Advanced Dynamic Resolution Technology for Alpha-Substituted-2-Aminoacetamides
The pharmaceutical and agrochemical industries rely heavily on the availability of high-purity chiral intermediates to ensure the efficacy and safety of final active ingredients. Patent CN101955439A introduces a groundbreaking methodology for the high-yield resolution of alpha-substituted-2-aminoacetamides, a class of compounds that serves as a pivotal backbone for numerous therapeutic agents and crop protection chemicals. Unlike conventional static resolution techniques that inherently cap production efficiency at 50%, this invention leverages a sophisticated dynamic kinetic resolution strategy. By integrating the asymmetric conversion of diastereomers directly into the crystallization process, the technology transforms what was once a wasteful separation into a highly efficient, near-quantitative synthesis. This report analyzes the technical merits of this approach, offering critical insights for R&D directors seeking robust synthetic routes and procurement leaders aiming for sustainable supply chain optimization.
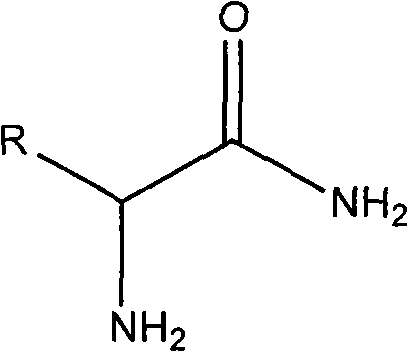
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of optically pure alpha-substituted-2-aminoacetamides has been plagued by significant inefficiencies inherent to classical resolution techniques. In a standard diastereomeric salt formation process, a racemic mixture is reacted with a single enantiomer of a resolving agent, resulting in two diastereomeric salts with different solubilities. While one salt crystallizes out, the other remains in the mother liquor and is typically discarded or subjected to a separate, often cumbersome, racemization process. This fundamental limitation restricts the maximum theoretical yield to merely 50% of the starting material, effectively doubling the raw material costs and waste generation for every kilogram of product produced. Furthermore, prior art methods, such as those described in CN1583721, attempted to address this by separating the unwanted isomer for subsequent racemization. However, these approaches frequently suffered from the co-precipitation of inorganic salts like sodium chloride or sodium tartrate due to the use of strong bases, complicating purification and reducing the optical purity of the final product. Other attempts involved complex multi-step racemization sequences that lowered overall throughput and introduced additional unit operations, thereby increasing capital expenditure and operational complexity.
The Novel Approach
The methodology disclosed in CN101955439A represents a paradigm shift by merging the resolution and racemization steps into a single, cohesive dynamic system. Instead of treating the unwanted enantiomer as waste, this process employs catalytic amounts of aldehydes or ketones to facilitate the continuous in situ interconversion of the stereocenters. This allows the "wrong" isomer remaining in the solution to constantly equilibrate and eventually convert into the "right" isomer that precipitates as the desired diastereomeric salt. By maintaining specific reaction conditions, including a controlled pH range of 4 to 7 and temperatures between 60°C and 80°C, the system drives the equilibrium towards the crystallizing product. This eliminates the need for isolating and separately processing the mother liquor isomer, effectively bypassing the 50% yield ceiling. The result is a streamlined workflow that maximizes atom economy, significantly reduces solvent consumption, and delivers optical purities (ee values) exceeding 90% with total yields approaching 98% when mother liquor recycling is employed.
Mechanistic Insights into Aldehyde/Ketone Catalyzed Dynamic Resolution
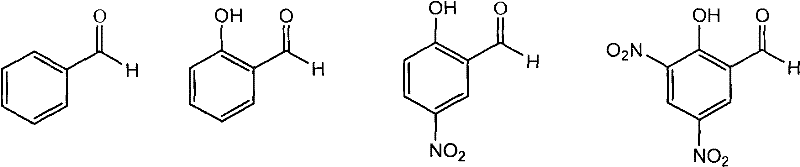
The core innovation of this technology lies in the catalytic role played by carbonyl compounds, specifically aromatic aldehydes like benzaldehyde and salicylaldehyde, or ketones such as acetone and butanone. Mechanistically, these additives interact with the free amine group of the alpha-substituted-2-aminoacetamide to form transient Schiff base intermediates (imines). The formation of the imine renders the alpha-proton adjacent to the imine nitrogen significantly more acidic than in the free amine form. This increased acidity facilitates rapid deprotonation and reprotonation events under the mildly acidic to neutral conditions maintained in the reactor. Consequently, the stereochemical integrity at the alpha-carbon is temporarily lost, allowing for racemization to occur dynamically within the reaction medium. As the chiral resolving agent selectively precipitates the desired enantiomer as an insoluble salt, Le Chatelier's principle drives the equilibrium forward, continuously pulling the racemizing species from the solution phase into the solid phase. This elegant interplay between reversible imine formation and selective crystallization ensures that virtually all starting material is funneled into the desired product stream.
Controlling the impurity profile is critical in pharmaceutical intermediate manufacturing, and this process offers distinct advantages in that regard. By avoiding the use of strong inorganic bases like sodium hydroxide, which were common in earlier resolution attempts, the formation of inorganic salt byproducts is effectively suppressed. The use of organic acids such as acetic acid, oxalic acid, or phosphoric acid to maintain the pH between 5 and 6 ensures that the reaction environment remains homogeneous and free from precipitating inorganics that could occlude within the crystal lattice of the product. Furthermore, the selection of specific resolving agents like L-tartrate or D-camphorsulfonic acid allows for fine-tuning the solubility differences between the diastereomers. For instance, the use of aromatic aldehydes has been observed to be more beneficial than aliphatic ones, likely due to the stability and electronic properties of the resulting aromatic imine intermediates, which optimize the rate of racemization without compromising the stability of the final amide bond. This precise control results in a product with high enantiomeric excess, often surpassing 94% ee in a single crystallization step.
