Advanced Hydrogenation Strategy for High-Purity Dibenzyl Ethylenediamine Acetate Manufacturing
The pharmaceutical industry's relentless demand for high-purity antibiotic precursors has driven significant innovation in the synthesis of key intermediates like dibenzyl ethylenediamine and its acetate salt. Patent CN101774927A introduces a transformative preparation method that addresses the longstanding inefficiencies of traditional alkylation routes. This technology leverages a sophisticated two-step sequence involving the formation of a Schiff base followed by catalytic hydrogenation, offering a robust alternative to corrosive and low-yielding legacy processes. The resulting dibenzyl ethylenediamine acetate is a critical building block for the manufacture of long-acting antibiotics such as benzathine penicillin V, benzathine penicillin G, and various cephalosporins. By shifting the synthetic paradigm from nucleophilic substitution to reductive amination, this patent outlines a pathway that not only enhances product quality but also aligns with modern green chemistry principles essential for sustainable pharmaceutical intermediates manufacturing.
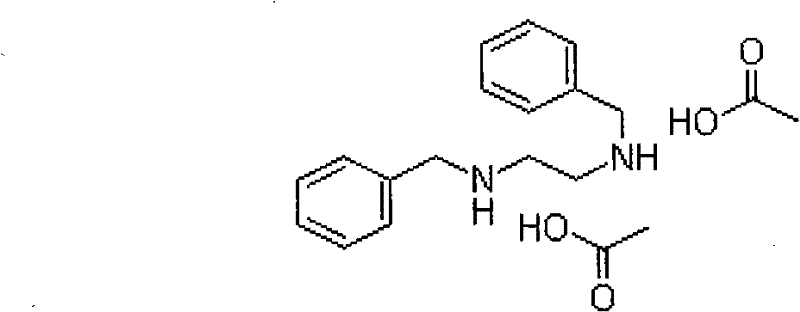
For R&D directors evaluating process feasibility, understanding the structural integrity and purity profile of the target molecule is paramount. The molecular architecture of dibenzyl ethylenediamine, as depicted in the structural analysis, requires precise control over the N-alkylation to prevent the formation of mono-substituted or tri-substituted impurities which can complicate downstream salt formation. The patent specifies that the new method achieves a product content of greater than 99%, with stringent controls on heavy metals (≤10ppm) and sulfated ash (≤0.1%). This level of purity is critical for API synthesis where impurity carryover can affect the stability and efficacy of the final drug product. The ability to consistently produce material that meets the Q-CCYH02-2000 standard demonstrates the robustness of the catalytic system employed, providing a reliable foundation for developing stable and effective long-acting antibiotic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of dibenzyl ethylenediamine has relied on three primary synthetic routes, each plagued by significant operational and economic drawbacks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. The first conventional method utilizes benzaldehyde but suffers from poor condensation yields ranging between 60% and 70%, coupled with substantial wastewater discharge that complicates environmental compliance. The second route, starting from benzylamine, is characterized by excessively long reaction times and difficult purification steps, with yields stagnating between 50% and 60%. Perhaps most problematic is the third route employing benzyl chloride, which generates large volumes of corrosive hydrochloric acid as a byproduct, necessitating expensive corrosion-resistant equipment and complex neutralization procedures. Furthermore, this chlorination route typically achieves yields of only around 75% and requires a large excess of ethylenediamine, driving up raw material costs and creating significant separation challenges during the distillation purification step.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach detailed in the patent utilizes a reductive amination strategy that fundamentally simplifies the process flow and enhances overall efficiency. By reacting benzaldehyde with ethylenediamine in an alcoholic solvent to form a Schiff base, followed by catalytic hydrogenation, the process achieves transformation efficiencies exceeding 95% and product purities greater than 99%. This method eliminates the need for harsh alkylating agents like benzyl chloride, thereby removing the generation of corrosive acidic waste and reducing the burden on waste treatment facilities. The reaction conditions are remarkably mild, operating at temperatures between 30°C and 70°C and low hydrogen pressures of 5-10 kPa, which significantly lowers the safety risks associated with high-pressure hydrogenation units. The streamlined workflow allows for direct salification of the crude alcoholic solution with glacial acetic acid, bypassing the energy-intensive distillation steps required in older methods to isolate the free base.
