Advanced Synthesis of Long-Wavelength Boron Tripyrrole Fluorescent Dyes for Commercial Scale-Up
The landscape of functional fluorescent materials is undergoing a significant transformation driven by the demand for deeper tissue penetration and higher sensitivity in bio-imaging applications. Patent CN102516799A introduces a groundbreaking class of novel long-wavelength boron tripyrrole fluorescent dyes that address critical limitations found in conventional BODIPY (boron-dipyrromethene) architectures. By strategically introducing pyrrolyl groups at the 3 and 5 positions of the parent structure and incorporating diverse substituents at the 8-position, this technology achieves a substantial expansion of the conjugated system. This structural innovation results in optimized spectral properties, including significantly red-shifted absorption and emission wavelengths, enhanced molar extinction coefficients, and improved fluorescence quantum yields. For R&D directors and procurement specialists seeking reliable functional fluorescent dye intermediates supplier partnerships, this patent represents a pivotal shift towards more efficient, scalable, and high-performance optical materials suitable for analytical chemistry, life sciences, and environmental monitoring.
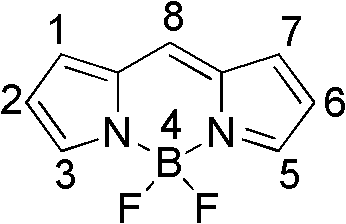
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of extended conjugation BODIPY derivatives has been plagued by significant technical and economic hurdles that hinder large-scale commercialization. Traditional strategies often relied on the introduction of bulky aromatic rings at the core structure, which inadvertently induced severe steric repulsion between molecular atoms. This repulsion disrupts the planarity of the conjugated system, leading to suboptimal spectral characteristics where the wavelength red-shift is limited compared to standard dyes. Furthermore, prior art methods for synthesizing fluoroboron tripyrrole derivatives, such as those reported by Kraut et al., necessitated the use of expensive noble metal catalysts like palladium. These processes typically involved lengthy multi-step synthetic routes with harsh reaction conditions, resulting in low overall yields and complex purification protocols. The reliance on transition metals also introduces stringent regulatory burdens regarding residual metal limits in pharmaceutical and biological applications, thereby increasing the cost reduction in electronic chemical manufacturing and complicating the supply chain for high-purity materials.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by enabling the direct construction of the tripyrrole framework through streamlined one-pot or solvent-free techniques. This approach eliminates the need for precious metal catalysts entirely, utilizing readily available acid chlorides and pyrrole as starting materials in a highly atom-economical process. The reaction conditions are markedly milder, often proceeding at temperatures between 40°C and 60°C in common organic solvents, or even under solvent-free conditions at elevated temperatures with oxygen oxidation. This simplification not only drastically reduces the operational complexity but also enhances the safety profile of the manufacturing process. By avoiding the use of palladium and minimizing solvent consumption, the novel route provides a robust pathway for the commercial scale-up of complex polymer additives and optical intermediates, ensuring consistent quality and supply continuity for downstream applications in cell imaging and biosensing.
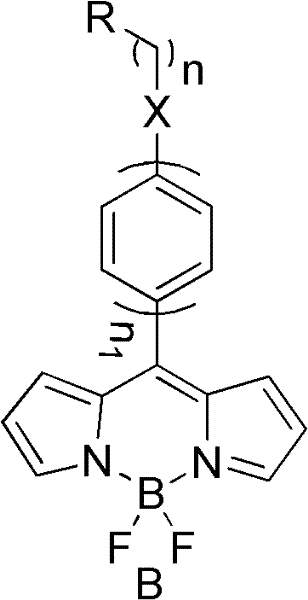
Mechanistic Insights into One-Pot Cyclization and Conjugation Extension
The core mechanistic advantage of this technology lies in the efficient formation of the tripyrrole methine bridge which extends the pi-conjugation system across the entire molecular backbone. In the one-pot synthesis, the initial condensation of acid chloride with excess pyrrole generates an uncoordinated dipyrrole intermediate. Subsequent complexation with boron trifluoride etherate in the presence of a base like triethylamine facilitates the cyclization and coordination of the boron atom, locking the structure into a rigid, planar conformation. This rigidity is crucial for minimizing non-radiative decay pathways, thereby maximizing the fluorescence quantum yield which can reach values as high as 0.72 in polar solvents like methanol. The introduction of the third pyrrole ring at the meso-position or via substitution effectively lowers the energy gap between the HOMO and LUMO orbitals, resulting in the observed bathochromic shift in both absorption and emission spectra. This precise control over electronic properties allows for the fine-tuning of the dye's color output, making it highly versatile for multiplexed detection assays.
