Scalable Synthesis of Trifluoromethyl Pyrazole Intermediates for Advanced Agrochemical and Pharmaceutical Applications
Scalable Synthesis of Trifluoromethyl Pyrazole Intermediates for Advanced Agrochemical and Pharmaceutical Applications
The rapid evolution of medicinal chemistry and crop protection science has increasingly demanded access to specialized heterocyclic building blocks that possess enhanced metabolic stability and lipophilicity. Patent CN101607941A introduces a groundbreaking methodology for the efficient construction of fluorine-containing pyrazole compounds, specifically targeting the 3-trifluoromethyl-5-(3'-mercaptopropylthio)pyrazole scaffold. This technological breakthrough addresses the critical need for reliable pharmaceutical intermediates supplier capabilities by offering a route that integrates the trifluoromethyl group directly during the ring-forming step, rather than through post-synthetic modification. The strategic incorporation of the trifluoromethyl moiety is paramount, as it significantly alters the electronic properties and binding affinity of the resulting molecules, thereby unlocking new possibilities for drug discovery and agrochemical formulation. By leveraging this patented acid-catalyzed cyclization strategy, manufacturers can achieve substantial improvements in process efficiency while maintaining the rigorous quality standards required for high-value fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for generating trifluoromethyl-substituted heterocycles often suffer from significant inefficiencies that hinder large-scale commercial adoption. Conventional methods frequently rely on the introduction of the fluorine atom after the heterocyclic core has been established, which typically necessitates the use of hazardous fluorinating agents, extreme reaction temperatures, or expensive transition metal catalysts. These legacy processes often result in poor atom economy, generate substantial quantities of toxic waste, and require complex purification protocols to remove residual heavy metals or unreacted fluorinating species. Furthermore, the structural diversity achievable through these older methods is frequently limited, as harsh reaction conditions can degrade sensitive functional groups or lead to unpredictable regioselectivity issues. For procurement managers seeking cost reduction in pharmaceutical intermediates manufacturing, these inefficiencies translate directly into higher raw material costs, extended production cycles, and increased regulatory burdens associated with waste disposal and safety compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a convergent synthesis strategy that constructs the pyrazole ring and installs the trifluoromethyl group simultaneously from a pre-functionalized precursor. This method employs fluorine-containing α-carbonyl enone dithioacetal compounds as key building blocks, which react smoothly with various hydrazine derivatives under mild acidic conditions. The reaction proceeds efficiently in common organic solvents such as ethanol or dimethylformamide at moderate temperatures around 80°C, eliminating the need for cryogenic conditions or high-pressure equipment. This streamlined protocol not only simplifies the operational workflow but also dramatically improves the overall yield and purity profile of the final product. By avoiding the use of exotic reagents and enabling a one-pot transformation, this technology represents a paradigm shift towards more sustainable and economically viable commercial scale-up of complex polymer additives and bioactive intermediates, ensuring a consistent supply of high-quality materials for downstream applications.
Mechanistic Insights into Acid-Catalyzed Cyclization
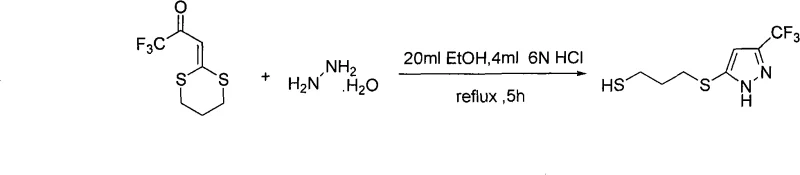
The core of this synthetic innovation lies in the precise mechanistic pathway governing the condensation and cyclization events. The reaction initiates with the nucleophilic attack of the hydrazine nitrogen on the electrophilic carbonyl carbon of the fluorinated dithioacetal precursor, facilitated by the protonation of the carbonyl oxygen by the acid catalyst. This initial addition forms a hydrazone intermediate, which subsequently undergoes an intramolecular cyclization via attack on the adjacent double bond or through tautomerization followed by ring closure. The presence of the electron-withdrawing trifluoromethyl group plays a crucial role in activating the system towards nucleophilic attack while stabilizing the resulting anionic intermediates throughout the transformation. The acid catalyst, typically hydrochloric acid or acetic acid, serves to lower the activation energy of the rate-determining steps, ensuring that the reaction proceeds to completion within a reasonable timeframe ranging from 5 to 48 hours depending on the specific substituents involved.
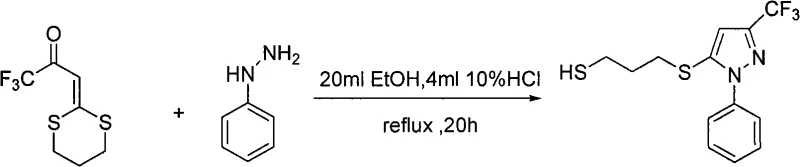
Following the formation of the pyrazole ring, the thioether side chain remains intact, preserving the valuable mercapto functionality for further derivatization or biological activity. The mechanism ensures high regioselectivity, predominantly yielding the 3-trifluoromethyl-5-substituted isomer, which is often the desired configuration for biological efficacy. Understanding this mechanistic nuance is vital for R&D directors aiming to optimize reaction parameters for new analogues, as slight modifications to the hydrazine component or the acid strength can fine-tune the reaction kinetics. The robustness of this mechanism allows for the tolerance of various functional groups, including nitro, chloro, and methyl substituents on the hydrazine aromatic ring, demonstrating the versatility of this catalytic system in generating diverse chemical libraries for screening purposes.