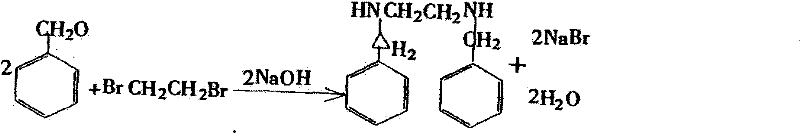
Mechanistic Insights into Catalytic Hydrogenation and Reductive Amination
The core of this technological advancement lies in the precise execution of the reductive amination mechanism, which proceeds through a well-defined Schiff base intermediate. In the first stage, benzaldehyde and ethylenediamine undergo a dehydration condensation reaction in an alcoholic medium, typically ethanol or methanol, at controlled temperatures of 30-60°C. This step is critical for establishing the carbon-nitrogen double bond characteristic of the imine functionality, which serves as the electrophilic center for the subsequent reduction. The choice of solvent plays a pivotal role here, as alcohols not only facilitate the dissolution of reactants but also participate in the equilibrium dynamics of water removal, driving the reaction towards the completion of the Schiff base formation. The patent highlights that maintaining the temperature within this specific range prevents polymerization side reactions while ensuring rapid kinetics, setting the stage for a highly efficient reduction phase.
Following the formation of the Schiff base, the mixture is subjected to catalytic hydrogenation using either palladium on carbon (Pd/C) or Raney nickel as the heterogeneous catalyst. This reduction step converts the unstable imine bond into a stable secondary amine linkage with exceptional selectivity. The patent specifies catalyst loading ratios, such as 100:1 for Pd/C or 100:3 for Raney nickel, optimized to maximize turnover frequency while minimizing metal residue in the final product. Operating at a low hydrogen pressure of 5-10 kPa is a key safety and economic feature, as it reduces the requirement for specialized high-pressure reactors and lowers the risk of over-reduction or hydrogenolysis of the benzyl groups. The resulting dibenzyl ethylenediamine remains in solution, allowing for a seamless transition to the final salification step without the need for intermediate isolation, thereby preserving the high conversion rate of 99% observed in the reduction phase.
How to Synthesize Dibenzyl Ethylenediamine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of solvent selection and temperature control to achieve optimal results. The process begins with the dissolution of benzaldehyde in ethanol, followed by the dropwise addition of ethylenediamine while maintaining the temperature between 40°C and 60°C to ensure complete conversion to the Schiff base. Once the intermediate is formed, the solution is transferred to a hydrogenation vessel where the catalyst is introduced, and hydrogen gas is supplied at low pressure until uptake ceases, indicating reaction completion. For those seeking to implement this technology, the detailed standardized synthesis steps see the guide below, which breaks down the specific molar ratios, agitation speeds, and filtration techniques required to replicate the high yields and purity reported in the intellectual property documentation.
- Condense benzaldehyde with ethylenediamine in an alcoholic solvent at 30-60°C to form the Schiff base intermediate.
- Perform catalytic hydrogenation of the Schiff base using Pd/C or Raney Nickel at low pressure (5-10 kPa) to obtain the free amine.
