Advanced Manufacturing of 2-Deoxy-L-Ribose: A Scalable Route from L-Arabinose for High-Purity Nucleoside Intermediates
Advanced Manufacturing of 2-Deoxy-L-Ribose: A Scalable Route from L-Arabinose for High-Purity Nucleoside Intermediates
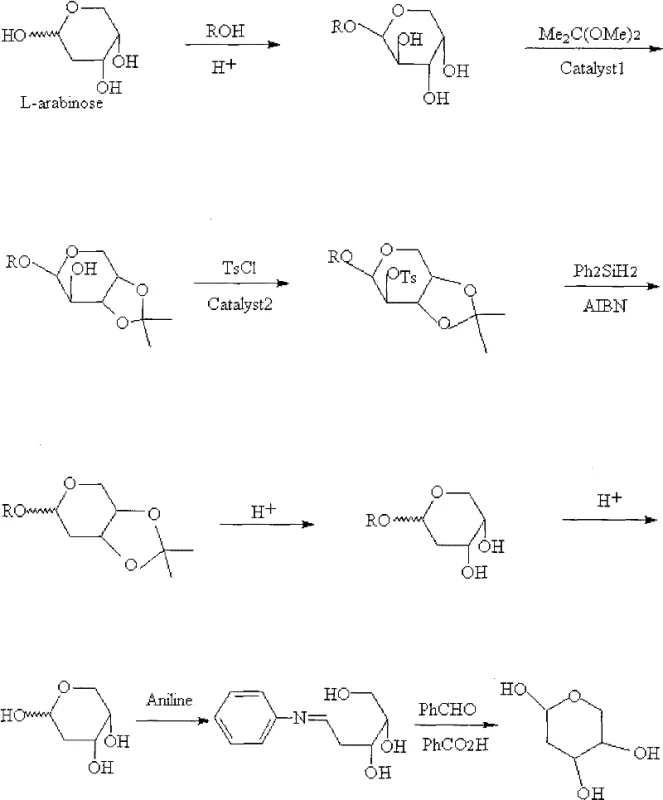
The pharmaceutical industry's relentless pursuit of novel antiviral and anticancer therapeutics has placed a premium on the availability of high-purity L-nucleoside intermediates. Central to this class of compounds is 2-deoxy-L-ribose, a critical building block for the synthesis of enantiomeric DNA and RNA analogs which exhibit superior metabolic stability and reduced toxicity compared to their natural D-counterparts. Patent CN102153600A introduces a transformative methodology for the preparation of 2-deoxy-L-ribose utilizing L-arabinose as the primary chiral feedstock. This innovation addresses long-standing challenges in carbohydrate chemistry by offering a route that circumvents the need for expensive reagents, harsh reaction conditions, and complex purification protocols that have historically plagued the commercial production of deoxy sugars. By leveraging a sequence of protection, activation, radical transformation, and a unique aniline-mediated purification strategy, this technology provides a robust framework for the reliable supply of pharmaceutical intermediates essential for next-generation oligonucleotide therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-deoxy-L-ribose has been fraught with significant technical and economic hurdles that impede efficient commercial scale-up. Traditional pathways often rely on starting materials like D-ribose or L-xylulose, which necessitate multiple stereochemical inversion steps to achieve the desired L-configuration, thereby drastically reducing overall atom economy and increasing waste generation. Furthermore, existing methodologies frequently employ highly toxic reagents such as tributyltin hydride for deoxygenation or require stringent conditions involving high vacuum and extreme temperatures that demand specialized equipment and pose safety risks in a manufacturing environment. The purification of the final sugar product is particularly problematic, as deoxy sugars are prone to degradation and difficult to separate from closely related impurities using standard crystallization techniques, often forcing manufacturers to resort to expensive preparative chromatography which is not viable for multi-ton production scales.
The Novel Approach
The methodology disclosed in CN102153600A represents a paradigm shift by utilizing L-arabinose, a readily available and inexpensive natural sugar, as the direct precursor, effectively eliminating the need for stereochemical inversions. This novel approach streamlines the synthesis into seven manageable steps that predominantly utilize acid-base catalysis and radical chemistry under mild conditions, typically ranging from room temperature to moderate reflux, without the requirement for high pressure or supervacuum systems. A standout feature of this process is the implementation of a radical deoxygenation strategy using diphenylsilane and azo-initiators, which replaces toxic tin reagents with safer, more manageable silicon-based hydrides. Additionally, the introduction of an aniline derivatization step for purification offers a clever chemical solution to isolation challenges, allowing the target molecule to be crystallized as a stable Schiff base derivative before being hydrolyzed back to the free sugar, ensuring exceptional purity levels suitable for sensitive biological applications.
Mechanistic Insights into Radical Deoxygenation and Aniline Purification
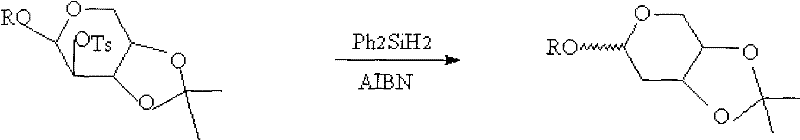
The core chemical transformation in this synthesis is the removal of the C2-hydroxyl group, achieved through a sophisticated radical mechanism that ensures high regioselectivity and yield. The process begins with the activation of the C2 position via tosylation, converting the hydroxyl group into an excellent leaving group (tosylate) while the 3,4-positions are protected as an isopropylidene acetal to prevent side reactions. Upon exposure to diphenylsilane and a radical initiator such as AIBN or diisopropyl azodicarboxylate, a radical chain reaction is initiated where the silyl radical abstracts the tosylate group, generating a carbon-centered radical at the C2 position which is subsequently quenched by hydrogen abstraction from the silane. This mechanism is highly advantageous for cost reduction in pharmaceutical intermediate manufacturing because it proceeds with high fidelity and minimizes the formation of elimination byproducts that are common in ionic deoxygenation pathways.
