Revolutionizing Triazole Intermediate Production via Green Electrochemical Cyclization Technology
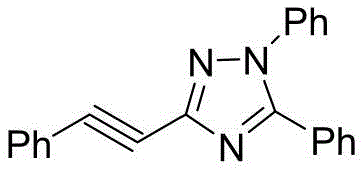
The pharmaceutical and fine chemical industries are constantly seeking sustainable alternatives to traditional cross-coupling reactions, particularly for the synthesis of nitrogen-rich heterocycles which are ubiquitous in drug discovery. Patent CN116178287A introduces a groundbreaking electrochemical methodology for the preparation of 3-phenylethynyl-1,2,4-triazole derivatives, a class of compounds known for their significant biological activities including anticancer and anti-inflammatory properties. This technology represents a paradigm shift from conventional thermal catalysis to electro-organic synthesis, utilizing electricity as the primary driving force to construct the triazole ring system under exceptionally mild conditions. By replacing stoichiometric oxidants and precious metal catalysts with electrons, this process not only aligns with the principles of green chemistry but also offers a robust pathway for generating diverse chemical libraries essential for modern medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-arylethynyl-1,2,4-triazole scaffolds has relied heavily on transition-metal catalyzed cross-coupling reactions, most notably the Sonogashira coupling. While effective, these traditional protocols suffer from inherent drawbacks that pose significant challenges for large-scale manufacturing and environmental compliance. The reliance on palladium and copper catalysts introduces the risk of heavy metal contamination in the final active pharmaceutical ingredients, necessitating complex and costly purification steps to meet stringent regulatory limits. Furthermore, these reactions often require elevated temperatures, inert gas protection, and the use of toxic organic solvents, which collectively increase the operational expenditure and carbon footprint of the synthesis. The need for pre-functionalized substrates, such as halogenated triazoles, adds additional synthetic steps, reducing the overall atom economy and extending the production lead time for critical intermediates.
The Novel Approach
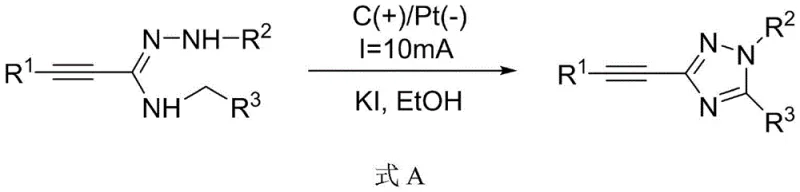
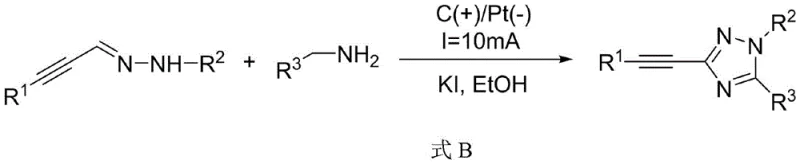
In stark contrast, the electrochemical strategy disclosed in the patent data offers a streamlined and environmentally benign alternative that circumvents the limitations of thermal catalysis. This novel approach enables the direct oxidative cyclization of readily available amidrazones or the condensation of hydrazones with benzylamines in a single electrochemical cell. The reaction proceeds at room temperature using simple halide salts like potassium iodide as mediators, eliminating the need for expensive transition metals entirely. By leveraging the anodic oxidation potential to generate reactive radical intermediates in situ, the process achieves high efficiency with yields consistently reported between 80% and 87% across a wide range of substrates. This method drastically simplifies the workflow, reduces waste generation, and provides a safer operating environment, making it an ideal candidate for the sustainable manufacturing of high-value pharmaceutical intermediates.
Mechanistic Insights into Electro-Oxidative Cyclization
The core of this innovative synthesis lies in the precise control of electrochemical potentials to drive the formation of the triazole ring without external chemical oxidants. In the presence of a halide catalyst such as KI, the anodic oxidation generates reactive iodine species which act as electron transfer mediators. These mediators facilitate the oxidation of the nitrogen-containing precursors, likely forming nitrogen-centered radicals or cationic intermediates that undergo intramolecular cyclization with the alkyne moiety. The use of a graphite anode and a platinum cathode ensures stable electron transfer while minimizing electrode degradation. This mechanism allows for the tolerance of various functional groups on the aromatic rings, including electron-donating and electron-withdrawing substituents, thereby expanding the scope of accessible chemical space for drug development without compromising reaction efficiency.
From an impurity control perspective, the absence of transition metals is a decisive advantage for pharmaceutical applications. Traditional palladium-catalyzed routes often leave trace amounts of metal residues that are difficult to remove and can catalyze unwanted degradation pathways in the final drug product. The electrochemical method described here produces a cleaner reaction profile, where the primary byproducts are typically hydrogen gas at the cathode and regenerated halide ions. This intrinsic cleanliness simplifies the downstream processing, as there is no need for specialized metal scavengers or extensive washing protocols. Consequently, the resulting 3-phenylethynyl-1,2,4-triazole derivatives exhibit high chemical purity, which is critical for ensuring the safety and efficacy of the final therapeutic agents derived from these intermediates.
