Advanced Synthesis of N-Difluoromethyl Azole Thiourea Derivatives for Commercial Scale-Up
Advanced Synthesis of N-Difluoromethyl Azole Thiourea Derivatives for Commercial Scale-Up
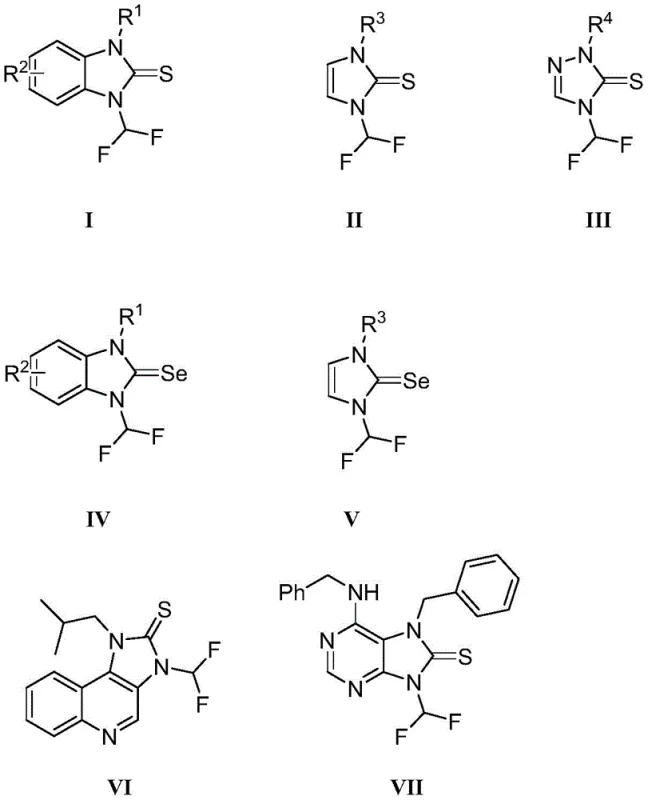
The strategic incorporation of fluorine atoms into organic molecules has revolutionized modern medicinal chemistry and agrochemical design, particularly through the introduction of the difluoromethyl group which acts as a unique lipophilic hydrogen bond donor. This structural motif significantly enhances membrane permeability, metabolic stability, and bioavailability in drug candidates, making the efficient synthesis of difluoromethylated heterocycles a critical priority for R&D teams globally. Patent CN112194634A discloses a groundbreaking preparation method for N-difluoromethyl imidazole sulfur and selenium urea derivatives, offering a robust pathway to access these high-value scaffolds. The invention covers a broad spectrum of compounds including N-difluoromethyl benzimidazole thiourea, imidazole thiourea, and triazole derivatives, all synthesized via a unified and economical protocol. This technological advancement addresses the longstanding challenge of introducing difluoromethyl groups under mild conditions without compromising functional group integrity.
For procurement managers and supply chain directors, the ability to source reliable pharmaceutical intermediates with consistent quality and reduced lead times is paramount. The disclosed technology leverages readily available starting materials such as elemental sulfur or selenium powder and ethyl bromodifluoroacetate, bypassing the need for exotic or hazardous reagents. This shift in synthetic strategy not only simplifies the supply chain logistics but also drastically reduces the raw material costs associated with producing complex fluorinated heterocycles. By adopting this method, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing while maintaining stringent purity specifications required for downstream drug synthesis. The versatility of the method allows for the production of a wide library of derivatives, facilitating rapid structure-activity relationship (SAR) studies for new drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-difluoromethyl azole thiourea compounds relied heavily on the use of Trimethylsilyl Fluorosulfonyl Difluoroacetate (TFDA) as the difluorocarbene source. While effective in generating the necessary reactive intermediates, TFDA is a complex reagent that is difficult to obtain commercially and often requires specialized handling due to its sensitivity and potential hazards. Furthermore, conventional protocols typically necessitate the use of strong bases to catalyze the reaction, which poses severe limitations regarding substrate compatibility. Strong basic conditions can lead to the degradation of sensitive functional groups present on the azole ring or side chains, resulting in poor yields and complex impurity profiles that are difficult to separate. This lack of functional group tolerance restricts the chemical space accessible to researchers, hindering the construction of diverse drug molecule libraries essential for identifying potent bioactive candidates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes ethyl bromodifluoroacetate (BrCF2CO2Et) in conjunction with elemental sulfur or selenium and a mild catalyst system comprising sulfinates or alkalis. This method operates under significantly milder conditions, typically at temperatures between 80°C and 120°C, which preserves the integrity of delicate functional groups such as esters, nitro groups, and halogens. The use of simple inorganic salts like sodium hydroxymethyl sulfinate or potassium carbonate as catalysts eliminates the need for expensive transition metals or harsh bases, streamlining the workup procedure and reducing waste generation. This broad-spectrum and economical reaction technology enables the efficient preparation of N-difluoromethyl azole thiourea derivatives with diverse structural modifications, opening new avenues for the development of herbicides, pharmaceuticals, and organic functional materials with enhanced biological activity.
