Advanced Synthesis of N,N-Dibenzyl-Ethylenediamin Diacetate for Commercial API Production
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for critical antibiotic intermediates. Patent CN101747206A introduces a transformative preparation method for N,N-dibenzyl-ethylenediamin diacetate (DBED), a pivotal building block in the synthesis of long-acting penicillins and cephalosporins. This technology shifts the paradigm from traditional, pollution-heavy alkylation methods to a cleaner, high-yield reductive amination strategy. By leveraging a three-step sequence involving condensation, catalytic hydrogenation, and salification, the process achieves yields exceeding 93% while maintaining exceptional control over impurity profiles. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for essential beta-lactam antibiotics, ensuring both regulatory compliance and cost-effectiveness in large-scale manufacturing environments.
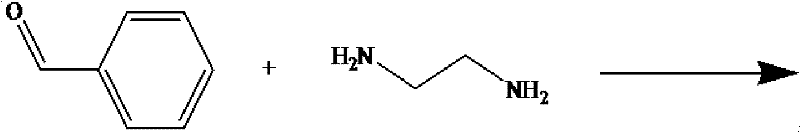
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N,N-dibenzyl-ethylenediamin diacetate relied heavily on the direct alkylation of ethylenediamine with benzyl chloride in the presence of strong bases such as sodium hydroxide. This legacy approach suffers from inherent thermodynamic and kinetic limitations that severely impact commercial viability. The reaction typically generates a complex mixture of mono-, di-, and tri-alkylated byproducts, leading to a notoriously difficult purification process and low overall yields. Furthermore, the stoichiometric requirement for strong bases results in the generation of massive quantities of saline wastewater, posing significant environmental disposal challenges and increasing operational costs for waste treatment facilities. The use of benzyl chloride, a potent lachrymator and alkylating agent, also introduces severe safety hazards regarding worker exposure and equipment corrosion, making the process less desirable for modern, safety-conscious manufacturing sites.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a reductive amination pathway starting from benzaldehyde and ethylenediamine. This route fundamentally alters the reaction landscape by first forming a stable Schiff base intermediate, N,N'-dibenzylidene ethylenediamine, which is subsequently reduced to the diamine. This strategic shift eliminates the need for hazardous alkyl halides and corrosive bases entirely. The process operates under milder conditions with readily available raw materials, significantly simplifying the reaction engineering requirements. By avoiding the statistical distribution of alkylation products inherent in the old method, this new approach ensures a much cleaner reaction profile, directly translating to higher purity crude products and reducing the burden on downstream purification units. This technological leap not only enhances safety but also aligns perfectly with green chemistry principles, offering a sustainable alternative for the production of high-value pharmaceutical intermediates.
Mechanistic Insights into Reductive Amination and Catalytic Hydrogenation
The core of this synthetic innovation lies in the precise control of the condensation and hydrogenation steps. Initially, benzaldehyde reacts with ethylenediamine in an organic solvent such as ethyl acetate or ethanol at temperatures ranging from 60°C to 120°C. This condensation proceeds via a nucleophilic attack of the amine nitrogen on the carbonyl carbon, followed by dehydration to form the bis-imine (Schiff base) structure. The choice of solvent is critical here; solvents like ethyl acetate facilitate the removal of water formed during the reaction, driving the equilibrium towards the imine product without requiring complex azeotropic distillation setups. This step is highly exothermic and requires careful temperature management to prevent polymerization or side reactions, ensuring the formation of a high-quality intermediate ready for reduction.
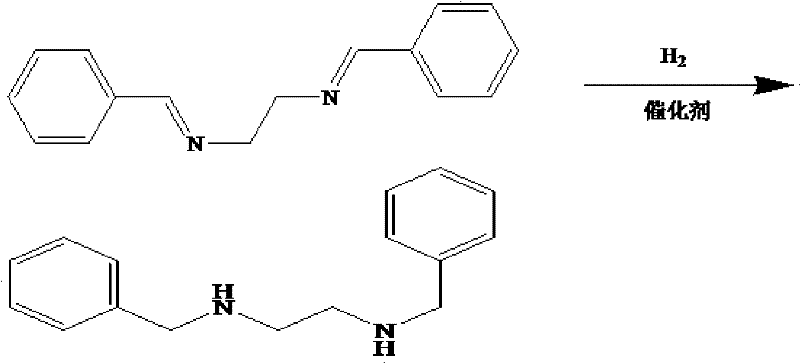
Following the formation of the imine, the process employs catalytic hydrogenation to reduce the C=N double bonds to C-N single bonds. This step is performed under hydrogen pressure ranging from 0.5 MPa to 10 MPa, typically using heterogeneous catalysts like Pd/C or Nickel at temperatures between 50°C and 150°C. The mechanism involves the adsorption of hydrogen and the imine substrate onto the catalyst surface, where the hydrogen atoms are transferred to the nitrogen-carbon bond. The patent highlights that catalyst loading can be kept remarkably low, between 0.1% and 1% of the substrate mass, yet still achieve complete conversion. This efficiency is crucial for cost reduction in API manufacturing, as it minimizes the consumption of precious metals and simplifies the filtration process to remove the spent catalyst. The final salification with glacial acetic acid is a straightforward acid-base reaction that precipitates the product as a stable crystalline salt, locking in the purity achieved during the previous steps.