How to Synthesize 3-Phenylethynyl-1,2,4-Triazole Derivatives Efficiently
The practical implementation of this electrochemical protocol is designed for ease of operation and reproducibility, making it accessible for both laboratory research and pilot-scale production. The process begins with the preparation of an electrolyte solution containing the substrate, a supporting electrolyte like KI, and a protic solvent such as ethanol. A constant current is then applied across the electrodes, driving the reaction to completion within a few hours at ambient temperature. This straightforward procedure eliminates the complexities associated with handling air-sensitive catalysts or maintaining high-pressure reactors. For detailed operational parameters and specific substrate examples, please refer to the standardized synthesis guide provided below.
- Prepare the electrolyte solution by dissolving the amidrazone substrate (or hydrazone and benzylamine precursors) along with a halide catalyst such as KI in an alcoholic solvent like ethanol.
- Insert a graphite anode and a platinum cathode into the reactor and apply a constant current of 8mA to 15mA at room temperature for 2 to 5 hours to drive the oxidative cyclization.
- Upon completion, concentrate the reaction mixture and purify the crude product using silica gel column chromatography to isolate the high-purity 3-phenylethynyl-1,2,4-triazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology translates into tangible strategic benefits that extend beyond mere technical novelty. The elimination of precious metal catalysts directly addresses one of the most volatile cost drivers in fine chemical manufacturing, as the prices of palladium and copper can fluctuate significantly based on global market dynamics. By substituting these expensive metals with abundant and inexpensive halide salts, the raw material cost structure is fundamentally optimized, leading to substantial cost savings in pharmaceutical intermediate manufacturing. Furthermore, the simplified process flow reduces the consumption of auxiliary materials and energy, contributing to a more lean and efficient production model that enhances overall margin potential.
- Cost Reduction in Manufacturing: The removal of palladium and copper catalysts from the synthesis route eliminates the need for costly metal recovery systems and specialized waste treatment protocols required for heavy metal disposal. This qualitative shift in the cost base allows for a more predictable pricing model for the final intermediates, shielding the supply chain from the volatility of precious metal markets. Additionally, the use of common solvents like ethanol instead of specialized anhydrous or toxic solvents further reduces procurement costs and simplifies inventory management for the manufacturing facility.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as graphite, platinum, and potassium iodide ensures a robust and resilient supply chain that is less susceptible to geopolitical disruptions or shortages of specialized reagents. Since the reaction operates at room temperature and atmospheric pressure, the equipment requirements are less stringent than those for high-pressure hydrogenation or high-temperature coupling reactions. This flexibility allows for faster deployment of production capacity and reduces the lead time for high-purity pharmaceutical intermediates, enabling quicker response to market demands.
- Scalability and Environmental Compliance: The electrochemical nature of this process is inherently scalable, particularly when adapted to continuous flow reactors which offer superior heat and mass transfer characteristics. This scalability supports the commercial scale-up of complex pharmaceutical intermediates from gram to ton quantities without the need for significant process re-engineering. Moreover, the green profile of the synthesis, characterized by the use of electrons as reagents and the generation of minimal waste, facilitates easier compliance with increasingly stringent environmental regulations, reducing the regulatory burden and associated compliance costs for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 3-phenylethynyl-1,2,4-triazole derivatives. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of this electrochemical method over traditional Sonogashira coupling?
A: Unlike traditional methods that require expensive palladium and copper catalysts, high temperatures, and inert atmospheres, this electrochemical approach operates at room temperature using electrons as the cleanest oxidant. This eliminates heavy metal contamination risks and significantly reduces raw material costs associated with precious metal catalysts.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process utilizes simple undivided cells with graphite and platinum electrodes and operates under mild conditions without strict pressure requirements. The use of common solvents like ethanol and stable halide salts makes the transition from laboratory scale to continuous flow electrochemical reactors highly feasible for large-scale manufacturing.
Q: What is the typical purity and yield profile for these triazole derivatives?
A: The patent data demonstrates consistent yields ranging from approximately 80% to 87% across various substituted derivatives. The absence of transition metal catalysts simplifies the purification process, allowing for the achievement of high-purity specifications suitable for pharmaceutical applications through standard silica gel chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Phenylethynyl-1,2,4-Triazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of critical pharmaceutical building blocks. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN116178287A can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-phenylethynyl-1,2,4-triazole derivatives delivered meets the highest quality standards required by global regulatory agencies.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to leverage this green technology for their drug development pipelines. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis to evaluate how switching to this electrochemical route can optimize your specific supply chain. We encourage you to contact us today to obtain specific COA data for our existing library of triazole intermediates and to discuss route feasibility assessments for your custom synthesis projects, ensuring a secure and sustainable supply of high-quality materials for your future success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
