Advanced Synthetic Route for 2,3-Dihydro-1-oxo-1H-indene-4-carbonitrile: Enhancing Safety and Scalability
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with operational safety, particularly for critical small molecule therapeutics. Patent CN111704559A, published on September 25, 2020, introduces a transformative methodology for preparing 2,3-dihydro-1-oxo-1H-indene-4-carbonitrile, a pivotal intermediate in the synthesis of Ozanimod and related therapeutic agents. This technical disclosure addresses long-standing challenges in the field by replacing hazardous cyanation reagents with a safer, multi-step sequence starting from 2-cyanobenzyl bromide. For R&D directors and procurement specialists, this innovation represents a significant shift towards greener chemistry without compromising the structural integrity required for downstream drug synthesis. The protocol leverages common industrial reagents such as Meldrum's acid derivatives and standard Lewis acids, effectively lowering the barrier to entry for commercial manufacturing while adhering to stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydro-1-oxo-1H-indene-4-carbonitrile has been plagued by severe safety and operational constraints that hinder scalable production. Prior art, including publications in the Journal of Organic Chemistry and Organic Letters, predominantly relies on the introduction of the cyano group via high-temperature reactions with cuprous cyanide or zinc cyanide. These heavy metal cyanides are notoriously toxic, posing acute health risks to operators and generating substantial volumes of hazardous wastewater that require complex and costly treatment protocols. Furthermore, the decarboxylation steps in these traditional routes often necessitate extreme thermal conditions, specifically temperatures reaching 165°C, which places immense stress on reactor materials and increases energy consumption. The combination of toxic reagents and harsh thermal requirements creates a bottleneck for reliable pharmaceutical intermediate supplier networks, as few facilities are equipped or permitted to handle such dangerous processes efficiently.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a strategic retrosynthetic disconnection that introduces the cyano group at the very beginning of the synthesis via commercially available 2-cyanobenzyl bromide. This fundamental shift eliminates the need for late-stage cyanation with toxic metal salts entirely. The process flows through a condensation with a cyclic malonate derivative, followed by hydrolysis and a remarkably mild decarboxylation step performed at just 100-105°C. This reduction in thermal demand not only preserves equipment longevity but also significantly lowers the energy footprint of the manufacturing process. The final cyclization is achieved through a classic Friedel-Crafts acylation, a well-understood transformation that offers high predictability and ease of purification. By integrating these steps, the new route provides a streamlined pathway that aligns perfectly with modern goals for cost reduction in API manufacturing.
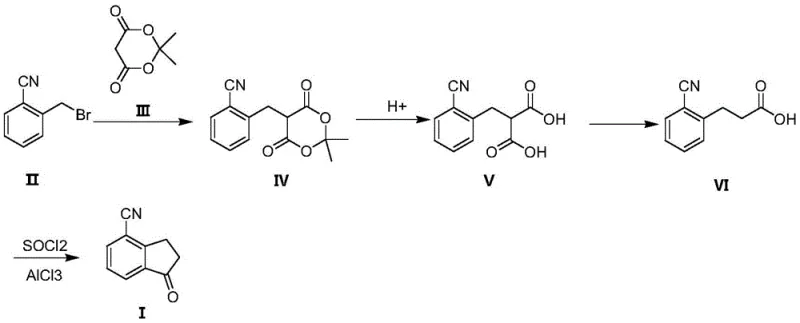
Mechanistic Insights into Friedel-Crafts Cyclization and Decarboxylation
The core of this synthetic strategy lies in the precise control of the decarboxylation and subsequent intramolecular acylation mechanisms. In the third step, the 2-(2-cyanophenyl)malonic acid intermediate undergoes thermal decarboxylation in a polar aprotic solvent like N-methylpyrrolidone, facilitated by a mild base such as pyridine. The mechanism involves the formation of a cyclic transition state that allows for the loss of carbon dioxide at relatively low temperatures compared to the 165°C required in older methods. This gentle removal of the carboxyl group prevents the degradation of the sensitive nitrile functionality and minimizes the formation of polymeric byproducts. Following this, the resulting 3-(2-cyanophenyl)propionic acid is activated using oxalyl chloride to form the corresponding acid chloride in situ. This highly reactive species then undergoes an intramolecular electrophilic aromatic substitution catalyzed by aluminum trichloride. The Lewis acid coordinates with the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon and driving the attack on the ortho-position of the aromatic ring to close the five-membered indanone ring system efficiently.
From an impurity control perspective, this mechanism offers distinct advantages over cyanide-based routes. Traditional methods often suffer from residual heavy metal contamination (copper or zinc) which is difficult to remove to ppm levels required for pharmaceutical grades. By avoiding these metals entirely, the new process inherently produces a cleaner crude product, simplifying the downstream purification workload. Additionally, the use of Meldrum's acid derivatives in the initial condensation step provides excellent regioselectivity, preventing the formation of unwanted isomers that could complicate the final crystallization. The hydrolysis step is conducted under acidic conditions which helps to protonate basic impurities, allowing them to be washed away during the aqueous workup. This multi-layered approach to impurity management ensures that the final high-purity OLED material or pharmaceutical intermediate meets the rigorous specifications demanded by global regulatory bodies without the need for extensive chromatographic purification.
