Revolutionizing Enamine Synthesis: A Metal-Free Route for High-Purity Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is documented in patent CN111423296A, which discloses a novel preparation method for β-haloenamic acid ester compounds. These compounds serve as critical building blocks in the construction of complex nitrogen-containing heterocycles found in numerous bioactive molecules, including alkaloids and antibiotics. The patent introduces a robust, metal-free catalytic system that operates under mild conditions, addressing many of the pain points associated with traditional enamine synthesis. By utilizing a multicomponent reaction strategy involving terminal alkynes, secondary amines, and N-haloimides, this technology enables the direct assembly of highly functionalized enamine scaffolds. This approach not only streamlines the synthetic route but also enhances the overall atom economy, making it a compelling choice for modern process chemistry.
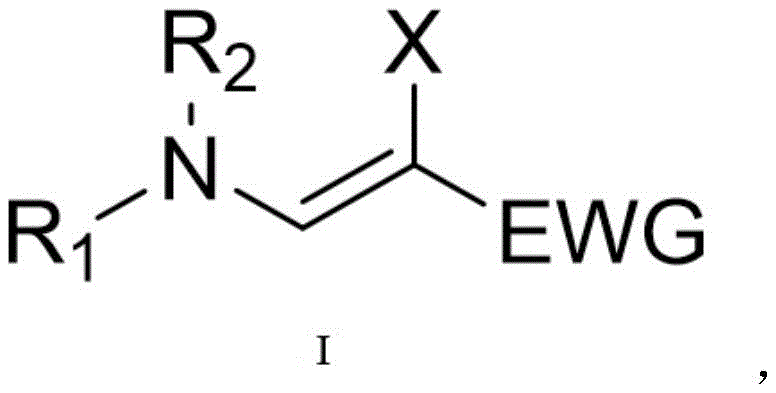
The structural versatility of these β-haloenamic acid esters, as depicted in the general formula, allows for extensive downstream functionalization. The presence of the halogen atom (Br or Cl) at the beta-position introduces a unique reactive handle that is absent in conventional enamine esters. This feature significantly expands the synthetic utility of the molecule, enabling further transformations such as cross-coupling reactions or nucleophilic substitutions. For R&D directors focused on developing new drug candidates, having access to such versatile intermediates can accelerate lead optimization campaigns. Furthermore, the ability to tune the electronic properties of the molecule through the selection of different electron-withdrawing groups (EWG) and amine substituents provides a powerful toolkit for medicinal chemists aiming to fine-tune the physicochemical properties of their target APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized enamines has been fraught with challenges that hinder their widespread adoption in large-scale manufacturing. Traditional methods often rely on condensation reactions between amines and carbonyl compounds, which can suffer from equilibrium issues and require harsh dehydration conditions. Alternatively, methods involving transition metal catalysis, while effective, introduce significant complications regarding product purity. The removal of trace heavy metals to meet regulatory standards for pharmaceutical ingredients is a costly and time-consuming process, often requiring specialized scavengers or multiple recrystallization steps. Moreover, many existing protocols necessitate strictly anhydrous and anaerobic environments, demanding specialized equipment and increasing the operational burden on production facilities. These factors collectively contribute to higher production costs and longer lead times, creating a bottleneck in the supply chain for high-value intermediates.
The Novel Approach
In stark contrast, the methodology described in patent CN111423296A offers a streamlined, one-pot solution that circumvents these traditional hurdles. By employing a metal-free catalytic system based on organic amines like DABCO, the process eliminates the risk of heavy metal contamination entirely. This shift not only simplifies the purification workflow but also aligns perfectly with the principles of green chemistry. The reaction proceeds efficiently at temperatures ranging from 0°C to 50°C, avoiding the energy-intensive heating or cryogenic cooling often required by older methods. Furthermore, the tolerance of this system towards a wide variety of substrates, including diverse terminal alkynes and secondary amines, demonstrates its robustness. This flexibility ensures that manufacturers can produce a broad library of derivatives without needing to re-optimize reaction conditions for each new analog, thereby enhancing process efficiency and reducing development timelines.
Mechanistic Insights into Metal-Free Multicomponent Halogenation
The success of this synthesis lies in its elegant mechanistic pathway, which leverages the nucleophilicity of secondary amines and the electrophilicity of N-haloimides. The reaction initiates with the nucleophilic attack of the secondary amine on the terminal alkyne, facilitated by the polar solvent DMF. This step generates a vinyl anion intermediate or a zwitterionic species that is stabilized by the adjacent electron-withdrawing group. Subsequently, the addition of the N-haloimide (such as NBS or NCS) serves as the halogen source. The DABCO catalyst plays a crucial role in activating the halogenating agent and facilitating the proton transfer steps necessary to establish the double bond geometry. The result is the highly selective formation of the Z-isomer of the β-haloenamine. This regio- and stereoselectivity is vital for ensuring consistent quality in downstream reactions, as mixed isomers can complicate purification and reduce overall yield in subsequent coupling steps.
