Scalable Chemical Synthesis of Potent PTP1B Inhibitors for Diabetes Treatment
Scalable Chemical Synthesis of Potent PTP1B Inhibitors for Diabetes Treatment
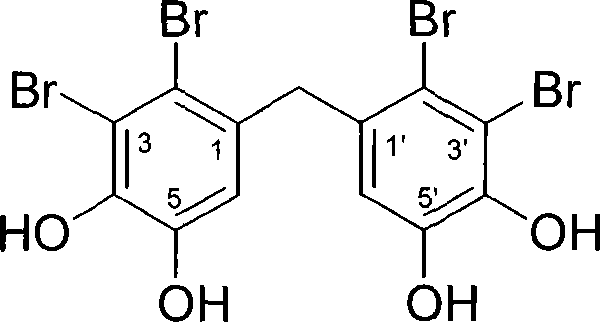
The global pharmaceutical landscape is increasingly focused on novel targets for Type 2 Diabetes Mellitus (T2DM), with Protein Tyrosine Phosphatase 1B (PTP1B) emerging as a critical negative regulator of insulin signaling. Patent CN101597213B discloses a robust chemical total synthesis method for Bis-(2,3-dibromo-4,5-dihydroxy-phenyl)-methane, a potent bromophenol derivative that acts as a highly effective PTP1B inhibitor. Unlike previous approaches that relied on the extraction of this bioactive compound from marine algae, which suffers from severe resource limitations and batch-to-batch variability, this patented methodology offers a fully synthetic pathway. By utilizing readily available industrial chemicals such as veratric acid and veratrole, the process establishes a reliable foundation for the mass production of high-purity pharmaceutical intermediates. This technological breakthrough not only secures the supply chain for next-generation antidiabetic agents but also significantly lowers the barrier to entry for clinical development by providing a cost-effective alternative to natural product isolation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of bioactive bromophenols like Bis-(2,3-dibromo-4,5-dihydroxy-phenyl)-methane has been heavily dependent on natural extraction from marine organisms, specifically certain species of red algae. This conventional approach presents profound challenges for the pharmaceutical industry, primarily due to the extremely low abundance of these compounds in nature, which necessitates the processing of massive quantities of biomass to obtain gram-scale amounts of the active ingredient. Furthermore, natural extraction is inherently susceptible to ecological fluctuations, seasonal variations, and geographical constraints, leading to inconsistent supply continuity that is unacceptable for Good Manufacturing Practice (GMP) production of drug substances. The purification of natural extracts is also notoriously difficult, often resulting in complex impurity profiles that require extensive and costly downstream processing to meet the stringent purity specifications required for human therapeutic use. Consequently, relying on natural sources creates a significant bottleneck that hinders the clinical translation of promising PTP1B inhibitors, making them economically unviable for widespread commercialization.
The Novel Approach
The synthetic strategy outlined in the patent represents a paradigm shift by replacing unpredictable biological sourcing with a deterministic, multi-step organic synthesis that guarantees reproducibility and scalability. This novel approach leverages classic organic transformations, including Friedel-Crafts acylation, electrophilic aromatic substitution, and ether cleavage, to construct the target molecule from simple, commodity-grade starting materials. By designing a route that introduces bromine atoms with high regioselectivity and preserves the sensitive phenolic functionality through protected methoxy intermediates, the method achieves a total yield that is commercially competitive while maintaining excellent control over the impurity profile. The elimination of biological variability ensures that every batch produced meets identical quality standards, thereby de-risking the supply chain for drug developers. Moreover, the use of standard solvents and reagents means that this process can be implemented in existing fine chemical manufacturing facilities without the need for specialized infrastructure, facilitating a rapid transition from laboratory discovery to industrial production.
Mechanistic Insights into Regioselective Bromination and Reduction
The core of this synthesis lies in the precise control of electrophilic aromatic substitution to install four bromine atoms at the 2, 2', 3, and 3' positions of the diphenylmethane scaffold. The process begins with the formation of a ketone intermediate via polyphosphoric acid-mediated acylation, which activates the aromatic rings for subsequent functionalization. The first bromination step utilizes molecular bromine in acetic acid at room temperature to selectively introduce bromine atoms at the positions ortho to the methoxy groups, driven by the strong activating effect of the electron-donating methoxy substituents. A second, more vigorous bromination step employs aluminum trichloride as a Lewis acid catalyst at elevated temperatures (75-85°C) to overcome the deactivating influence of the initially installed bromine atoms, ensuring complete dibromination on both rings. This sequential bromination strategy is critical for avoiding the formation of mono-brominated or tri-brominated byproducts, which would be difficult to separate and could compromise the biological activity of the final API. Following the construction of the halogenated skeleton, the ketone bridge is reduced to a methylene group using trifluoroacetic acid and triethylsilane, a mild yet effective system that avoids the harsh conditions of traditional Clemmensen or Wolff-Kishner reductions.