How to Synthesize N,N-Dibenzyl-Ethylenediamin Diacetate Efficiently
Implementing this synthesis requires a systematic approach to reactor design and process control to maximize the benefits outlined in the patent. The procedure begins with the condensation of benzaldehyde and ethylenediamine, followed by a high-pressure hydrogenation step, and concludes with salt formation. Each stage demands specific attention to temperature, pressure, and stoichiometry to ensure the high yields reported in the embodiments. The following guide outlines the standardized operational parameters derived from the patent data, providing a clear roadmap for process engineers to replicate these results in a pilot or production setting. Detailed standard operating procedures for each unit operation are essential to maintain consistency and safety throughout the campaign.
- Condense benzaldehyde and ethylenediamine in an organic solvent at 60-120°C to form N,N'-dibenzylidene ethylenediamine.
- Perform catalytic hydrogenation on the imine intermediate using Pd/C or Ni catalyst at 50-150°C and 0.5-10 MPa pressure.
- React the resulting amine with glacial acetic acid in solvent at 60-90°C to crystallize the final diacetate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages beyond mere technical feasibility. The shift away from benzyl chloride mitigates significant supply chain risks associated with the handling and transportation of hazardous regulated chemicals. Additionally, the dramatic improvement in yield directly correlates to a reduction in raw material consumption per kilogram of finished product, effectively lowering the variable cost of goods sold. The simplified work-up procedure, which primarily involves filtration and drying rather than complex distillations or extractions, reduces energy consumption and cycle times, thereby enhancing overall plant throughput and asset utilization rates.
- Cost Reduction in Manufacturing: The elimination of sodium hydroxide and the associated waste treatment costs provides a substantial economic benefit. Traditional alkylation methods generate large volumes of saline effluent that require expensive neutralization and disposal; by replacing this with a catalytic hydrogenation process, the facility avoids these hidden environmental costs. Furthermore, the high selectivity of the reductive amination means less raw material is wasted on byproducts, optimizing the atom economy of the process. The ability to use lower catalyst loadings without sacrificing conversion rates further contributes to margin expansion, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Benzaldehyde and ethylenediamine are commodity chemicals with robust, multi-vendor supply chains, unlike specialized alkylating agents that may face availability fluctuations. This raw material security ensures continuous production capability even during market disruptions. The process tolerance for various solvents, including ethanol and ethyl acetate, provides additional flexibility for procurement teams to source materials based on regional availability and pricing dynamics. This adaptability is critical for maintaining uninterrupted supply to downstream antibiotic manufacturers who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard batch reactors and hydrogenation autoclaves found in most fine chemical facilities. The absence of corrosive reagents extends equipment lifespan and reduces maintenance downtime. From an environmental perspective, the cleaner reaction profile simplifies regulatory compliance, as the facility generates significantly less hazardous waste. This alignment with green chemistry standards not only reduces the risk of regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims within Patent CN101747206A, providing a reliable basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for upgrading existing production lines.
Q: What are the primary advantages of the reductive amination route over the traditional alkylation method?
A: The reductive amination route described in Patent CN101747206A eliminates the use of hazardous benzyl chloride and strong bases like sodium hydroxide. This results in significantly higher product yields (up to 95%), drastically reduced impurity profiles, and a much cleaner environmental footprint due to the absence of heavy salt waste streams.
Q: Which catalysts are suitable for the hydrogenation step in this synthesis?
A: The patent specifies that effective hydrogenation can be achieved using a variety of transition metal catalysts, including Nickel (Ni), Palladium on Carbon (Pd/C), Platinum (Pt), Rhodium (Rh), or Ruthenium (Ru). Preferred embodiments utilize Pd/C or Raney Nickel at loadings between 0.1% and 1% relative to the substrate mass.
Q: How does this process impact the scalability of antibiotic intermediate production?
A: The process utilizes standard unit operations such as condensation, high-pressure hydrogenation, and crystallization, which are easily scalable from laboratory to multi-ton production. The high yield and simple work-up procedures, involving basic filtration and drying, ensure robust supply chain continuity for downstream antibiotic manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Dibenzyl-Ethylenediamin Diacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering N,N-dibenzyl-ethylenediamin diacetate with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the demanding requirements of antibiotic synthesis. Our infrastructure is designed to support the complex catalytic hydrogenation and crystallization steps required by this advanced patent, guaranteeing consistent quality and supply continuity for our partners.
We invite you to collaborate with us to optimize your supply chain for beta-lactam antibiotic production. By leveraging our expertise in reductive amination technologies, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
