Advanced Metal-Free Synthesis of Diaryl Formyl Substituted Pyrazole Compounds for Commercial Scale-up
Advanced Metal-Free Synthesis of Diaryl Formyl Substituted Pyrazole Compounds for Commercial Scale-up
The pharmaceutical and agrochemical industries continuously demand robust, scalable, and environmentally benign synthetic routes for heterocyclic scaffolds, particularly pyrazole derivatives which serve as critical nuclei in numerous bioactive molecules. Patent CN112876416B, published in March 2022, introduces a groundbreaking preparation method for diaryl formyl substituted pyrazole compounds that addresses long-standing inefficiencies in conventional manufacturing. This technology leverages a novel (2+1+1+1) cyclization strategy between diazo tetrafluoroborate and sulfur ylides, eliminating the need for toxic heavy metal catalysts and harsh reaction conditions. The significance of this innovation is underscored by the prevalence of the pyrazole skeleton in high-value commercial drugs such as Fipronil, Celebrex, and Doramapimod, highlighting the massive market potential for improved intermediates.
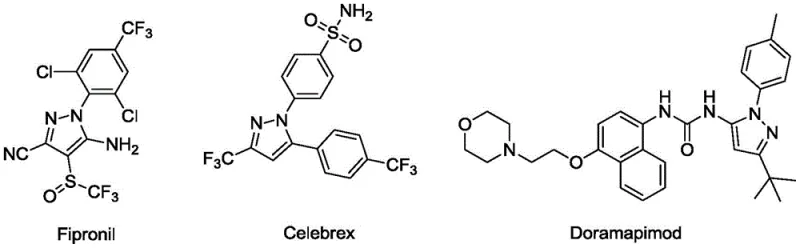
For R&D directors and process chemists, the ability to access diverse polysubstituted pyrazoles with high functional group tolerance is paramount for lead optimization and scale-up. The disclosed method operates under mild temperatures ranging from 0°C to 40°C, utilizing readily available starting materials that bypass the complex preparation of unstable halogenated hydrazones. By shifting the paradigm from metal-catalyzed cycloadditions to base-promoted organic transformations, this patent offers a pathway to significantly reduce impurity profiles related to metal leaching, thereby simplifying downstream purification and ensuring compliance with stringent ICH Q3D guidelines for elemental impurities in drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyrazole compounds has been fraught with technical hurdles that impede efficient commercial production. Traditional strategies predominantly rely on the cyclization of halogenated hydrazones with alkynes, a process severely hampered by the inherent instability and water sensitivity of the hydrazone precursors. Furthermore, the preparation of the requisite alkyne substrates is often complex and limited in scope, restricting the structural diversity accessible to medicinal chemists. Another prevalent approach involves metal-catalyzed cycloaddition, which, while effective, introduces significant environmental and safety liabilities due to the use of heavy metals that require costly removal steps to meet pharmaceutical purity standards.
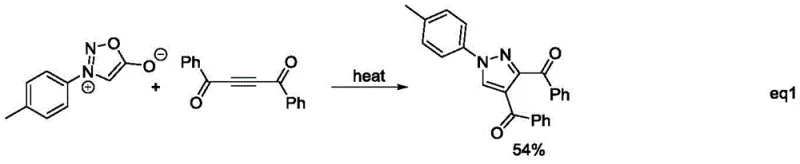
Specific prior art methods illustrate these deficiencies vividly. For instance, one common route involves refluxing intermediate ionic compounds with aryl-substituted butynedione, a process that suffers from severe reaction conditions and the handling of pungent, hazardous butynedione reagents. The intermediate ionic compounds themselves are notoriously difficult to prepare and exhibit poor stability, leading to inconsistent batch quality and low throughput. Another strategy employs chlorinated hydrazones and enaminones under high temperature and high pressure, creating significant safety risks for plant operators and requiring specialized high-pressure equipment that increases capital expenditure. These legacy methods collectively fail to meet the modern industry's demand for green, safe, and versatile synthetic platforms.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the invention disclosed in CN112876416B presents a streamlined, metal-free methodology that fundamentally reshapes the synthesis landscape for diaryl formyl substituted pyrazoles. The core innovation lies in the direct cyclization of sulfur ylides with diazo tetrafluoroborate salts under the influence of a mild base, proceeding through a unique (2+1+1+1) mechanism. This approach completely circumvents the need for unstable halogenated hydrazones or hazardous alkynes, utilizing instead stable, commercially accessible sulfur ylide precursors that tolerate a wide array of functional groups. The reaction proceeds smoothly at ambient or near-ambient temperatures (0-40°C), drastically reducing energy consumption compared to the high-temperature reflux or high-pressure conditions of the past.
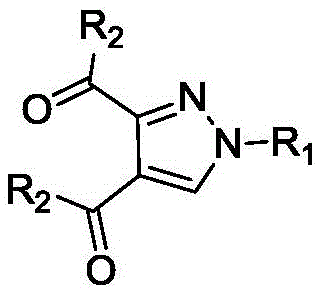
The versatility of this new platform is evidenced by its broad substrate scope, successfully accommodating variously substituted phenyl, naphthyl, and heterocyclic aryl groups without compromising yield or purity. By eliminating transition metals from the catalytic cycle, the process inherently produces cleaner crude products, minimizing the burden on purification teams and reducing the generation of heavy metal waste streams. The resulting diaryl formyl substituted pyrazole compounds are obtained in good to excellent yields, with the patent reporting separation yields frequently exceeding 60% and reaching up to 90% in optimized examples. This represents a substantial leap forward in process efficiency, offering a reliable, scalable, and environmentally responsible solution for the manufacture of high-value pyrazole intermediates.
