Advanced One-Pot Synthesis of 3,3'-Diaminobenzophenone for High-Performance Polyimide Manufacturing
The global demand for high-performance thermoplastic polyimide (TPI) films, particularly for flexible copper-clad laminates in the electronics sector, has necessitated a rigorous re-evaluation of monomer synthesis strategies. Patent CN109400481B introduces a transformative one-pot methodology for producing 3,3'-diaminobenzophenone, a critical diamine monomer, addressing the severe limitations of legacy manufacturing processes. This innovative route leverages dinitrogen pentoxide (N2O5) for selective nitration followed by in-situ catalytic hydrogenation, achieving yields exceeding 75% and purity levels greater than 99%. For R&D directors and procurement specialists, this patent represents a pivotal shift away from corrosive mixed-acid systems toward a greener, more efficient paradigm that aligns with modern sustainability mandates. By integrating nitration and reduction into a single reactor sequence, the process minimizes material handling losses and significantly curtails the generation of hazardous waste streams, offering a compelling value proposition for reliable electronic chemical supplier partnerships aiming to secure long-term supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 3,3'-diaminobenzophenone has been plagued by inefficient multi-step protocols that rely on harsh reagents and generate substantial environmental liabilities. Traditional nitration methods utilizing mixed acids (concentrated sulfuric and nitric acid) suffer from poor atom economy and severe equipment corrosion, often resulting in yields as low as 52% due to extensive side reactions and oxidative degradation. Furthermore, the subsequent reduction step typically employs tin(II) chloride (SnCl2) in hydrochloric acid, a process that not only yields less than 40% conversion but also produces massive quantities of toxic tin-containing sludge and acidic wastewater. These legacy techniques impose heavy burdens on waste treatment facilities and increase the total cost of ownership through expensive disposal fees and frequent reactor maintenance, rendering them increasingly untenable for cost reduction in polyimide monomer manufacturing in a regulated global market.
The Novel Approach
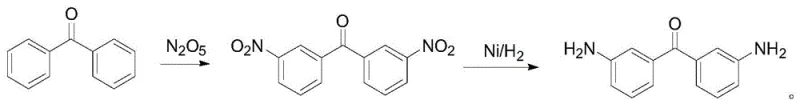
In stark contrast, the novel one-pot strategy disclosed in the patent utilizes dinitrogen pentoxide dissolved in dichloromethane as a mild yet potent nitrating agent, facilitated by an HZSM-4 molecular sieve catalyst. This system allows for the quantitative conversion of benzophenone to the intermediate 3,3'-dinitrobenzophenone without the formation of waste acid, operating under mild temperatures of 25-30°C. Crucially, the process eliminates the need for intermediate isolation; instead, nickel powder is introduced directly into the reaction vessel to catalyze the hydrogenation of the nitro groups to amines under moderate hydrogen pressure (0.5-1.5 MPa). This telescoped approach not only streamlines the workflow by removing unit operations but also ensures that the final product achieves a purity of over 99% after simple recrystallization, demonstrating superior scalability and environmental compliance compared to conventional batch processes.
Mechanistic Insights into N2O5-Mediated Nitration and Ni-Catalyzed Hydrogenation
The core chemical innovation lies in the electrophilic aromatic substitution mechanism driven by the nitronium ion (NO2+) generated from dinitrogen pentoxide. Unlike proton-based acid systems, N2O5 provides a non-aqueous nitration environment that suppresses hydrolysis and oxidation side reactions, ensuring high regioselectivity for the meta-position on the benzophenone rings. The HZSM-4 zeolite catalyst plays a dual role, acting as a solid acid to promote the formation of the active nitronium species while its porous structure may offer shape selectivity that further enhances the ratio of the desired 3,3'-isomer. Following nitration, the introduction of nickel powder initiates a heterogeneous catalytic cycle where molecular hydrogen is adsorbed and dissociated on the metal surface. This activated hydrogen is then transferred to the nitro groups of the dissolved intermediate, sequentially reducing them through nitroso and hydroxylamine intermediates to the final amine functionality, all within the same solvent matrix.
From an impurity control perspective, this mechanism is exceptionally clean because it avoids the redox-active metal salts found in tin-based reductions, which often leave difficult-to-remove metal residues in the final API or polymer precursor. The use of a heterogeneous nickel catalyst allows for easy removal via simple filtration post-reaction, preventing metal contamination that could degrade the thermal or electrical properties of the resulting polyimide film. Additionally, the mild reaction conditions prevent the thermal decomposition of the sensitive benzophenone backbone, a common issue in high-temperature acid nitrations. This precise control over the reaction pathway ensures a narrow impurity profile, which is critical for applications requiring high-purity 3,3'-diaminobenzophenone where trace contaminants can act as chain terminators or defects in the polymer matrix.
