Scalable Synthesis of (S)-3-Hydroxypyrrolidine: A Strategic Upgrade for API Manufacturing
Introduction to Advanced Chiral Pyrrolidine Synthesis
The pharmaceutical industry's demand for high-purity chiral building blocks continues to drive innovation in process chemistry, particularly for urology therapeutics like Darifenacin. Patent CN101759619A introduces a robust and industrially viable preparation method for (S)-3-hydroxypyrrolidine and its N-derivatives, addressing critical bottlenecks in existing manufacturing protocols. This technology leverages the chiral pool strategy, utilizing L-Malic acid as a foundational starting material to construct the pyrrolidine core with exceptional stereochemical fidelity. By shifting away from hazardous reagents and multi-step protection-deprotection sequences, this methodology offers a streamlined pathway that aligns with modern green chemistry principles while maintaining rigorous quality standards. The strategic value of this route lies in its ability to deliver complex heterocyclic intermediates with reduced environmental impact and enhanced operational safety, making it an ideal candidate for commercial scale-up.
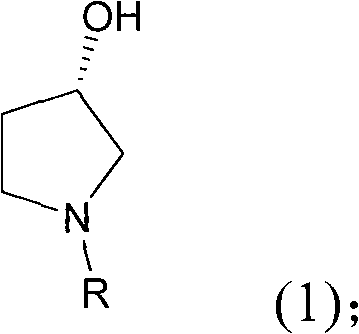
For R&D directors and procurement specialists, understanding the structural versatility of this intermediate is paramount. The process allows for the generation of various N-substituted derivatives where the R group can be hydrogen, methyl, ethyl, or benzyl, providing flexibility for downstream diversification. This adaptability ensures that the synthesis can be tailored to specific API requirements without necessitating a complete process redesign. Furthermore, the retention of the hydroxyl group at the 3-position with high optical purity is critical for the biological activity of the final drug substance. The patent data underscores a commitment to quality, reporting enantiomeric excess values exceeding 99%, which drastically reduces the burden on downstream purification processes. As a reliable pharmaceutical intermediate supplier, integrating such efficient routes into the supply chain is essential for maintaining competitiveness in the global API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-3-hydroxypyrrolidine has been plagued by inefficiencies that hinder large-scale production. Prior art, such as methods described in Tetrahedron Letters and Journal of Medicinal Chemistry, often relies on the conversion of L-Malic acid into intramolecular acid anhydrides using excess acetyl chloride. This approach is not only atom-uneconomical but also introduces significant safety hazards associated with handling large volumes of corrosive acyl chlorides. Additionally, the subsequent ring-closing steps with benzylamine often result in complex reaction mixtures that are difficult to purify, leading to suboptimal total recovery rates. Another prevalent conventional method utilizes Lithium Aluminum Hydride (LiAlH4) for the reduction of pyrrolidine precursors. While chemically effective on a small scale, LiAlH4 is notoriously dangerous for industrial application due to its pyrophoric nature and the generation of flammable hydrogen gas during quenching. These factors collectively increase the cost of goods sold (COGS) through expensive safety infrastructure, specialized waste treatment, and lower overall yields, creating a fragile supply chain vulnerable to disruptions.
The Novel Approach
In stark contrast, the methodology outlined in CN101759619A presents a paradigm shift towards safer and more efficient manufacturing. By bypassing the formation of unstable acid anhydrides and instead proceeding through a stable acid amide intermediate, the process simplifies the reaction profile and minimizes side-product formation. The transition to a dicarbonyl imide intermediate via thermal cyclization is a key innovation, creating a robust scaffold that is highly amenable to selective reduction. Crucially, the replacement of LiAlH4 with metal borohydrides (such as sodium borohydride) in the presence of acid promoters, or alternatively using Red-Al, mitigates the severe safety risks associated with traditional hydride reductions. This novel approach not only enhances operator safety but also facilitates easier temperature control and workup procedures. The result is a synthesis that is shorter, higher yielding, and fundamentally more compatible with the rigorous safety and environmental standards required for cost reduction in API manufacturing today.
