Advanced Synthesis of Benzimidazole Proton Pump Inhibitor Intermediates via Direct Chlorination
Advanced Synthesis of Benzimidazole Proton Pump Inhibitor Intermediates via Direct Chlorination
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of Proton Pump Inhibitors (PPIs), a class of drugs essential for managing gastric acid-related disorders. Patent CN103664885A introduces a transformative approach to synthesizing key benzimidazole-type PPI intermediates, specifically targeting the thioether linkage formation found in major drugs like Omeprazole, Rabeprazole, and Lansoprazole. This technology addresses long-standing inefficiencies in the supply chain by replacing cumbersome multi-step sequences with a streamlined, direct chlorination strategy. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates while drastically reducing the environmental footprint and operational costs associated with traditional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-chloromethylpyridine derivatives, which are pivotal precursors for PPI synthesis, has relied on a convoluted and inefficient pathway. The conventional method typically involves the reaction of 2-methylpyridine N-oxide derivatives with acetic anhydride to induce an intramolecular rearrangement, forming an acetyloxymethyl intermediate. This species must then undergo hydrolysis in sodium hydroxide solution to yield a hydroxymethyl derivative, which is finally chlorinated using reagents such as thionyl chloride. This three-step sequence is not only operationally繁琐 (tedious) but also suffers from cumulative yield losses at each isolation stage. Furthermore, the use of acetic anhydride and subsequent hydrolysis generates significant aqueous waste, complicating environmental compliance and increasing the overall cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN103664885A simplifies this landscape by employing phosphorus oxychloride (POCl3) as a superior chlorinating agent that acts directly on the 2-methylpyridine N-oxide substrate. This innovation eliminates the need for the initial acylation and subsequent hydrolysis steps, collapsing the synthesis into a single, highly efficient chlorination event. By utilizing a specific solvent system and controlled addition protocols, the process achieves high conversion rates without generating the complex by-product profiles associated with the older acetic anhydride route. This direct approach not only accelerates the production timeline but also enhances the overall atom economy, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to offer cost-effective solutions to global API manufacturers.
Mechanistic Insights into POCl3-Mediated Direct Chlorination
The core of this technological advancement lies in the unique activation of the methyl group adjacent to the pyridine nitrogen through the N-oxide functionality. When phosphorus oxychloride is introduced to the reaction mixture containing the 2-methylpyridine N-oxide derivative, it facilitates the substitution of a hydrogen atom on the methyl group with a chlorine atom. Crucially, the presence of triethylamine acts as an acid-binding agent that serves a dual purpose: it scavenges the hydrogen chloride generated during the reaction to drive the equilibrium forward, and its lone pair electrons interact with the oxygen atom of the N-oxide. This interaction enhances the electron-withdrawing capability of the N-oxide moiety, thereby increasing the acidity of the adjacent methyl protons and making them more susceptible to chlorination. This mechanistic nuance ensures that the reaction proceeds with high specificity towards the desired 2-chloromethyl product rather than attacking the pyridine ring itself.
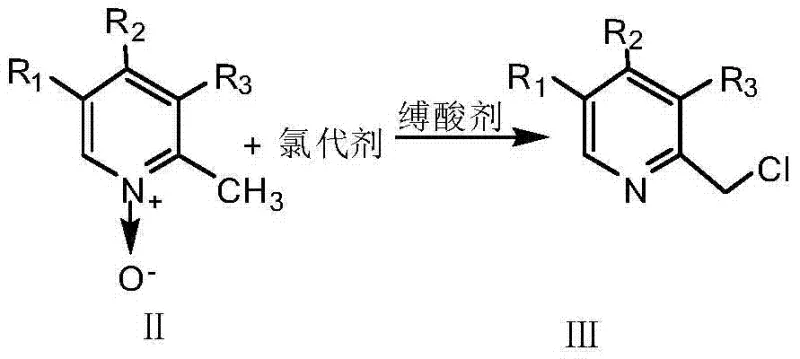
Furthermore, the patent highlights a critical procedural detail regarding the order of reagent addition that is vital for maintaining product integrity. If phosphorus oxychloride were added directly to a solution containing excess triethylamine, it would preferentially react with the amine, deactivating the chlorinating agent. Conversely, if the N-oxide reacts with phosphorus oxychloride for too long without the acid scavenger, there is a risk of forming unwanted 6-chloropyridine derivatives due to electrophilic attack on the ring. The optimized protocol dictates an initial partial addition of the chlorinating agent followed by the simultaneous, rate-controlled dropwise addition of the acid-binding agent. This precise kinetic control ensures that the concentration of free acid is managed effectively, suppressing side reactions and guaranteeing that the 2-chloromethylpyridine derivative is generated as the predominant species, ready for the subsequent condensation step.