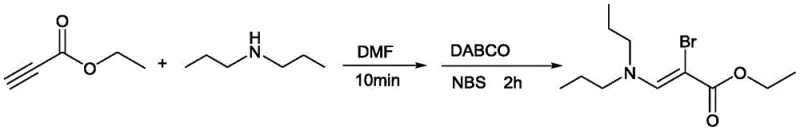
Understanding the impurity profile is equally critical for process validation. The mild conditions of this reaction minimize side reactions such as polymerization of the alkyne or over-halogenation, which are common pitfalls in more aggressive halogenation protocols. The use of saturated brine for quenching effectively stops the reaction without generating excessive heat or hazardous byproducts. Purification via neutral alumina chromatography, as specified in the patent examples, provides a gentle yet effective means of isolating the product, preserving the integrity of the sensitive enamine double bond. For quality control teams, this translates to a cleaner crude product with fewer difficult-to-remove impurities, simplifying the analytical burden and ensuring that the final material meets the stringent specifications required for GMP manufacturing environments.
How to Synthesize Beta-Haloenamic Acid Esters Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural details to maximize yield and purity. The process begins with the careful selection of dry glassware and reagents to prevent moisture interference, although the system is relatively robust compared to organometallic alternatives. The sequential addition of reagents—first the amine and alkyne, followed by the catalyst and halogen source—is key to controlling the reaction kinetics. Maintaining the temperature within the specified 0-50°C window ensures optimal conversion while preventing thermal degradation of the product. The workup procedure involving ethyl acetate extraction and drying over anhydrous sodium sulfate is standard but effective for removing polar byproducts. For detailed operational parameters and stoichiometric ratios, refer to the standardized guide below.
- Mix dimethylformamide (DMF), a terminal alkyne compound, and a secondary amine or derivative in a dry pressure-resistant tube at 0-25°C.
- Stir the mixture for 5-30 minutes to allow initial adduct formation, then add triethylenediamine (DABCO) and an N-haloimide (NBS or NCS).
- React at 0-50°C for 0.5-4 hours, quench with saturated brine, extract with ethyl acetate, and purify via neutral alumina chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free synthesis route offers substantial strategic benefits for procurement and supply chain management. The elimination of precious metal catalysts represents a direct reduction in raw material costs, as palladium, copper, or rhodium complexes are notoriously expensive and subject to volatile market pricing. Furthermore, the simplified purification process reduces the consumption of auxiliary materials such as metal scavengers and specialized filtration media. This efficiency gain extends to waste management, as the absence of heavy metals simplifies the disposal of chemical waste, lowering environmental compliance costs. For supply chain heads, the reliance on commodity chemicals like DMF, DABCO, and common amines ensures a stable and resilient supply base, mitigating the risk of disruptions associated with specialty reagent shortages.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound. By removing the need for expensive transition metal catalysts, the direct material cost of the synthesis is significantly lowered. Additionally, the one-pot nature of the reaction reduces solvent usage and energy consumption, as there is no need for intermediate isolation or extensive heating cycles. The simplified downstream processing further contributes to cost savings by reducing labor hours and equipment occupancy time. These cumulative efficiencies allow for a more competitive pricing structure for the final intermediate, providing a clear margin advantage in the highly price-sensitive pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain stability is paramount for continuous API production. This method utilizes widely available, bulk-grade chemicals that are sourced from a global network of suppliers, reducing dependency on single-source vendors for exotic catalysts. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental controls or reagent quality, leading to more consistent batch-to-batch performance. This reliability translates into predictable lead times and the ability to scale production volumes rapidly in response to market demand without the lengthy qualification processes often required for new catalytic systems.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently scalable due to its mild exothermic profile and lack of hazardous reagents. The reaction can be safely performed in standard stainless steel reactors without the need for specialized lining to prevent metal leaching. From an environmental standpoint, the process aligns with increasingly strict regulations regarding heavy metal discharge and solvent emissions. The reduced waste generation and lower energy footprint make this route attractive for companies aiming to improve their sustainability metrics and achieve green manufacturing certifications, which are becoming a key differentiator in B2B negotiations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the synthesis and application of these compounds. The following insights are derived directly from the experimental data and technical specifications outlined in the patent literature. These questions address critical aspects such as substrate compatibility, reaction safety, and product stability, providing a comprehensive overview for decision-makers considering the integration of this route into their manufacturing portfolio.
Q: What are the key advantages of this metal-free synthesis method?
A: The primary advantage is the elimination of expensive and toxic transition metal catalysts, which simplifies purification and ensures the final product meets stringent heavy metal limits required for pharmaceutical applications. Additionally, the one-pot nature reduces solvent waste and operational time.
Q: Can this method produce both bromo and chloro derivatives?
A: Yes, the method is highly versatile. By selecting either N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS) as the halogen source, manufacturers can selectively synthesize either beta-bromo or beta-chloro enamine acid esters with high regioselectivity.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The reaction conditions are mild (0-50°C) and utilize cheap, readily available raw materials. The absence of sensitive catalysts and the robustness of the one-pot procedure make it ideal for scaling up from laboratory to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Haloenamic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN111423296A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from benchtop to marketplace. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of beta-haloenamic acid ester meets the highest industry standards. Our commitment to quality assurance ensures that the complex impurity profiles associated with enamine chemistry are meticulously managed.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to request specific COA data for our available intermediates and to discuss route feasibility assessments for your target molecules. Together, we can optimize your supply chain and accelerate the delivery of life-saving therapies to patients worldwide.