Beyond the synthesis itself, the patent details a unique purification mechanism that significantly enhances the impurity profile of the final product. The crude 2-deoxy-L-ribose is reacted with aniline to form a crystalline aniline derivative, likely a glycosylamine or Schiff base, which precipitates out of the solution, leaving soluble impurities behind in the mother liquor. This solid intermediate is then subjected to hydrolysis in the presence of benzaldehyde and benzoic acid, which serves to regenerate the free sugar while driving the equilibrium towards product formation. This two-step purification logic is critical for R&D directors focused on purity specifications, as it effectively removes colored impurities and trace metal residues without the need for silica gel chromatography, thereby simplifying the downstream processing and ensuring the material meets the stringent quality standards required for GMP production of active pharmaceutical ingredients.
How to Synthesize 2-Deoxy-L-Ribose Efficiently
The synthesis of 2-deoxy-L-ribose via this patented route involves a logical sequence of functional group manipulations designed to maximize yield and operational simplicity. The process initiates with the Fischer glycosidation of L-arabinose to protect the anomeric center, followed by acetal formation to lock the ring conformation and protect adjacent hydroxyls. Subsequent activation of the C2 position sets the stage for the key radical deoxygenation event, after which global deprotection reveals the target sugar skeleton. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and temperature profiles for each of the seven stages, are outlined in the guide below to facilitate immediate technology transfer and process validation.
- Protect L-arabinose by reacting with alcohol under acid catalysis to form 1-O-alkyl-L-arabinose glycosides.
- Form the 3,4-O-isopropylidene acetal using dimethyl acetal or acetone with strong-acid cation exchange resin catalysis.
- Activate the C2 position by tosylation using TsCl and organic base to prepare the sulfonate intermediate.
- Perform radical deoxygenation using diphenylsilane and a radical initiator like AIBN or DIAD to remove the C2 oxygen.
- Deprotect the acetal and glycoside groups under acidic conditions to yield crude 2-deoxy-L-ribose.
- Purify the crude product via aniline derivatization followed by reduction with benzaldehyde and benzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers substantial strategic benefits that extend beyond mere technical feasibility. The reliance on L-arabinose as a starting material leverages a commodity chemical with a stable and abundant global supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized precursors. The elimination of high-pressure reactors and cryogenic conditions reduces the capital expenditure required for manufacturing infrastructure, allowing for production in standard glass-lined or stainless steel vessels commonly found in multipurpose chemical plants. Furthermore, the replacement of toxic tin reagents with silanes and the use of recyclable ion-exchange resins for catalysis aligns with modern environmental compliance standards, significantly lowering the costs associated with waste treatment and regulatory reporting.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing cheap, commercially available reagents such as methanol, acetone, and tosyl chloride, while avoiding the use of precious metal catalysts or expensive chiral auxiliaries. The high yields reported in the patent embodiments, particularly in the protection and deprotection steps, minimize raw material consumption and reduce the cost of goods sold (COGS). Additionally, the simplified purification protocol eliminates the need for expensive chromatographic resins and large volumes of organic solvents, leading to substantial savings in both material costs and solvent recovery operations.
- Enhanced Supply Chain Reliability: By basing the synthesis on L-arabinose, a bulk chemical derived from biomass, the supply chain becomes inherently more resilient to fluctuations in the petrochemical market. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without compromising product quality, ensures consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical clients who depend on a steady flow of high-quality nucleoside intermediates.
- Scalability and Environmental Compliance: The absence of hazardous reagents and the use of mild reaction parameters make this process exceptionally scalable from pilot plant to commercial tonnage without the need for extensive re-engineering. The "green" nature of the chemistry, characterized by low toxicity and the potential for solvent recycling, facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. This alignment with sustainability goals not only lowers operational risks but also enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-deoxy-L-ribose using this specific patented methodology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and product quality attributes.
Q: Why is L-arabinose preferred over D-ribose for synthesizing 2-deoxy-L-ribose?
A: L-arabinose is a naturally abundant and cost-effective chiral pool starting material that possesses the correct stereochemistry for L-nucleoside synthesis, avoiding the need for complex inversion steps required when starting from D-sugars.
Q: How does the aniline purification step improve product quality?
A: The formation of a crystalline aniline derivative allows for the physical separation of impurities through filtration, followed by hydrolysis to release the pure sugar, significantly enhancing the purity profile compared to standard chromatography.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions without high temperature or pressure, employs cheap and non-toxic reagents, and avoids difficult separation steps, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Deoxy-L-Ribose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity sugar intermediates play in the development of life-saving antiviral and oncology drugs. Our team of expert process chemists has thoroughly evaluated the route described in CN102153600A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory benchtop to industrial reactor. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-deoxy-L-ribose meets the exacting standards required for GMP synthesis of nucleoside analogs.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in carbohydrate chemistry can accelerate your drug development timeline while ensuring a secure and cost-effective supply of this vital intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
