Advanced Catalytic Hydrogenation for Ultra-High Purity Clindamycin Phosphate Manufacturing
The pharmaceutical industry constantly grapples with the challenge of removing structurally similar impurities from Active Pharmaceutical Ingredients (APIs), a struggle that directly impacts drug safety and efficacy. Patent CN102964401A introduces a transformative approach to the preparation of Clindamycin Phosphate, a critical lincosamide antibiotic used globally for treating serious bacterial infections. Traditional manufacturing routes often leave behind trace amounts of dehydro-clindamycin phosphate, an unsaturated analog that is notoriously difficult to separate due to its physicochemical similarity to the target molecule. This patent discloses a sophisticated catalytic hydrogenation strategy that does not merely separate the impurity but chemically converts it into the desired product. By leveraging palladium-catalyzed reduction in aqueous or alcoholic media, manufacturers can achieve unprecedented purity levels, effectively turning a liability into an asset. This technical breakthrough offers a robust solution for reliable Clindamycin Phosphate supplier networks seeking to enhance their product quality profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Clindamycin Phosphate has relied heavily on repetitive crystallization and chromatographic techniques to reduce impurity levels. However, the structural proximity of dehydro-clindamycin phosphate to the final API creates a significant thermodynamic barrier to separation. In conventional processes, the dehydro-impurity often co-crystallizes with the product, leading to batches that fail stringent regulatory specifications for related substances. Furthermore, aggressive purification attempts to remove this specific impurity often result in substantial yield losses, driving up the cost of goods sold and creating supply bottlenecks. The reliance on complex solvent systems and multiple recrystallization steps also increases the environmental footprint and operational complexity of the manufacturing process. For procurement managers, these inefficiencies translate into higher costs and less predictable lead times for high-purity Clindamycin Phosphate batches.
The Novel Approach
The methodology outlined in CN102964401A represents a paradigm shift from separation to conversion. Instead of fighting to isolate the dehydro-impurity, the process utilizes catalytic hydrogenation to saturate the exocyclic double bond characteristic of the contaminant. By dissolving the crude material in water or a water-containing alcoholic liquid and introducing hydrogen gas in the presence of a palladium carbon catalyst, the dehydro-clindamycin phosphate is fully reduced to Clindamycin Phosphate. This chemical transformation ensures that the impurity is not just removed but is actually added to the yield of the final product. The reaction conditions are mild, typically proceeding at ambient temperatures with hydrogen pressures that are manageable in standard industrial reactors. This approach simplifies the downstream processing significantly, as the subsequent crystallization step yields a product virtually free of the dehydro-species, thereby streamlining the path to commercial scale-up of complex antibiotic intermediates.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation
The core of this innovation lies in the selective reduction of the carbon-carbon double bond located on the propyl side chain of the pyrrolidine ring, without affecting other sensitive functional groups such as the amide linkage, the glycosidic bond, or the phosphate ester. The mechanism involves the adsorption of both the hydrogen gas and the dehydro-clindamycin phosphate substrate onto the surface of the palladium nanoparticles supported on carbon. The palladium catalyst activates the molecular hydrogen, facilitating its addition across the alkene moiety in a syn-addition manner. This heterogeneous catalysis is highly efficient because the steric environment of the catalyst surface favors the reduction of the exposed exocyclic double bond while leaving the more hindered and electronically distinct regions of the molecule intact. The use of water or aqueous alcohol as the solvent medium further enhances the selectivity by modulating the solubility of the substrate and the interaction with the catalyst surface.
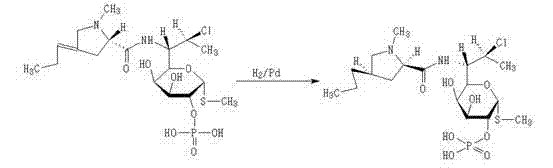
Controlling the impurity profile is critical for R&D directors focused on regulatory compliance. The conversion mechanism ensures that the specific impurity peak associated with the unsaturated analog disappears from the HPLC profile, replaced by the main product peak. This eliminates the need for specialized scavengers or complex extraction protocols that are often required to remove trace metals or organic impurities in other synthetic routes. The robustness of the Pd/C catalyst allows for consistent performance across different batches, provided that the hydrogen flow and stirring rates are maintained to ensure adequate mass transfer. By understanding this mechanistic pathway, process chemists can fine-tune reaction parameters such as catalyst loading (ranging from 1/5 to 1/100 of the substrate weight) and reaction time (1 to 48 hours) to optimize throughput without compromising the stringent purity specifications required for parenteral formulations.