How to Synthesize 2,3-Dihydro-1-oxo-1H-indene-4-carbonitrile Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control across the four distinct stages to maximize overall yield. The process begins with the alkylation of the methylene group in the cyclic malonate, followed by hydrolytic cleavage of the ester protecting groups. The subsequent decarboxylation must be monitored closely to ensure complete gas evolution before proceeding to the final cyclization. While the general workflow is robust, specific parameters regarding solvent choice and catalyst loading are critical for reproducibility on a large scale. For detailed operational procedures, including exact reagent quantities and workup protocols, please refer to the standardized synthesis guide below which encapsulates the preferred embodiments of the patent.
- Condense 2-cyanobenzyl bromide with Meldrum's acid derivative in DMF using a base like potassium carbonate.
- Hydrolyze the resulting intermediate in water with trifluoroacetic acid at 60-70°C to form the malonic acid derivative.
- Perform thermal decarboxylation in N-methylpyrrolidone with pyridine at 100-105°C to yield 3-(2-cyanophenyl)propionic acid.
- Execute Friedel-Crafts acylation using oxalyl chloride and aluminum trichloride at 130-135°C to cyclize into the final indanone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible operational benefits and risk mitigation. The most immediate impact is the elimination of cuprous cyanide and zinc cyanide from the bill of materials. These reagents are not only expensive due to their hazardous nature but also subject to strict transportation and storage regulations that can delay shipments and increase logistics costs. By substituting them with stable, non-toxic organic precursors, the supply chain becomes more resilient and less prone to regulatory interruptions. Furthermore, the reduction in reaction temperature from 165°C to roughly 100-135°C means that the process can be run in standard glass-lined or stainless steel reactors without the need for specialized high-temperature heating media, thereby expanding the pool of eligible contract manufacturing organizations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of waste treatment and the avoidance of expensive heavy metal scavengers. Since no toxic cyanide salts are used, the cost associated with neutralizing and disposing of hazardous wastewater is drastically reduced. Additionally, the high yield of the decarboxylation step, reported at nearly quantitative levels in the patent examples, minimizes raw material loss. The use of common solvents like dichloromethane and DMF, which are readily available in bulk, further stabilizes the variable costs of production. This holistic reduction in operational overhead allows for significant cost savings in fine chemical intermediates manufacturing without sacrificing product quality.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commodity chemicals that have stable global supply chains. 2-Cyanobenzyl bromide and Meldrum's acid are produced by multiple vendors worldwide, reducing the risk of single-source dependency. The milder reaction conditions also imply less wear and tear on production equipment, leading to fewer unplanned maintenance shutdowns and more consistent batch turnover rates. For supply chain planners, this predictability is crucial for maintaining inventory levels of critical API intermediates. The ability to scale this process from laboratory benchtop to multi-ton production without changing the fundamental chemistry ensures that lead times remain short even as demand for the final drug product fluctuates.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety flaws, but this route is inherently designed for expansion. The exothermic profiles of the reactions are manageable, and the absence of highly toxic gases reduces the need for complex scrubbing systems. From an environmental compliance standpoint, the process generates significantly less hazardous waste, aligning with increasingly strict global emissions standards. This 'green' profile facilitates faster regulatory approvals for new manufacturing sites and reduces the likelihood of environmental fines. Consequently, the commercial scale-up of complex pharmaceutical intermediates becomes a smoother, more predictable endeavor, securing long-term supply continuity for downstream partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from a detailed analysis of the experimental data and process descriptions provided in the source documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines.
Q: Why is this new synthesis route safer than conventional methods?
A: Conventional methods rely on highly toxic cuprous cyanide or zinc cyanide for cyano group introduction and require extreme decarboxylation temperatures of 165°C. This novel patent avoids toxic cyanide salts entirely by starting with 2-cyanobenzyl bromide and utilizes milder decarboxylation conditions around 100-105°C, significantly reducing environmental hazards and equipment stress.
Q: What are the typical yields for this four-step process?
A: According to the experimental data in patent CN111704559A, the process demonstrates robust efficiency with step yields of approximately 77.4% for condensation, 74.4% for hydrolysis, an excellent 99.6% for decarboxylation, and 68.2% for the final cyclization, ensuring a viable overall production yield for commercial scale-up.
Q: How does this method impact supply chain reliability for Ozanimod production?
A: By eliminating the need for specialized high-temperature reactors capable of sustaining 165°C and removing the regulatory burdens associated with handling剧毒 cyanide reagents, this method simplifies procurement and logistics. It allows for production in standard stainless steel facilities, thereby enhancing supply continuity and reducing lead times for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydro-1-oxo-1H-indene-4-carbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications like Ozanimod. Our technical team has thoroughly analyzed the methodology described in CN111704559A and possesses the expertise to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of 2,3-dihydro-1-oxo-1H-indene-4-carbonitrile meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthesis for your upcoming projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this safer route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive efficiency and safety in your supply chain, ensuring a seamless flow of high-quality materials for your global operations.