How to Synthesize Alpha-Substituted-2-Aminoacetamide Efficiently
The implementation of this dynamic resolution process requires careful attention to solvent selection, temperature control, and the stoichiometric balance between the substrate, the chiral resolving agent, and the carbonyl catalyst. The patent outlines a robust procedure where the racemic amide is first dissolved in a lower alcohol, followed by the addition of the catalyst and acid before the slow introduction of the resolving agent. This sequence is vital to establish the dynamic equilibrium before precipitation begins. For a detailed breakdown of the specific operational parameters, reagent grades, and workup procedures required to replicate these high-yield results in a pilot or production setting, please refer to the standardized synthesis guide below.
- Dissolve the racemic alpha-substituted-2-aminoacetamide in a lower alcohol solvent such as methanol or ethanol under stirring.
- Add a catalytic amount of an aldehyde (e.g., benzaldehyde, salicylaldehyde) or ketone (e.g., acetone) along with a weak acid to adjust the pH to between 4 and 7.
- Heat the mixture to 60-80°C and slowly add a solution of a chiral resolving agent (e.g., L-tartrate, D-camphorsulfonic acid) to precipitate the desired diastereomeric salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from static to dynamic resolution technologies offers profound economic and logistical benefits. The primary value driver is the drastic improvement in material efficiency; by pushing yields from a theoretical maximum of 50% to nearly 100%, the demand for raw starting materials is effectively halved for the same output volume. This reduction in raw material intensity directly translates to significant cost savings and a smaller environmental footprint, aligning with modern green chemistry mandates. Additionally, the elimination of separate racemization steps simplifies the manufacturing workflow, reducing the number of required reactors, filtration units, and drying cycles. This process intensification not only lowers capital investment requirements for new production lines but also shortens the overall cycle time per batch, enhancing the agility of the supply chain to respond to market fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the complete utilization of the racemic feedstock. In traditional processes, half of the valuable chiral precursor is essentially wasted, representing a direct loss of capital. By recovering this material through in situ conversion, the effective cost per kilogram of the active intermediate is substantially reduced. Furthermore, the avoidance of inorganic salt byproducts simplifies the downstream purification and waste treatment processes. There is no need for expensive ion-exchange resins or extensive washing protocols to remove entrapped salts, which reduces utility consumption and waste disposal fees. The use of catalytic amounts of relatively inexpensive aldehydes or ketones, rather than stoichiometric quantities of exotic reagents, further optimizes the bill of materials, ensuring a highly competitive cost structure for large-scale production.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex, multi-step syntheses that have multiple points of failure. This streamlined one-pot dynamic resolution minimizes operational risks by reducing the number of unit operations and intermediate isolations. The robustness of the process, demonstrated by its tolerance to various substituents on the R-group (from simple alkyls to aromatic rings), ensures that the same platform technology can be applied to a wide portfolio of intermediates. This versatility allows manufacturers to utilize existing equipment for multiple products without extensive retooling. Moreover, the high yields mean that less inventory of starting materials needs to be held in stock to meet production targets, freeing up working capital and reducing the risk of supply disruptions associated with sourcing large volumes of precursors.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden bottlenecks related to heat transfer and mixing, particularly in heterogeneous systems. This resolution method operates in a predominantly homogeneous liquid phase until the final crystallization, making it highly amenable to scale-up from laboratory to multi-tonne commercial reactors. The mild reaction conditions (60-80°C) and the use of common solvents like methanol and ethanol reduce energy consumption compared to high-temperature or high-pressure alternatives. From an environmental perspective, the near-quantitative atom economy significantly reduces the E-factor (mass of waste per mass of product). The absence of heavy metal catalysts or hazardous inorganic bases simplifies effluent treatment, ensuring compliance with increasingly stringent global environmental regulations regarding wastewater discharge and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dynamic resolution technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What is the primary advantage of this dynamic resolution method over traditional resolution?
A: Traditional resolution is limited to a maximum theoretical yield of 50% because the unwanted enantiomer is discarded. This novel method utilizes catalytic aldehydes or ketones to facilitate the asymmetric conversion of the unwanted isomer in situ, allowing for total utilization of the racemic starting material and achieving yields up to 98%.
Q: Which resolving agents are compatible with this process?
A: The process is highly versatile and supports a wide range of chiral acids including L- or D-tartrates, oxysuccinic acid (malic acid), and camphorsulfonic acid. The choice depends on the specific solubility characteristics of the target diastereomeric salt.
Q: Can this method be applied to the synthesis of Levetiracetam intermediates?
A: Yes, this method is specifically effective for resolving alpha-ethyl-2-aminoacetamide, which is the critical chiral intermediate for the antiepileptic drug Levetiracetam. It overcomes previous issues with inorganic salt contamination and low racemization yields found in older patents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Substituted-2-Aminoacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of chiral drugs depends on access to advanced synthetic technologies like the dynamic resolution process described in CN101955439A. Our R&D team has extensively studied this methodology and possesses the expertise to adapt it for the specific needs of your target molecule, whether it be for antiepileptic agents like Levetiracetam or novel agrochemical actives. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields observed in the lab are faithfully reproduced on an industrial scale. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed pharmacopeial standards.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. By leveraging our technical capabilities, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot batches and to discuss route feasibility assessments for your proprietary compounds. Let us help you transform complex chiral synthesis challenges into reliable, cost-effective commercial realities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
