Advanced Metal-Free Synthesis of 3,8-Dipyrrole-BODIPY for High-Performance Optical Applications
The landscape of fluorescent dye manufacturing is undergoing a significant transformation driven by the demand for near-infrared (NIR) emitters with superior photostability and quantum yields. Patent CN111187288B introduces a groundbreaking preparation method for 3,8-dipyrrole-BODIPY fluorescent dyes, addressing critical bottlenecks in traditional synthetic routes. This technology leverages a direct oxidative nucleophilic reaction between a specific BOPHY precursor and pyrrole, bypassing the complex multi-step sequences that have historically plagued this chemical class. For R&D directors and procurement specialists in the optoelectronic and fine chemical sectors, this innovation represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms. The ability to access high-purity NIR dyes without relying on scarce transition metals opens new avenues for scalable production of advanced imaging agents and sensor materials.
Furthermore, the structural integrity of the resulting 3,8-dipyrrole-BODIPY core ensures exceptional performance in qualitative detection applications, particularly for hypochlorite ions. The patent details a robust protocol that not only enhances the fluorescence emission wavelength through extended conjugation but also guarantees a cleaner impurity profile. This is crucial for applications requiring stringent quality control, such as biological tissue imaging and electronic display components. By integrating this novel synthetic strategy, manufacturers can achieve a reliable supply of high-performance fluorescent materials while adhering to increasingly rigorous environmental and safety standards.
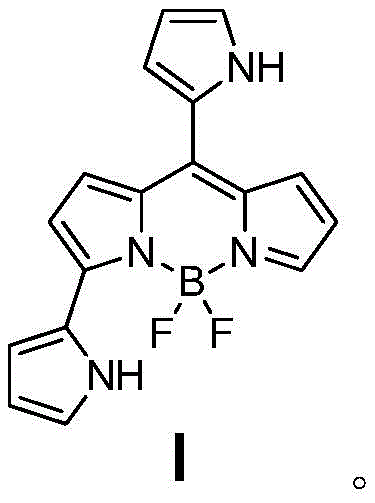
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of modified BODIPY cores has been fraught with inefficiencies that hinder commercial viability. Prior art, such as the methodology reported by the Ravikanth group in 2016, relied on a cumbersome sequence involving the initial synthesis of tripyrrole followed by oxidation with DDQ and subsequent nucleophilic substitution. This multi-step approach suffered from extremely low overall yields, often hovering around 5%, primarily due to the formation of unstable intermediate species that degraded before final conversion. Additionally, the reliance on harsh oxidants and the necessity for precise stoichiometric control increased the operational complexity and waste generation. For industrial-scale operations, these factors translate into prohibitive costs and significant supply chain vulnerabilities, making the consistent production of high-quality BODIPY derivatives a challenging endeavor.
The Novel Approach
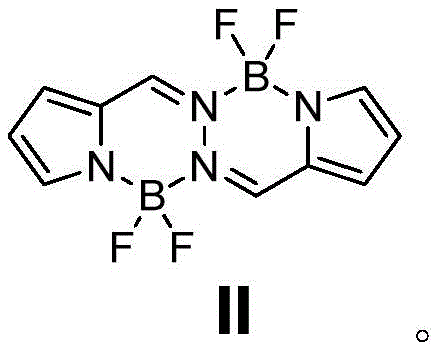
In stark contrast, the method disclosed in patent CN111187288B streamlines the entire process into a single, efficient oxidative nucleophilic reaction. By utilizing a pre-formed BOPHY compound (Formula II) as the starting material, the reaction directly installs the requisite pyrrole groups at the 3 and 8 positions in one pot. This strategic simplification eliminates the isolation of unstable intermediates, thereby preserving material throughput and significantly boosting the final isolated yield to approximately 19%. The absence of transition metal catalysts further distinguishes this approach, aligning perfectly with green chemistry principles. This reduction in synthetic complexity not only lowers the barrier to entry for production but also enhances the reproducibility of the process, ensuring that every batch meets the high standards required for commercial distribution.
Mechanistic Insights into Oxidative Nucleophilic Substitution
The core of this technological advancement lies in the unique reactivity of the BOPHY scaffold under thermal conditions. The reaction mechanism involves the activation of the BOPHY core, which acts as an electrophilic center, facilitating the attack by the nucleophilic pyrrole molecules. Unlike traditional methods that require external oxidants like DDQ to drive the aromatization and coupling steps, this thermal process promotes the necessary oxidative transformations intrinsically within the reaction matrix. The elevated temperature range of 120-160°C provides the activation energy needed to overcome the kinetic barriers, allowing for the smooth displacement of leaving groups and the subsequent formation of the stable boron-dipyrromethene complex. This mechanistic pathway avoids the generation of radical species that often lead to polymerization or side reactions, resulting in a cleaner reaction profile.