How to Synthesize Rabeprazole Intermediate Efficiently
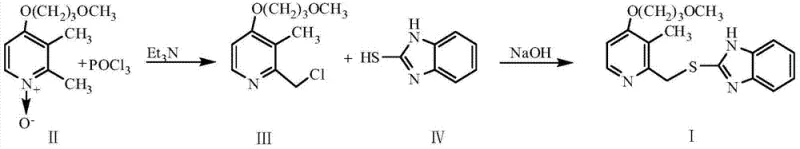
To demonstrate the practical applicability of this novel route, we examine the synthesis of the Rabeprazole intermediate, 2-{[4-(3-methoxypropoxy)-3-methylpyridin-2-yl]methylthio}-1H-benzimidazole. The process begins by dissolving the specific pyridine N-oxide precursor in benzene and heating the mixture to a controlled temperature range of 60-90°C. Phosphorus oxychloride and triethylamine, separately diluted in dichloromethane, are introduced according to the strict addition protocol mentioned previously to generate the reactive 2-chloromethyl intermediate in situ. Without isolating this potentially unstable chloromethyl species, the reaction mixture is processed to remove inorganic salts and then directly subjected to condensation with 2-mercaptobenzimidazole in an alkaline ethanol medium. This telescoped operation minimizes handling losses and exposure to moisture, which is critical for maintaining the stability of the chloromethyl group before it forms the stable thioether bond.
- Dissolve 2-picoline N-oxide derivative in benzene and react with phosphorus oxychloride and triethylamine at 60-90°C using a specific dropwise addition protocol to form the chloromethyl intermediate.
- Without purification, react the crude chloromethyl intermediate with a 2-mercapto-benzimidazole derivative in sodium hydroxide ethanol solution under reflux.
- Perform workup via extraction with dichloromethane, followed by recrystallization using ethyl acetate and petroleum ether to isolate the high-purity thioether product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. The elimination of multiple reaction steps and intermediate isolations translates directly into a reduction in processing time and labor costs. By removing the requirement for acetic anhydride and the subsequent hydrolysis phase, the process significantly lowers the consumption of raw materials and reduces the volume of wastewater generated, aligning with modern green chemistry principles. This efficiency gain allows for a more agile response to market demand fluctuations, ensuring a steady flow of high-quality intermediates without the bottlenecks typical of legacy manufacturing processes.
- Cost Reduction in Manufacturing: The substitution of the traditional multi-step sequence with a direct chlorination method using phosphorus oxychloride results in substantial cost savings. Phosphorus oxychloride is a widely available and inexpensive reagent compared to the combined cost of acetic anhydride and thionyl chloride used in older methods. Moreover, the ability to telescope the reaction—using the crude chloromethyl intermediate directly in the next step without purification—drastically reduces solvent usage and energy consumption associated with drying and distillation, leading to a lower overall cost base for the production of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the number of potential failure points in the manufacturing process. With fewer unit operations and less reliance on sensitive intermediate isolations, the risk of batch failures due to contamination or degradation is minimized. This robustness ensures a more predictable lead time for the delivery of high-purity pharmaceutical intermediates. Additionally, the reagents employed are standard industrial chemicals with stable supply chains, mitigating the risk of raw material shortages that could disrupt production schedules for critical gastro-intestinal medications.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents like benzene (or suitable alternatives) and dichloromethane which are easily recovered and recycled in large-scale reactors. The reduction in waste generation, particularly the avoidance of acetate-heavy waste streams from the acetic anhydride route, simplifies effluent treatment requirements. This makes the technology highly suitable for commercial scale-up of complex pharmaceutical intermediates in facilities operating under strict environmental regulations, ensuring long-term sustainability and compliance without compromising on output volume.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis pathway. Understanding these nuances is essential for process chemists and engineers looking to integrate this technology into their existing manufacturing frameworks. The answers are derived directly from the experimental data and mechanistic explanations provided in the patent literature, ensuring accuracy and relevance for industrial application.
Q: Why is phosphorus oxychloride preferred over traditional acetic anhydride methods?
A: Phosphorus oxychloride allows for direct chlorination of the methyl group on the pyridine N-oxide, eliminating the need for a multi-step rearrangement, hydrolysis, and subsequent chlorination sequence, thereby reducing operational complexity and cost.
Q: What is the critical parameter for controlling by-products in this synthesis?
A: The order of reagent addition is critical; adding phosphorus oxychloride prior to the simultaneous dropwise addition of the acid-binding agent prevents the formation of unwanted 6-chloropyridine derivatives and ensures high regioselectivity.
Q: How does this method impact the purity of the final PPI intermediate?
A: By avoiding complex intermediate isolations and utilizing a telescoped process where the crude chloromethyl species reacts directly, the method minimizes product loss and achieves HPLC purity levels exceeding 95-98% after simple recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of generic and branded pharmaceutical production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs which utilize advanced analytical techniques to verify the identity and quality of every batch of benzimidazole intermediates we supply to our global partners.
We invite you to collaborate with us to optimize your supply chain for Proton Pump Inhibitor intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance the efficiency and profitability of your API production lines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
