Advanced Immobilized Transaminase Technology for Efficient Sitagliptin Intermediate Production
The pharmaceutical industry continuously seeks robust biocatalytic solutions to enhance the efficiency of synthesizing complex active pharmaceutical ingredients (APIs). Patent CN115466756A introduces a significant advancement in this domain by disclosing novel immobilized transaminases specifically engineered for the preparation of sitagliptin and its key intermediates. This technology addresses critical limitations associated with traditional liquid enzyme systems, particularly regarding stability in organic solvents and reusability. By leveraging specific amino acid mutations at the 241st position of the transaminase sequence and immobilizing them on epoxy resins, the invention achieves superior catalytic performance. This breakthrough offers a compelling value proposition for manufacturers aiming to optimize their production lines for DPP-4 inhibitors, ensuring higher yields and more sustainable processing conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the biocatalytic synthesis of sitagliptin has relied heavily on liquid transaminase systems or earlier generations of immobilized enzymes that exhibited suboptimal stability under industrial conditions. Prior art, such as the methods disclosed in US8293507 and US9617573, often required stringent control over solvent water content, typically necessitating water-saturated isopropyl acetate (IPAc) to maintain enzyme activity. This requirement introduces significant operational complexity, as controlling water activity in large-scale reactors is challenging and prone to variability. Furthermore, liquid enzymes cannot be easily recovered, leading to high consumption costs and complicated downstream purification steps to remove protein residues from the final product. The instability of previous immobilized variants in high concentrations of organic solvents further restricted substrate loading capacities, limiting overall process efficiency and throughput.
The Novel Approach
The technology described in CN115466756A overcomes these hurdles through a dual strategy of protein engineering and advanced immobilization. By introducing specific point mutations, such as R241A, R241H, or R241N, into the transaminase backbone, the inventors have created variants that retain high activity even when covalently bound to resin supports like SEPABEADS EC-HFA. This novel approach allows the use of a simpler aqueous isopropanol solvent system, eliminating the need for pre-saturating solvents with water. The resulting immobilized biocatalyst demonstrates exceptional robustness, maintaining structural integrity and catalytic function over multiple reaction cycles. This shift not only simplifies the operational workflow but also drastically reduces the environmental footprint by minimizing waste generation associated with single-use enzyme preparations.
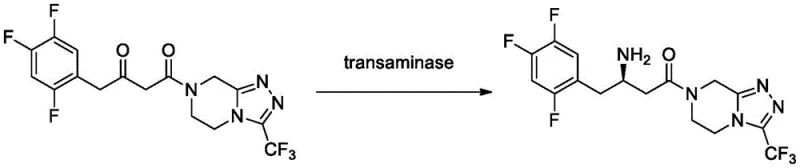
Mechanistically, the enhanced performance stems from the stabilization of the enzyme's tertiary structure upon immobilization, which prevents denaturation in the presence of organic cosolvents. The transamination reaction proceeds via a ping-pong bi-bi mechanism involving the cofactor pyridoxal phosphate (PLP). In this cycle, the amino group from the donor, typically isopropylamine, is transferred to the PLP to form pyridoxamine phosphate, which then transfers the amino group to the ketoamide substrate to generate the chiral amine product. The specific mutations at position 241 likely optimize the local electrostatic environment or steric hindrance near the active site, facilitating better substrate access or product release. Additionally, the covalent attachment to the epoxy resin ensures that the enzyme remains fixed within the reactor, preventing leaching and allowing for straightforward filtration and reuse. This mechanistic stability is crucial for maintaining high enantiomeric excess (ee > 99.9%) throughout the production campaign.
Impurity control is another critical aspect where this technology excels. The high stereoselectivity of the engineered transaminase minimizes the formation of the unwanted (S)-enantiomer, which is a difficult-to-remove impurity in downstream crystallization steps. Furthermore, the robustness of the immobilized system reduces the likelihood of enzyme degradation products contaminating the reaction mixture. By operating at moderate temperatures (30-60°C) and using a clean solvent system, the process avoids side reactions such as non-enzymatic hydrolysis or racemization that can occur under harsher chemical conditions. This results in a cleaner crude product profile, reducing the burden on purification units and ensuring that the final API intermediate meets stringent regulatory specifications for chirality and purity.