How to Synthesize 3,3'-Diaminobenzophenone Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the nitrating agent and the pressure parameters during the hydrogenation phase to maximize yield and safety. The process begins with the dissolution of benzophenone in a chlorinated solvent such as dichloromethane or 1,2-dichloroethane, followed by the controlled addition of the N2O5 solution to manage the exotherm effectively. Once the nitration is complete, the system is purged and pressurized with hydrogen in the presence of the nickel catalyst, maintaining a temperature range of 25-30°C to ensure complete reduction without over-hydrogenation of the carbonyl group. The detailed standardized synthetic steps, including specific molar ratios, stirring rates, and workup procedures derived from the patent examples, are outlined below for technical reference.
- Dissolve benzophenone in dichloromethane with HZSM-4 catalyst and react with dinitrogen pentoxide solution at 25-30°C to form 3,3'-dinitrobenzophenone.
- Without isolation, add nickel powder to the reaction mixture and introduce hydrogen gas at 0.5-1.5 MPa pressure to reduce the nitro groups.
- Filter the catalyst, evaporate the solvent, and recrystallize the crude product from ethanol/water to obtain high-purity 3,3'-diaminobenzophenone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this one-pot methodology offers profound strategic benefits beyond mere technical elegance, primarily driven by the simplification of the manufacturing footprint and the elimination of hazardous waste liabilities. By consolidating two distinct chemical transformations into a single reactor campaign, manufacturers can drastically reduce cycle times and energy consumption associated with heating, cooling, and solvent swapping between steps. The avoidance of corrosive mixed acids and toxic tin salts translates directly into lower capital expenditure for specialized corrosion-resistant equipment and significantly reduced operational costs for waste neutralization and disposal. Furthermore, the ability to recycle the chlorinated solvent after simple distillation enhances the overall material efficiency, contributing to substantial cost savings and a smaller environmental footprint, which is increasingly vital for meeting corporate sustainability goals in the electronic materials sector.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like concentrated sulfuric acid and tin chloride removes significant line items from the bill of materials, while the high atom economy of the N2O5 nitration ensures that raw material spend is optimized. The one-pot nature of the process reduces labor hours and utility costs by minimizing the number of unit operations, such as filtration, drying, and solvent exchange, required to isolate intermediates. Consequently, the overall production cost per kilogram is significantly lowered, allowing for more competitive pricing structures in the global market for high-purity electronic chemicals without compromising on quality margins.
- Enhanced Supply Chain Reliability: Relying on readily available starting materials like benzophenone and robust catalysts like nickel powder mitigates the risk of supply disruptions often associated with specialty reagents or regulated precursors. The mild reaction conditions (ambient temperature and moderate pressure) reduce the likelihood of unplanned shutdowns due to thermal runaways or equipment failures, ensuring a consistent and predictable production schedule. This operational stability is crucial for reducing lead time for high-purity electronic chemicals, enabling suppliers to respond more agilely to fluctuating demand from downstream polyimide film manufacturers and securing long-term contracts with key accounts.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex polymer intermediates, as the absence of strong exotherms and the use of heterogeneous catalysis simplify heat management and product separation at larger volumes. The significant reduction in three-waste emissions (waste water, waste gas, and solid waste) aligns with stringent environmental regulations, minimizing the regulatory burden and permitting risks associated with expanding production capacity. This eco-friendly profile not only future-proofs the manufacturing site against tightening legislation but also enhances the brand reputation of the supplier as a responsible partner in the green chemistry initiative.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, drawing directly from the experimental data and beneficial effects reported in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this technology for their own production lines or for procurement professionals assessing the quality assurances of potential suppliers. The answers provided reflect the specific advantages of the N2O5/Ni-H2 system over traditional methods, focusing on yield optimization, purity standards, and operational safety.
Q: What are the advantages of using N2O5 over mixed acid for nitration?
A: Using dinitrogen pentoxide (N2O5) eliminates the generation of large quantities of waste acid associated with traditional sulfuric/nitric acid mixtures, significantly reducing environmental burden and equipment corrosion risks while improving atom economy.
Q: How does the one-pot method impact production costs?
A: The one-pot approach avoids the isolation and purification of the intermediate 3,3'-dinitrobenzophenone, saving solvent usage, energy consumption, and labor time, thereby drastically lowering the overall manufacturing cost.
Q: Is the nickel catalyst suitable for large-scale industrial production?
A: Yes, nickel powder is a robust, heterogeneous catalyst that operates under mild conditions (25-30°C) and moderate pressure, making it highly scalable and safer than homogeneous reduction methods involving tin chloride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Diaminobenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of advanced polyimide materials, and we possess the technical expertise to translate laboratory innovations like Patent CN109400481B into robust industrial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3,3'-diaminobenzophenone meets the exacting standards required for flexible copper-clad laminate applications, providing our partners with unwavering confidence in material consistency.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains and leverage these advanced synthetic methodologies for tangible business growth. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments to validate the commercial viability of this process for your operations. Let us help you secure a sustainable and cost-effective source of this vital electronic chemical, driving innovation and efficiency in your polymer manufacturing endeavors.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