Mechanistic Insights into Sulfinate-Catalyzed Difluoromethylation
The core of this innovative synthesis lies in the generation of difluoromethyl radicals or carbenoid species from ethyl bromodifluoroacetate mediated by the sulfinate or alkali catalyst. In the presence of elemental sulfur, the reaction likely proceeds through a radical mechanism where the difluoromethyl species attacks the sulfur atom to form a reactive intermediate, which subsequently undergoes cyclization or addition to the nitrogen atom of the azole ring. This mechanistic pathway is distinct from traditional nucleophilic substitutions and offers superior regioselectivity, ensuring that the difluoromethyl group is installed precisely at the desired position on the heterocyclic scaffold. The compatibility of this mechanism with various azole substrates, including benzimidazoles, imidazoles, and triazoles, demonstrates the robustness of the catalytic system in activating the C-H or N-H bonds of the heterocycle without requiring pre-functionalization.
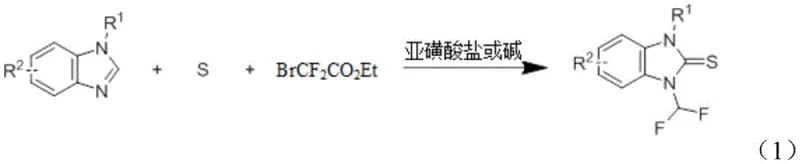
From an impurity control perspective, the mild reaction conditions play a crucial role in minimizing side reactions such as polymerization or over-fluorination which are common in harsher fluoroalkylation processes. The use of stoichiometric amounts of elemental sulfur ensures that the thiocarbonyl formation is efficient, while the choice of polar aprotic solvents like N,N-Dimethylacetamide (DMA) facilitates the solubility of both organic substrates and inorganic catalysts, promoting homogeneous reaction kinetics. Detailed analysis of the reaction products reveals high purity levels with minimal byproduct formation, simplifying the downstream purification steps such as flash column chromatography. This high level of control over the reaction trajectory is essential for meeting the rigorous quality standards of the pharmaceutical industry, where even trace impurities can impact the safety and efficacy of the final active pharmaceutical ingredient.
How to Synthesize N-Difluoromethyl Benzimidazole Thiourea Efficiently
The synthesis protocol described in the patent provides a straightforward and scalable route for producing high-purity N-difluoromethyl benzimidazole thiourea derivatives, which are valuable intermediates in the synthesis of antifungal agents and other bioactive compounds. The process involves mixing the azole precursor with sulfur powder and a catalyst in a sealed tube, followed by the addition of the fluorinating agent and heating. This standardized approach ensures reproducibility across different batches and scales, making it an ideal candidate for technology transfer from laboratory to pilot plant. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below which outlines the critical steps for maximizing yield and purity.
- Combine the azole substrate (e.g., 1-benzylbenzimidazole), elemental sulfur powder, and a sulfinate or alkali catalyst in a reaction vessel with an organic solvent like DMA.
- Inject ethyl bromodifluoroacetate (BrCF2CO2Et) into the mixture and heat the reaction to a temperature range of 80-120°C for approximately 24 hours.
- Upon completion, cool the mixture, extract with ethyl acetate and brine, dry over anhydrous sodium sulfate, and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthesis route offers substantial strategic benefits for procurement and supply chain operations, primarily driven by the simplification of the raw material portfolio and the elimination of hazardous reagents. By replacing expensive and hard-to-source TFDA with commodity chemicals like ethyl bromodifluoroacetate and sulfur powder, companies can achieve significant cost savings in their raw material expenditure without compromising on product quality. The stability and ease of handling of these reagents also reduce the logistical complexities and storage costs associated with hazardous chemical management, leading to a more resilient and agile supply chain capable of responding quickly to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex fluorinating reagents directly translates to lower production costs per kilogram. The use of inexpensive inorganic bases and elemental sulfur further drives down the variable costs, allowing for competitive pricing in the global market for fine chemical intermediates. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable manufacturing process.
- Enhanced Supply Chain Reliability: Reliance on widely available commodity chemicals mitigates the risk of supply disruptions that often plague specialty reagent markets. Ethyl bromodifluoroacetate and sulfur powder are produced by multiple suppliers globally, ensuring a stable and continuous supply of key inputs. This diversification of the supply base enhances the overall reliability of the production schedule, reducing lead times for high-purity pharmaceutical intermediates and enabling manufacturers to meet tight delivery deadlines with confidence.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process highly scalable and environmentally friendly. The ability to run the reaction at moderate temperatures reduces energy consumption, while the use of benign catalysts simplifies wastewater treatment and compliance with environmental regulations. This alignment with green chemistry principles not only reduces the environmental footprint but also future-proofs the manufacturing process against increasingly stringent regulatory requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity on its practical application and benefits. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industry professionals seeking to optimize their production workflows.
Q: What are the advantages of using ethyl bromodifluoroacetate over TFDA?
A: Ethyl bromodifluoroacetate is significantly more economical and commercially available compared to Trimethylsilyl Fluorosulfonyl Difluoroacetate (TFDA). Furthermore, this novel method avoids the use of strong bases required by TFDA protocols, thereby preserving sensitive functional groups on the substrate.
Q: Can this method be applied to selenium-containing derivatives?
A: Yes, the methodology is versatile. By substituting elemental sulfur powder with elemental selenium powder under similar reaction conditions, N-difluoromethyl azole selenourea derivatives can be successfully synthesized with good yields.
Q: Is this process suitable for large-scale manufacturing?
A: The process utilizes common organic solvents like DMA or DMF and operates at moderate temperatures (100°C), making it highly amenable to scale-up. The use of inexpensive reagents like sulfur powder further enhances its viability for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Difluoromethyl Imidazole Thiourea Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation pharmaceuticals and agrochemicals. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee batch-to-batch consistency and regulatory compliance.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, helping you identify opportunities for optimization and efficiency gains. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline and enhance your competitive advantage in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