Mechanistic Insights into L-Malic Acid Derived Cyclization
The core of this synthetic strategy relies on the precise manipulation of the L-Malic acid backbone to preserve chirality while constructing the nitrogen-containing ring. The initial amidation step involves the nucleophilic attack of an amine or ammonia on the carboxylic acid groups of L-Malic acid. This reaction, conducted in solvents like methanol or ethanol at temperatures ranging from 55°C to 150°C, forms an open-chain acid amide. The stoichiometry is carefully controlled, with a molar ratio of amine to L-Malic acid between 0.6:1 and 1.5:1, ensuring complete conversion while minimizing excess reagent waste. Following amidation, the system undergoes a thermal cyclization in high-boiling solvents such as toluene or xylene at 80°C to 250°C. This step drives the dehydration and ring closure to form the 3-hydroxy-N-substituted-2,5-dicarbonyl imide. The thermodynamic stability of this imide ring is crucial, as it locks the stereochemistry in place and prepares the molecule for the subsequent reduction.
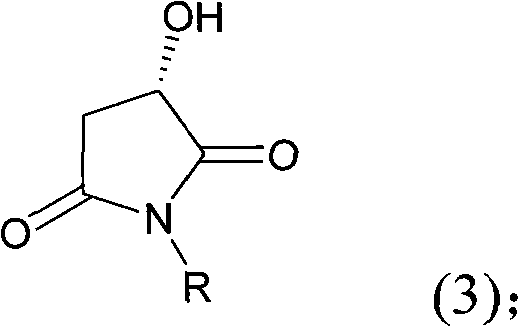
The reduction mechanism represents the most critical phase for impurity control and yield optimization. The dicarbonyl imide is subjected to reduction using metal borohydrides in the presence of Lewis acids like LiCl, AlCl3, or iodine, or alternatively using Red-Al in toluene. The presence of the acid promoter activates the carbonyl groups, facilitating hydride transfer while preventing over-reduction or racemization. Reaction temperatures are maintained between -30°C and 70°C to balance reaction kinetics with selectivity. For the N-benzyl protected intermediate, a final hydrogenation step using palladium on carbon or Raney nickel at 0.2 to 5 MPa pressure cleanly removes the benzyl group. This catalytic hydrogenation is highly selective, preserving the sensitive 3-hydroxyl group and the chiral center. The mechanistic elegance of this route ensures that impurities arising from epimerization or over-reduction are minimized, resulting in a product profile that meets the stringent purity specifications demanded by regulatory bodies for pharmaceutical intermediates.
How to Synthesize (S)-3-Hydroxypyrrolidine Efficiently
Implementing this synthesis requires a disciplined approach to reaction conditions and reagent selection to maximize the benefits of the patented route. The process begins with the straightforward amidation of L-Malic acid, followed by azeotropic water removal to drive the cyclization to the imide. The reduction step demands careful attention to temperature control and reagent addition rates to manage exotherms effectively. For the final deprotection, standard hydrogenation equipment capable of handling moderate pressures is sufficient. The detailed standardized synthesis steps, including specific solvent choices, temperature ramps, and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency route.
- React L-Malic acid with an amine or ammonia in a solvent like methanol or ethanol at 55-150°C to form the corresponding acid amide intermediate.
- Perform a thermal cyclization of the acid amide in a high-boiling solvent such as toluene at 80-250°C to generate the 3-hydroxy-N-substituted-2,5-dicarbonyl imide.
- Reduce the dicarbonyl imide using metal borohydrides with acid promoters or Red-Al at controlled temperatures (-30 to 70°C) to obtain the N-substituted pyrrolidine.