How to Synthesize Clindamycin Phosphate Efficiently
The synthesis protocol described in the patent offers a straightforward yet highly effective route for producing pharmaceutical-grade Clindamycin Phosphate. The process begins with the dissolution of the crude starting material, which inherently contains the dehydro-impurity, into a suitable solvent system such as water or a mixture of water and ethanol. Once dissolved, a precise amount of palladium on carbon catalyst is introduced to the reaction vessel. The system is then purged and pressurized with hydrogen gas, initiating the reduction reaction under continuous stirring. Following the completion of the hydrogenation, indicated by the cessation of hydrogen uptake or the elapsed reaction time, the catalyst is removed via filtration. The filtrate is then subjected to concentration and crystallization, typically by adding anti-solvents like dehydrated alcohol and controlling the temperature to induce precipitation of the pure product.
- Dissolve crude Clindamycin Phosphate containing dehydro-impurities in water or a water-alcohol mixture.
- Add Palladium on Carbon (Pd/C) catalyst (1/5 to 1/100 ratio) and introduce hydrogen gas under stirring.
- React for 1 to 48 hours, filter the catalyst, and perform crystallization to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrogenation technology offers substantial strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By converting the major impurity into the product, the number of unit operations required to meet quality standards is significantly reduced. This reduction in processing steps directly correlates to lower operational expenditures, as there is less solvent consumption, reduced energy usage for heating and cooling cycles, and decreased labor requirements for monitoring and handling multiple crystallization crops. Furthermore, the yield improvement derived from converting the impurity means that less raw material is needed to produce the same amount of finished API, providing a direct lever for cost reduction in antibiotic manufacturing.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the recovery of yield from what was previously waste material leads to significant economic efficiency. Since the process avoids the use of exotic reagents and relies on recoverable heterogeneous catalysts, the overall cost structure is optimized. The ability to use water and common alcohols as solvents further reduces raw material costs compared to processes requiring chlorinated or aromatic solvents. Additionally, the removal of the need for extensive chromatographic purification or multiple recrystallizations lowers the capital intensity of the production line.
- Enhanced Supply Chain Reliability: The robustness of the catalytic hydrogenation process ensures consistent batch-to-batch quality, which is vital for maintaining uninterrupted supply to downstream formulation partners. The use of standard hydrogenation equipment means that the process can be easily transferred between manufacturing sites or scaled up without requiring specialized, hard-to-source machinery. This flexibility mitigates the risk of supply disruptions caused by equipment failure or capacity constraints at specific facilities. Moreover, the high purity of the resulting intermediate reduces the risk of batch rejection during quality control testing, ensuring smoother logistics and inventory management.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles by utilizing aqueous solvent systems and minimizing waste generation. The heterogeneous nature of the Pd/C catalyst allows for easy filtration and potential recycling, reducing the burden of heavy metal waste disposal. The process generates fewer organic solvent emissions and wastewater loads compared to traditional extraction-heavy methods. This environmental compatibility facilitates easier regulatory approval and supports corporate sustainability goals, making it an attractive option for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology for Clindamycin Phosphate production. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing supply chains.
Q: How does this method remove dehydro-clindamycin phosphate?
A: Unlike traditional purification which attempts to separate the impurity, this method uses catalytic hydrogenation to chemically convert the dehydro-clindamycin phosphate impurity directly into Clindamycin Phosphate, effectively eliminating the contaminant while increasing yield.
Q: What catalysts are required for this synthesis?
A: The process utilizes Palladium on Carbon (Pd/C) as the heterogeneous catalyst. The loading can vary from 1/5 to 1/100 of the raw material weight, offering flexibility in cost and reaction speed management.
Q: Is this process scalable for industrial production?
A: Yes, the method employs standard hydrogenation equipment and common solvents like water and ethanol. The reaction conditions (ambient to moderate pressure) are highly compatible with large-scale pharmaceutical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clindamycin Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of life-saving antibiotics like Clindamycin Phosphate. Our technical team has extensively analyzed the catalytic hydrogenation route described in CN102964401A and possesses the expertise to implement this advanced purification strategy at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of detecting trace impurities down to the ppm level, guaranteeing that every batch meets stringent purity specifications required by global pharmacopoeias.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains using this superior manufacturing technology. By leveraging our process engineering capabilities, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can ensure a stable, high-quality supply of Clindamycin Phosphate that meets the evolving demands of the global healthcare market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