Mechanistic Insights into Base-Promoted (2+1+1+1) Cyclization
The mechanistic elegance of this transformation lies in its stepwise construction of the pyrazole ring without external metal coordination. The reaction initiates with the nucleophilic attack of the sulfur ylide on the diazonium species, facilitated by the presence of a base such as sodium hydroxide or potassium carbonate. This interaction triggers a cascade of bond formations and cleavages, effectively assembling the five-membered heterocyclic core through a concerted (2+1+1+1) cyclization pathway. Following the initial ring closure, the intermediate undergoes spontaneous deacylation and aromatization processes, driven by the thermodynamic stability of the aromatic pyrazole system. This sequence ensures that the final product is formed directly with the desired oxidation state, avoiding the need for additional oxidative or reductive workup steps that often complicate multi-step syntheses.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed alternatives. The absence of metal catalysts eliminates the risk of metal-mediated side reactions, such as homocoupling or over-reduction, which can generate difficult-to-remove trace impurities. Furthermore, the mild basic conditions preserve sensitive functional groups like esters, nitriles, and halides, which might otherwise degrade under the harsh acidic or thermal conditions of traditional methods. The use of aprotic solvents like acetonitrile further enhances reaction homogeneity and rate, ensuring consistent kinetics across different scales. This robust mechanistic profile translates directly to a cleaner impurity spectrum, simplifying the analytical characterization and release testing required for GMP manufacturing of pharmaceutical intermediates.
How to Synthesize Diaryl Formyl Substituted Pyrazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory discovery and pilot plant operations. The general procedure involves dissolving the sulfur ylide and tetrafluoroborate diazonium salt in a suitable solvent, followed by the addition of a stoichiometric amount of base. The mixture is then stirred at a controlled temperature until conversion is complete, typically monitored by TLC or HPLC. Upon completion, the solvent is removed under reduced pressure, and the crude residue is purified via standard silica gel column chromatography to afford the target pyrazole derivative as a high-purity solid. This straightforward workflow minimizes unit operations and reduces the overall processing time, aligning perfectly with the needs of agile process development teams.
- Dissolve sulfur ylide, tetrafluoroborate diazonium salt, and a base such as sodium hydroxide in an aprotic solvent like acetonitrile.
- Stir the reaction mixture at a mild temperature between 0°C and 40°C until the sulfur ylide is fully consumed.
- Remove the solvent under reduced pressure and purify the resulting crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of expensive noble metal catalysts such as palladium or rhodium removes a significant variable cost component from the bill of materials, while also eradicating the need for specialized scavenger resins or complex extraction protocols required to meet residual metal specifications. This simplification of the downstream processing train not only reduces solvent consumption and waste disposal costs but also shortens the overall production cycle time, allowing for faster turnaround on custom synthesis orders and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts and the use of mild reaction conditions significantly lower the operational expenditure associated with pyrazole production. By avoiding high-pressure reactors and energy-intensive heating protocols, the process reduces utility costs and equipment depreciation. Furthermore, the high functional group tolerance minimizes the need for protecting group strategies, shortening the synthetic linear sequence and reducing the cumulative loss of material across multiple steps. These factors combine to deliver a substantially more cost-effective manufacturing process compared to traditional alkyne-based or metal-catalyzed routes.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials like sulfur ylides and diazonium salts mitigates the risk of supply disruptions often associated with specialized or hazardous reagents. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the incidence of failed batches and the need for re-processing. This reliability is crucial for maintaining continuous supply lines for critical pharmaceutical intermediates, ensuring that downstream API manufacturing schedules are not compromised by upstream variability or raw material shortages.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with increasingly stringent global environmental regulations regarding heavy metal discharge and waste management. Scaling this process from gram to tonnage quantities does not introduce the complex engineering challenges associated with handling pyrophoric reagents or high-pressure gases. The simplified workup and purification procedures facilitate easier technology transfer between sites and reduce the environmental footprint of the manufacturing facility, supporting corporate sustainability goals and enhancing the company's reputation as a responsible chemical supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios or for initiating new development projects focused on pyrazole-containing targets.
Q: What are the main limitations of traditional pyrazole synthesis methods?
A: Traditional methods often rely on halogenated hydrazones which are water-sensitive and difficult to prepare, or alkynes which have a narrow substrate range. Additionally, metal-catalyzed routes introduce heavy metal contamination risks.
Q: Does this new synthesis method require expensive noble metal catalysts?
A: No, the disclosed method operates without any heavy or noble metal catalysts, utilizing a base-promoted cyclization of sulfur ylides and diazonium salts instead.
Q: What kind of yields can be expected from this process?
A: The patent reports that the separation yield for most products exceeds 60%, with specific examples achieving yields as high as 90% under optimized conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Formyl Substituted Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies disclosed in CN112876416B for the production of high-value pyrazole intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this metal-free route are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of diaryl formyl substituted pyrazole meets the exacting standards required by top-tier pharmaceutical clients, delivering consistency and quality that you can trust for your critical drug development programs.
We invite you to collaborate with our technical team to explore how this advanced synthesis can optimize your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or detailed technical support for process validation, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations that demonstrate the tangible value of partnering with a leader in fine chemical innovation.