From an impurity control perspective, the metal-free nature of this catalysis is paramount. Traditional transition metal-catalyzed couplings often leave trace residues of palladium, copper, or nickel, which can quench fluorescence or cause toxicity in biological applications. By eliminating these metals entirely, the process inherently produces a product with a superior purity profile, reducing the burden on downstream purification units. The structural analysis confirms that the newly formed C-N bonds maintain normal bond lengths, and the dihedral angles between the pyrrole rings provide steric protection against aggregation. This structural rigidity not only enhances solubility in various organic solvents but also stabilizes the excited state, leading to the observed redshift in fluorescence emission and improved photophysical properties essential for high-end optical applications.
How to Synthesize 3,8-Dipyrrole-BODIPY Efficiently
The implementation of this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process begins with the charging of the BOPHY solid powder into a Schlenk tube under an inert atmosphere, typically argon, to prevent oxidative degradation of the sensitive precursors. Following the establishment of an oxygen-free environment, a molar excess of pyrrole is introduced to drive the equilibrium towards the desired product. The reaction mixture is then heated to an optimal temperature window of 135-145°C for a duration of 4 to 6 hours. Post-reaction workup involves standard extraction techniques using dichloromethane, followed by drying and solvent removal. The final purification is achieved through silica gel column chromatography, utilizing a specific gradient of ethyl acetate and petroleum ether to isolate the target dye with high homogeneity.
- Charge the reactor with BOPHY compound (Formula II) under inert gas protection.
- Add excess pyrrole and heat the mixture to 135-145°C for 4-6 hours.
- Purify the crude product via dichloromethane extraction and silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical specifications. The elimination of expensive transition metal catalysts represents a direct reduction in raw material costs, removing the volatility associated with precious metal markets. Furthermore, the simplification of the synthetic route from multiple steps to a single pot operation drastically reduces labor hours, energy consumption, and solvent usage. These efficiencies compound to lower the overall cost of goods sold (COGS), allowing for more competitive pricing in the global market for specialty chemicals and electronic materials. The robustness of the process also minimizes the risk of batch failures, ensuring a more predictable and reliable supply schedule for downstream customers.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and harsh oxidants like DDQ significantly lowers the input cost per kilogram of product. By avoiding the need for specialized metal scavenging resins and complex waste treatment protocols associated with heavy metals, the downstream processing costs are substantially reduced. This streamlined approach allows for a more lean manufacturing model, where resources are allocated more efficiently, ultimately driving down the unit price of the final fluorescent dye without compromising on quality or performance metrics.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as pyrrole and the BOPHY precursor mitigates the risk of supply disruptions often caused by niche reagent shortages. The simplified one-pot reaction reduces the number of intermediate handling steps, which in turn decreases the potential for logistical delays and inventory bottlenecks. This agility enables manufacturers to respond more rapidly to fluctuations in market demand, ensuring that clients in the pharmaceutical and electronics sectors receive their orders on time, every time, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with modern environmental regulations and sustainability goals. The absence of heavy metal waste simplifies effluent treatment and reduces the environmental footprint of the manufacturing facility. This compliance advantage facilitates easier permitting for scale-up activities, allowing production to move seamlessly from pilot plant quantities to multi-ton commercial scales. The inherent safety of operating without pyrophoric catalysts or highly toxic oxidants also improves workplace safety standards, reducing liability and insurance costs for the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,8-dipyrrole-BODIPY fluorescent dyes. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this material into their existing product lines or research pipelines.
Q: What is the primary advantage of this BODIPY synthesis method over prior art?
A: The primary advantage is the significant increase in yield, reaching up to 19% compared to the historical 5% benchmark, achieved through a simplified one-pot oxidative nucleophilic reaction that eliminates unstable intermediates.
Q: Does this process require expensive transition metal catalysts?
A: No, the patented method operates entirely without transition metal catalysts, which drastically reduces raw material costs and simplifies the downstream purification process by removing the need for heavy metal scavenging.
Q: How does the structural modification affect the dye's performance?
A: The introduction of conjugated pyrrole groups at the 3 and 8 positions effectively redshifts the fluorescence emission wavelength, making the dye suitable for near-infrared applications and enhancing its utility in biological imaging and sensing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,8-Dipyrrole-BODIPY Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis route described in patent CN111187288B for the next generation of optical materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,8-dipyrrole-BODIPY delivered meets the highest industry standards for fluorescence quantum yield and photostability.
We invite you to collaborate with our technical team to explore how this advanced synthesis can optimize your supply chain and reduce your overall manufacturing costs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