How to Synthesize Sitagliptin Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for implementing this technology in a pilot or production setting. The process begins with the preparation of the immobilized enzyme, followed by the setup of the reaction mixture with precise control over substrate concentration and amino donor ratios. The simplicity of the aqueous isopropanol system makes it highly adaptable to existing infrastructure. Detailed standard operating procedures for enzyme immobilization and reaction execution are essential for reproducibility. For a comprehensive guide on the specific parameters and step-by-step execution, please refer to the standardized synthesis protocol below.
- Prepare the immobilized transaminase by contacting the enzyme solution containing specific R241 mutations with SEPABEADS EC-HFA epoxy resin under controlled pH and temperature conditions.
- Set up the reaction system using an aqueous isopropanol solvent containing the ketoamide substrate, isopropylamine as the amino donor, and pyridoxal phosphate as a cofactor.
- Conduct the transamination reaction at 30-60°C with stirring, allowing for multiple reuse cycles of the immobilized enzyme while maintaining high conversion rates and stereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this immobilized transaminase technology translates into tangible strategic benefits beyond mere technical performance. The ability to reuse the biocatalyst over multiple batches fundamentally alters the cost structure of the synthesis. Unlike liquid enzymes that are consumed in a single run, the immobilized form acts as a durable processing aid, significantly lowering the cost of goods sold (COGS) related to biocatalysts. Moreover, the simplified solvent system reduces the complexity of solvent recovery and disposal, contributing to substantial cost reduction in pharmaceutical manufacturing. The robustness of the enzyme also means fewer batch failures due to catalyst deactivation, ensuring a more predictable and reliable supply of critical intermediates.
- Cost Reduction in Manufacturing: The primary economic driver here is the reusability of the immobilized enzyme. Since the biocatalyst can be recovered and reused for numerous cycles without significant loss of activity, the effective cost per kilogram of product attributed to the enzyme is drastically reduced. Additionally, the elimination of complex solvent saturation steps reduces energy consumption and labor hours associated with solvent preparation. The high conversion rates achieved minimize the amount of unreacted starting material that needs to be recovered or disposed of, further enhancing the overall material efficiency of the process.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the variability of biological reagents. The enhanced stability of these immobilized variants ensures consistent performance across different production lots, reducing the risk of supply disruptions. The use of commercially available resins and standard solvents like isopropanol means that raw material sourcing is straightforward and less susceptible to geopolitical or logistical bottlenecks. This reliability allows for tighter production scheduling and reduced safety stock requirements for critical intermediates.
- Scalability and Environmental Compliance: Scaling biocatalytic processes can be challenging due to mass transfer limitations and enzyme stability issues. This technology addresses scalability by providing a solid catalyst that is easy to handle in large reactors. The reduced use of hazardous solvents and the generation of less biological waste align with green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. This sustainability profile is becoming a key differentiator for suppliers seeking to partner with major pharmaceutical companies committed to carbon neutrality.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this biocatalytic process is essential for successful implementation. Common inquiries often revolve around the specific operational parameters, the scope of substrates compatible with the enzyme, and the long-term stability of the immobilized preparation. The following answers are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on how this technology performs under various conditions.
Q: What are the key advantages of the immobilized transaminase described in CN115466756A?
A: The immobilized transaminase offers significantly improved stability in organic solvent systems compared to liquid enzymes, allows for repeated reuse over multiple batches without significant loss of activity, and achieves high conversion rates exceeding 90% for specific substrates.
Q: Which specific mutations enhance the enzyme performance for sitagliptin synthesis?
A: The patent highlights variants where the arginine at position 241 of the amino acid sequence (SEQ ID NO: 5) is mutated to alanine, histidine, asparagine, serine, leucine, threonine, cysteine, or methionine, with specific combinations showing optimal activity.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for industrial scalability, utilizing robust immobilization on commercial resins and demonstrating consistent performance over at least 10 reaction batches, which simplifies downstream processing and reduces operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in modern drug manufacturing. Our team of experts is well-versed in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the immobilized transaminase technology described in CN115466756A can be seamlessly integrated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of sitagliptin intermediate meets the highest quality standards required by global regulatory bodies. Our commitment to technical excellence ensures that you receive a product that is not only cost-effective but also consistently reliable.
We invite you to explore how our capabilities can optimize your production of DPP-4 inhibitor intermediates. By leveraging our expertise in enzyme engineering and process development, we can help you achieve significant efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring a smooth transition to this advanced manufacturing platform.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