Furthermore, the patent details a unique microwave-assisted deboronation strategy that converts the stable fluoroboron tripyrrole dyes into free tripyrrole derivatives. This transformation involves treating the dye with a strong base such as potassium tert-butoxide in tert-butanol under microwave irradiation at 92°C. The mechanism likely involves the nucleophilic attack on the boron center followed by the elimination of the BF2 group, yielding a tripyrrole scaffold that shares structural homology with prodigiosin analogs known for their anticancer activity. This dual utility—serving as both a high-performance fluorophore and a precursor for bioactive natural product analogs—adds significant value to the synthetic platform. The ability to access these distinct chemical spaces from a common intermediate underscores the flexibility of the chemistry and opens new avenues for drug discovery and development pipelines.
How to Synthesize Boron Tripyrrole Dye Efficiently
The synthesis of these advanced optical materials is designed for reproducibility and scalability, leveraging standard laboratory equipment that can be easily adapted for pilot plant operations. The process begins with the careful control of stoichiometry, typically employing a ten-fold excess of pyrrole relative to the acid chloride to drive the condensation reaction to completion. Following the formation of the dipyrrole, the addition of the boron source must be rapid to ensure efficient complexation before side reactions can occur. The workup procedure involves standard aqueous extraction and drying, followed by purification via silica gel chromatography using hexane and ethyl acetate gradients. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Mix substituted acid chloride and pyrrole in a 1: 10 molar ratio in an organic solvent like dichloromethane under oxygen atmosphere at 0-5°C.
- Stir at room temperature for 6-12 hours to form uncoordinated conjugated dipyrrole, then add triethylamine and boron trifluoride etherate.
- Raise temperature to 40-60°C for 12-24 hours, then purify the crude product via silica gel column chromatography to obtain the final dye.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthetic route offers profound benefits that extend beyond mere technical performance. The elimination of noble metal catalysts represents a direct removal of a major cost driver and a significant supply chain risk factor, as palladium prices are notoriously volatile and subject to geopolitical constraints. By shifting to a base-metal-free or metal-free organic synthesis, manufacturers can achieve substantial cost savings in raw material procurement and reduce the capital expenditure associated with metal scavenging and waste treatment infrastructure. This simplification of the bill of materials enhances the predictability of production costs, allowing for more stable pricing models for long-term contracts.
- Cost Reduction in Manufacturing: The one-pot nature of the synthesis consolidates multiple reaction steps into a single vessel operation, which significantly reduces labor hours, energy consumption, and solvent usage. This intensification of the process leads to a smaller physical footprint for production facilities and lowers the overall cost of goods sold. Additionally, the avoidance of expensive chromatography media in favor of simpler crystallization or extraction techniques for certain derivatives further drives down operational expenses, making the final dye products more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: The starting materials, primarily substituted acid chlorides and pyrrole, are commodity chemicals available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source specialty reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and atmospheric conditions, minimizes the risk of batch failures due to minor process deviations. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream customers in the diagnostics and imaging sectors.
- Scalability and Environmental Compliance: The solvent-free variant of this synthesis offers an exceptional opportunity for green chemistry implementation, virtually eliminating volatile organic compound (VOC) emissions associated with large-scale solvent handling and recovery. This aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. The high thermal stability of the intermediates and the final products ensures safe handling during transport and storage, reducing the logistical complexities and insurance costs associated with hazardous chemical shipments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this fluorescent dye technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear understanding of the material's capabilities and limitations.
Q: What are the advantages of this new boron tripyrrole synthesis over traditional BODIPY methods?
A: Unlike traditional methods requiring noble metal catalysts like palladium, this novel approach utilizes a one-pot or solvent-free strategy that significantly reduces raw material costs and eliminates expensive metal removal steps, while achieving superior red-shifted emission wavelengths.
Q: How does the spectral performance of these dyes compare to standard fluorophores?
A: These dyes exhibit optimized spectral properties including larger maximum absorption and emission wavelengths, significant Stokes shift values ranging from 613 to 1057 cm⁻¹, and high fluorescence quantum yields, making them ideal for deep-tissue imaging and sensing applications.
Q: Can these intermediates be further modified for specific pharmaceutical applications?
A: Yes, the structure allows for extensive modification at the 8-position with alkyl, halogen, or aromatic groups, and the tripyrrole derivatives can be converted into prodigiosin analogs with potential anticancer activity via microwave-assisted deboronation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boron Tripyrrole Dye Supplier
As the demand for high-performance optical materials continues to surge across the biotechnology and electronics sectors, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market deployment is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of boron tripyrrole dye meets the exacting standards required for sensitive analytical and biological applications.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this novel route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your R&D and supply chain teams to make informed decisions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
