from the outset. This "quality by design" approach allows the crude vinorelbine to be purified effectively without the need for destructive column chromatography. The result is a streamlined workflow where the final product is obtained through crystallization of the free base followed by salt formation with tartaric acid. This shift from separation-based purification to crystallization-based purification is a hallmark of mature, scalable chemical manufacturing, offering a direct path to cost reduction in API manufacturing while simultaneously improving the environmental footprint by reducing solvent waste.
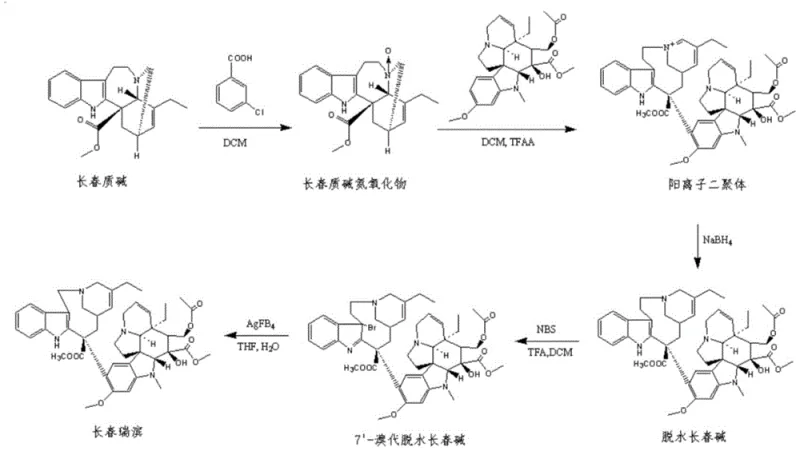
Mechanistic Insights into Cryogenic Reduction and Selective Bromination
The core chemical breakthroughs in this patent revolve around the precise manipulation of reaction thermodynamics to suppress specific side reactions. In the synthesis of dehydrated vinblastine (Step 2), the patent specifies dissolving the cationic dimer at temperatures between -30°C and -10°C, followed by cooling to -65°C ± 5°C before the addition of sodium borohydride. This extreme cryogenic condition is not arbitrary; it is essential for kinetic control. At higher temperatures, the cationic dimer reacts with the alcohol solvent (methanol or ethanol) to form ether by-products, which subsequently convert into the notorious "RRT0.72" impurity during later stages. This impurity is structurally similar to the target molecule and notoriously difficult to separate. By maintaining the reaction at -65°C, the nucleophilic attack by the solvent is effectively halted, ensuring that the sodium borohydride selectively reduces the iminium ion to the desired amine. Additionally, the patent highlights a drastic reduction in the equivalent of sodium borohydride used (down to roughly 1.1 equivalents), which minimizes the formation of boron ester by-products that could complicate downstream workups.
Similarly, the bromination step (Step 3) demonstrates sophisticated control over electrophilic substitution selectivity. Conventional methods often operate at extremely low temperatures (-90°C to -60°C) with high loads of trifluoroacetic acid (TFA) to drive the reaction. However, the new process operates at a warmer -45°C ± 5°C while significantly reducing the TFA ratio to between 0.047 and 0.113 relative to the substrate. This counter-intuitive adjustment actually enhances selectivity. Excessive acidity and extreme cold in prior methods promoted non-selective bromination, leading to the formation of the "17-bromo-vinorelbine" impurity. By moderating the acidity and temperature, the reaction favors bromination at the desired 7' position. Furthermore, the quenching protocol involves adding methanol prior to the bicarbonate wash, acting as an antifreeze to prevent local freezing and peracid formation that could degrade the sensitive bromo-intermediate. These mechanistic refinements ensure that the crude product entering the purification stage is already of high purity, validating the feasibility of skipping silica gel chromatography.
How to Synthesize Vinorelbine Tartrate Efficiently
The synthesis of vinorelbine tartrate via this patented route requires strict adherence to temperature profiles and intermediate monitoring to ensure the high yields reported. The process begins with the coupling of catharanthine-N-oxide and vindoline, followed by the critical cryogenic reduction and selective bromination steps detailed above. The elimination of column chromatography relies entirely on the success of these upstream controls; if impurity levels exceed thresholds, the crystallization steps will fail to deliver pharmaceutical-grade material. Therefore, real-time HPLC monitoring is integrated into the workflow to determine exact reaction endpoints, ensuring that starting materials are consumed without over-reacting to form degradation products. The following guide outlines the standardized operational framework derived from the patent examples, serving as a baseline for process engineers looking to implement this technology.