How to Synthesize 3-Trifluoromethyl-5-(3'-mercaptopropylthio)pyrazole Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to specific stoichiometric ratios and thermal profiles to maximize conversion and minimize byproduct formation. The general procedure involves mixing the fluorinated α-carbonyl ketene dithioacetal with the chosen hydrazine derivative in a molar ratio of approximately 1:1.25 to ensure complete consumption of the limiting reagent. The reaction mixture is then heated to reflux, typically around 80°C, in the presence of a catalytic amount of acid, such as 10% hydrochloric acid solution, for a duration determined by the reactivity of the specific hydrazine used. Upon completion, the workup involves standard extraction techniques using ethyl acetate, followed by washing with brine and drying over anhydrous magnesium sulfate to remove residual water and inorganic salts. The crude product is then purified via silica gel column chromatography using a gradient of petroleum ether and ethyl acetate to isolate the pure fluorinated pyrazole compound.
- Mix fluorine-containing alpha-carbonyl ketene dithioacetal and hydrazine derivatives in a molar ratio of 1: 1.25 within an organic solvent such as ethanol.
- Add acid catalyst (e.g., 10% HCl) and maintain the reaction system temperature at 80°C under reflux conditions for 5 to 48 hours depending on substrate reactivity.
- Remove solvent, extract residue with ethyl acetate, wash with brine, dry over magnesium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers compelling advantages that directly impact the bottom line and operational resilience of chemical supply chains. The elimination of complex multi-step sequences and hazardous fluorination reagents translates into a significantly simplified manufacturing process that requires less specialized equipment and lower energy input. This streamlining of the production workflow inherently reduces the risk of batch failures and quality deviations, thereby enhancing the reliability of supply for critical downstream customers who depend on consistent material availability for their own formulation processes. Furthermore, the use of commodity chemicals such as ethanol and hydrochloric acid as solvents and catalysts ensures that the raw material supply chain is robust and less susceptible to geopolitical disruptions or market volatility associated with specialty reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy and the avoidance of expensive post-synthetic fluorination steps. By utilizing a pre-fluorinated building block that reacts efficiently in a single pot, manufacturers can drastically reduce the consumption of solvents, reagents, and labor hours per kilogram of product. The mild reaction conditions also extend the lifespan of reactor vessels and reduce maintenance costs associated with corrosion from harsh acids or bases. Additionally, the high yields reported in the patent examples suggest that waste generation is minimized, leading to lower disposal costs and a smaller environmental footprint, which aligns with modern sustainability goals and regulatory incentives for green chemistry practices.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available starting materials, such as hydrazine hydrates and substituted phenylhydrazines, ensures that production schedules can be maintained without interruption due to raw material shortages. The flexibility of the synthesis to accommodate various substituents allows for the rapid switching between different product variants within the same production line, providing agility in responding to changing market demands. This adaptability is crucial for maintaining service levels in the fast-paced pharmaceutical and agrochemical sectors, where time-to-market is a critical competitive factor. The robust nature of the reaction also implies that scale-up from laboratory to commercial production can be achieved with minimal re-optimization, reducing the time and capital investment required to bring new products to market.
- Scalability and Environmental Compliance: The process is inherently scalable due to its homogeneous nature and the absence of gas-evolving steps or highly exothermic events that are difficult to control on a large scale. The use of standard extraction and chromatography techniques for purification facilitates easy integration into existing manufacturing infrastructure without the need for major capital expenditures. From an environmental standpoint, the reduction in hazardous waste and the use of less toxic solvents contribute to easier compliance with increasingly stringent environmental regulations. This proactive approach to environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible partner in the global supply chain, appealing to customers who prioritize sustainability in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated pyrazole intermediates, based on the detailed specifications provided in the patent literature. Understanding these nuances helps stakeholders make informed decisions about integrating this technology into their existing portfolios. The answers reflect the specific capabilities and limitations observed during the experimental validation of the process, ensuring transparency and accuracy in our technical communications.
Q: What are the primary advantages of this fluorinated pyrazole synthesis method?
A: The method described in patent CN101607941A offers a simple preparation process with great structural adjustability. It utilizes readily available starting materials and mild acidic conditions, resulting in low production costs and environmental friendliness compared to traditional multi-step fluorination routes.
Q: What is the scope of substituents allowed on the pyrazole ring?
A: The synthesis supports a wide range of substituents. R1 can be a perfluoro C1-C4 alkyl group, while R2 accommodates hydrogen, halogens, nitro, or cyano groups. The N-substituent (R4) can vary from hydrogen and methyl to aryl and substituted aryl groups, allowing for diverse library generation.
Q: What are the potential applications for these compounds?
A: These fluorine-containing pyrazole compounds serve as novel target compounds widely applicable in the medical and pesticide fields. The trifluoromethyl group enhances lipophilicity and metabolic stability, making them ideal candidates for bioactive molecule development in both pharmaceutical and agrochemical sectors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine-Containing Pyrazole Compounds Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced synthesis technology to deliver high-performance intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of even the largest multinational corporations. We operate state-of-the-art facilities equipped with rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of fluorinated pyrazole compound meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to quality and consistency makes us the preferred partner for companies seeking to secure their supply chain for critical building blocks.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate the potential of these fluorine-containing compounds for your next generation of bioactive products with confidence and clarity.