- Directly add glacial acetic acid to the alcoholic solution of the amine to precipitate the high-purity acetate salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers compelling advantages that directly address the pain points of cost volatility and supply continuity in the pharmaceutical intermediates sector. The elimination of benzyl chloride as a starting material removes a significant hazard from the supply chain, as benzyl chloride is a regulated substance in many jurisdictions due to its toxicity and potential use as a chemical warfare precursor. By switching to benzaldehyde and ethylenediamine, both of which are commodity chemicals with stable global supply chains, manufacturers can mitigate the risk of regulatory shutdowns and ensure a consistent flow of raw materials. Furthermore, the patent explicitly claims a cost reduction of more than 20% compared to traditional methods, driven primarily by the higher yields and the elimination of expensive purification steps like fractional distillation. This economic efficiency translates into a more competitive pricing structure for the final acetate salt, providing a buffer against market fluctuations in raw material costs.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from multiple factors, including the high atom economy of the reductive amination reaction and the recyclability of the alcoholic solvents. Unlike the benzyl chloride route which consumes large excesses of ethylenediamine to drive the reaction and requires energy-intensive distillation to recover it, the new method operates with near-stoichiometric ratios and allows the solvent to be recovered and reused after simple filtration and crystallization. The avoidance of corrosive byproducts also extends the lifespan of reactor vessels and piping, reducing capital expenditure on maintenance and equipment replacement. Additionally, the direct salification step means that the energy cost associated with drying and isolating the free base is completely eliminated, further compressing the overall manufacturing budget and enhancing the margin profile for bulk producers.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions contributes significantly to supply chain resilience, as the process does not rely on specialized high-pressure infrastructure or exotic catalysts that might face supply bottlenecks. The use of common catalysts like Pd/C or Raney nickel ensures that sourcing remains straightforward, while the mild operating parameters allow for production in a wider range of multipurpose chemical facilities. This flexibility reduces the lead time for scaling up production capacity in response to surges in demand for downstream antibiotics like penicillin. Moreover, the high purity of the crude product reduces the likelihood of batch failures during quality control testing, ensuring that shipment schedules are met consistently without the delays associated with re-processing off-spec material.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial tonnage is facilitated by the inherent safety of the low-pressure hydrogenation step, which minimizes the engineering controls required for explosion protection. The environmental profile is vastly superior to legacy methods, as the process generates minimal aqueous waste and avoids the discharge of chloride ions that contribute to soil and water salinity. The recyclability of the ethanol or methanol solvent aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and solvent waste disposal. This green chemistry advantage not only simplifies the permitting process for new manufacturing sites but also enhances the corporate sustainability credentials of the supplier, a factor that is becoming increasingly important for multinational pharmaceutical buyers auditing their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, drawing directly from the experimental data and beneficial effects described in the patent literature. These insights are designed to clarify the operational parameters and quality expectations for stakeholders considering the adoption of this route for commercial production. Understanding these details is crucial for assessing the feasibility of integrating this process into existing manufacturing workflows and for ensuring that the final product meets the rigorous specifications required for pharmaceutical applications.
Q: What are the critical impurities controlled in this novel synthesis route?
A: The novel catalytic hydrogenation route significantly minimizes the formation of mono-benzylated byproducts and polymeric impurities often associated with the traditional benzyl chloride alkylation method, ensuring single benzyl impurities remain below 1.5%.
Q: How does the new process improve environmental compliance compared to older methods?
A: By eliminating the use of benzyl chloride, the process avoids the generation of large quantities of corrosive hydrochloric acid wastewater, and the alcoholic solvents used are fully recyclable, drastically reducing the environmental footprint.
Q: Is this method suitable for large-scale commercial production of antibiotic intermediates?
A: Yes, the process operates at mild temperatures (30-70°C) and very low hydrogen pressures (5-10 kPa), making it inherently safer and easier to scale up for multi-ton production of benzathine penicillin precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzyl Ethylenediamine Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering dibenzyl ethylenediamine acetate that adheres to stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the necessary standards for heavy metals, moisture content, and assay potency. Our facility is equipped to handle the specific requirements of catalytic hydrogenation processes, providing a safe and compliant environment for the production of this sensitive pharmaceutical intermediate.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By leveraging our expertise in process optimization and supply chain management, we can help you realize the economic and operational benefits of this advanced synthesis route. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term strategic goals in the pharmaceutical sector.