- Optionally, subject the N-benzyl intermediate to catalytic hydrogenation using Pd/C or Raney Nickel at 0.2-5 MPa to remove the protecting group and yield free (S)-3-hydroxypyrrolidine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this L-Malic acid-based route offers profound advantages for procurement managers and supply chain heads seeking to optimize their sourcing strategies. The primary driver of value is the utilization of L-Malic acid, a commodity chemical produced on a massive scale via fermentation, which ensures a stable and low-cost supply of the chiral starting material. Unlike synthetic chiral auxiliaries or expensive enzymatic resolutions, L-Malic acid provides inherent chirality at a fraction of the cost, directly translating to substantial cost savings in the final API production. Furthermore, the elimination of hazardous reagents like LiAlH4 and excess acetyl chloride reduces the need for specialized containment systems and expensive waste disposal protocols. This simplification of the safety infrastructure allows for production in standard multipurpose reactors, enhancing asset utilization and reducing capital expenditure requirements for new capacity.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from both material efficiency and operational simplicity. By shortening the synthetic route and avoiding complex protection-deprotection sequences, the overall consumption of solvents and reagents is significantly decreased. The use of safer reducing agents lowers the insurance and compliance costs associated with handling pyrophoric materials. Additionally, the high yields reported in the patent examples indicate a robust process with minimal material loss, further driving down the unit cost of the intermediate. These factors combine to create a highly competitive cost structure that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, non-proprietary starting materials. L-Malic acid and common amines are sourced from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent grades, ensures consistent production output even when minor variations in feedstock quality occur. This reliability is critical for maintaining continuous API supply, preventing costly production stoppages, and meeting Just-In-Time delivery commitments to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations such as distillation, crystallization, and filtration that are standard in fine chemical manufacturing. The avoidance of heavy metal catalysts in the main synthetic sequence (using boron or aluminum-based reagents instead) simplifies the removal of trace metals, a critical quality attribute for drug substances. Moreover, the reduced generation of hazardous waste aligns with increasingly strict environmental regulations, minimizing the ecological footprint of the manufacturing site. This environmental compatibility not only ensures regulatory compliance but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the technical disclosures in patent CN101759619A and provide clarity on the feasibility and advantages of the technology. Understanding these details is essential for technical teams evaluating the potential for technology transfer or contract manufacturing partnerships.
Q: Why is the L-Malic acid route superior to traditional acetyl chloride methods for (S)-3-hydroxypyrrolidine?
A: Traditional methods often require excess acetyl chloride to form anhydrides, leading to complex workups and lower total recovery. The L-Malic acid route described in CN101759619A utilizes a direct amidation and cyclization strategy that eliminates hazardous anhydride formation steps, resulting in a shorter synthetic sequence and significantly improved operational safety.
Q: How does this process ensure high enantiomeric purity for Darifenacin production?
A: By starting from L-Malic acid, a naturally occurring chiral pool material, the stereochemistry is inherently preserved throughout the synthesis. The patent data demonstrates an enantiomeric excess (ee) of 99.66% in the final hydrogenation step, ensuring the product meets the stringent purity specifications required for urology API intermediates without the need for costly chiral resolution.
Q: What are the safety advantages of using Red-Al or Borohydrides over Lithium Aluminum Hydride (LiAlH4)?
A: Prior art methods relying on LiAlH4 present significant industrial hazards due to its pyrophoric nature and difficult quenching requirements. This novel approach employs safer reducing agents like sodium borohydride with acid promoters or Red-Al (sodium bis(2-methoxyethoxy)aluminum hydride), which offer manageable reactivity profiles, easier temperature control, and reduced risk of thermal runaway during large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Hydroxypyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering (S)-3-hydroxypyrrolidine with stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest industry standards. Our expertise in chiral pool synthesis allows us to optimize this specific L-Malic acid route for maximum yield and cost-efficiency, providing you with a secure supply of this vital urology intermediate.
We invite you to collaborate with us to explore how this advanced synthesis can enhance your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