- Synthesize the cationic dimer by reacting catharanthine-N-oxide with vindoline in dichloromethane using trifluoroacetic anhydride at controlled low temperatures.
- Perform cryogenic reduction of the cationic dimer to dehydrated vinblastine using sodium borohydride at -65°C to minimize side reactions.
- Execute selective bromination at -45°C with reduced trifluoroacetic acid to form 7'-bromo-dehydrated vinblastine, followed by cyclization and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chromatography-dependent synthesis to crystallization-based manufacturing offers profound strategic benefits beyond simple yield metrics. The most immediate impact is on the cost structure of the final API. Silica gel chromatography is a resource-intensive operation that demands high-purity solvents, expensive stationary phases, and significant energy for solvent recovery. By eliminating these steps, the manufacturing process becomes significantly leaner. The reduction in solvent volume directly correlates to lower raw material costs and reduced expenses associated with hazardous waste disposal. Moreover, the shortened production cycle means that manufacturing assets (reactors, dryers) have higher turnover rates. A process that previously took ten days can now be completed in a fraction of that time, effectively increasing the annual production capacity of existing facilities without the need for capital expenditure on new equipment. This efficiency gain translates into a more competitive pricing structure for the final high-purity vinorelbine tartrate.
- Cost Reduction in Manufacturing: The elimination of multi-step silica gel chromatography removes one of the most expensive unit operations in fine chemical synthesis. Chromatography requires large volumes of high-grade organic solvents and specialized media, both of which carry high procurement and disposal costs. By shifting to crystallization, the process utilizes standard, scalable unit operations that are far more economical. The reduction in sodium borohydride equivalents and trifluoroacetic acid usage further lowers the bill of materials. Additionally, the higher overall molar yield (nearly doubling from ~30% to ~60%) means that less starting material (vindoline and catharanthine) is required to produce the same amount of final product, drastically reducing the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on production lead times. Traditional methods with long chromatography runs create bottlenecks that make it difficult to respond to sudden spikes in demand. The streamlined nature of this new process allows for faster batch completion, enabling manufacturers to maintain lower inventory levels while still meeting delivery commitments. The robustness of the crystallization steps also ensures consistent batch-to-batch quality, reducing the risk of failed batches that can disrupt supply schedules. For buyers of pharmaceutical intermediates, this reliability is crucial for maintaining their own production schedules for finished dosage forms.
- Scalability and Environmental Compliance: Scaling up column chromatography from pilot to commercial scale is technically challenging and often requires disproportionate increases in equipment footprint. In contrast, crystallization and filtration are linear processes that scale predictably from grams to tons. This makes the technology ideal for commercial scale-up of complex oncology intermediates. From an environmental perspective, the significant reduction in solvent usage aligns with green chemistry principles and increasingly stringent environmental regulations. Lower solvent consumption means reduced VOC emissions and a smaller carbon footprint, helping pharmaceutical companies meet their sustainability goals while securing a compliant supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is vital for R&D teams evaluating technology transfer and procurement teams assessing supplier capabilities. The answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance to industrial application.
Q: How does the new process improve purity compared to traditional methods?
A: The patented process eliminates multi-step silica gel chromatography, which often leads to product degradation. By optimizing reaction temperatures (e.g., -65°C for reduction) and reagent ratios, it significantly reduces difficult-to-remove impurities like RRT0.72 and 17-bromo-vinorelbine, allowing purification via simple crystallization.
Q: What are the key cost drivers reduced in this manufacturing route?
A: The primary cost reduction comes from removing extensive column chromatography steps. This drastically lowers solvent consumption, reduces silica gel/alumina usage, and shortens the production cycle from over ten days to a much shorter timeframe, enhancing overall throughput.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the replacement of chromatographic purification with crystallization and alumina decolorization makes the process highly scalable. Crystallization is a standard unit operation in GMP facilities, ensuring consistent quality and easier regulatory compliance compared to batch chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinorelbine Tartrate Supplier
The technological advancements detailed in patent CN114524830B underscore the complexity and precision required to manufacture high-value oncology intermediates efficiently. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such sophisticated laboratory processes into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of cryogenic reactions and crystallization kinetics is maintained at scale. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of vinorelbine tartrate meets the exacting standards required for global pharmaceutical registration.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain needs. By leveraging our optimized manufacturing protocols, we can help you reduce lead times and secure a stable supply of this critical API intermediate. Contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments that demonstrate how our capabilities align with your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