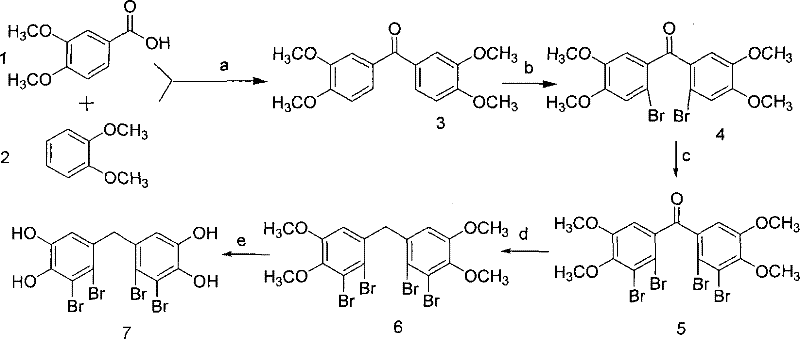
Impurity control is meticulously managed throughout the synthetic sequence, particularly during the final demethylation step where boron tribromide is used to cleave the methyl ethers to reveal the free phenolic hydroxyl groups. The choice of boron tribromide in dichloromethane at low temperatures allows for the selective removal of methyl groups without causing debromination or degradation of the sensitive aromatic core, a common side reaction with stronger acids or nucleophiles. The patent specifies a molar ratio of compound to boron tribromide of approximately 1:9 to 1:10, ensuring that the reagent is in sufficient excess to drive the reaction to completion while minimizing the formation of boron-containing complexes that could trap the product. Workup procedures involving ice-water quenching and careful pH control during extraction further ensure that acidic byproducts and residual boron species are effectively removed. This rigorous attention to mechanistic detail results in a final product with a defined melting point and spectral characteristics that confirm high chemical purity, essential for passing regulatory scrutiny in pharmaceutical applications.
How to Synthesize Bis-(2,3-dibromo-4,5-dihydroxy-phenyl)-methane Efficiently
The efficient production of this high-value pharmaceutical intermediate requires strict adherence to the optimized reaction parameters detailed in the patent to maximize yield and minimize waste. The synthesis is divided into five distinct operational stages, beginning with the condensation of veratric acid and veratrole, followed by two stages of bromination, a reduction step, and a final deprotection. Each stage requires precise temperature control and stoichiometric management of reagents, particularly the hazardous bromine and boron tribromide, to ensure operator safety and reaction consistency. The following guide outlines the critical process flow, emphasizing the specific conditions required to achieve the reported yields and purity levels suitable for downstream drug formulation.
- Friedel-Crafts acylation of veratric acid and veratrole using polyphosphoric acid at 75-85°C to form the ketone intermediate.
- Regioselective bromination using bromine in acetic acid, followed by further bromination with aluminum trichloride catalyst.
- Reduction of the ketone to methylene using trifluoroacetic acid and triethylsilane, followed by demethylation with boron tribromide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from natural extraction to this fully synthetic route offers transformative advantages in terms of cost stability and supply security. The reliance on marine biomass previously introduced significant volatility into the cost structure of PTP1B inhibitors, whereas this chemical synthesis utilizes commodity chemicals that are produced on a multi-ton scale globally, insulating the supply chain from ecological shocks. By eliminating the need for complex extraction and purification from biological matrices, the manufacturing process becomes significantly streamlined, reducing the number of unit operations and the associated labor and energy costs. This simplification of the production workflow directly translates to a lower cost of goods sold (COGS), enabling more competitive pricing for the final drug product and improving margins for pharmaceutical partners. Furthermore, the synthetic route's compatibility with standard chemical reactors means that production can be scaled up rapidly to meet surging demand without the long lead times associated with cultivating or harvesting natural resources.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive starting materials like veratric acid and veratrole, which are widely available from bulk chemical suppliers at stable prices. The elimination of expensive chromatography steps in the early stages, replaced by efficient crystallization and washing protocols, drastically reduces solvent consumption and waste disposal costs. Additionally, the avoidance of precious metal catalysts or exotic reagents further lowers the raw material expenditure, making the overall process highly cost-efficient compared to biotechnological or extraction-based alternatives. These cumulative savings allow for substantial cost reduction in pharmaceutical intermediate manufacturing, providing a strong financial incentive for adopting this synthetic pathway.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly enhanced by decoupling production from the limitations of natural sources, which are subject to seasonal availability and environmental regulations. The synthetic route relies on a robust network of chemical suppliers for reagents like bromine and polyphosphoric acid, ensuring that raw material shortages are unlikely to disrupt production schedules. The ability to produce the compound on demand in a controlled factory environment guarantees consistent lead times and inventory availability, which is critical for maintaining continuous drug manufacturing lines. This reliability reduces the risk of stockouts and allows for better long-term planning and contract negotiation with downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plants to large-scale commercial reactors without significant re-engineering. The use of common organic solvents like dichloromethane and acetic acid facilitates efficient solvent recovery and recycling systems, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process. Waste streams are primarily aqueous and organic phases that can be treated using standard industrial wastewater treatment protocols, ensuring compliance with strict environmental regulations. This ease of scale-up and environmental manageability makes the process an attractive option for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this PTP1B inhibitor, based on the data provided in the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of this intermediate for drug development programs. The answers reflect the specific chemical properties and process advantages identified in the intellectual property.
Q: What is the primary advantage of this synthetic route over natural extraction?
A: The synthetic route overcomes the bottleneck of limited natural sources (seaweed) by using cheap, commercially available starting materials like veratric acid, ensuring consistent supply and lower costs.
Q: How is regioselectivity controlled during the bromination steps?
A: The process utilizes specific conditions, such as using acetic acid as a solvent and controlling the molar ratio of bromine, along with Lewis acid catalysts like aluminum trichloride, to achieve precise 2,3-dibromo substitution.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent describes standard organic unit operations like filtration, extraction, and column chromatography, which are easily adaptable from laboratory scale to multi-ton commercial production without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-(2,3-dibromo-4,5-dihydroxy-phenyl)-methane Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-quality intermediates for the development of next-generation antidiabetic therapies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped to handle the specific reagents and conditions required for this bromination-heavy synthesis, adhering to stringent purity specifications and rigorous QC labs to guarantee batch-to-batch consistency. We are committed to supporting your R&D efforts by providing reliable access to this critical PTP1B inhibitor intermediate, enabling you to focus on clinical outcomes rather than supply chain uncertainties.
We invite you to engage with our technical team to discuss how our manufacturing capabilities can optimize your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how partnering with NINGBO INNO PHARMCHEM can accelerate your path to market while maximizing value.
