Advanced Synthesis of Di(2-ethylhexyl) 2-ethylhexylphosphonate for Commercial Scale-up
The chemical industry is constantly evolving towards safer and more efficient synthetic pathways, particularly for high-volume intermediates used in hydrometallurgy and specialty applications. A pivotal advancement in this domain is detailed in patent CN115028660A, which discloses a novel method for synthesizing di(2-ethyl)hexyl 2-ethylhexylphosphonate, commonly known as the middle ester intermediate for the renowned extractant P-507. This technology represents a significant departure from conventional protocols by utilizing sodium 2-ethylhexanoxide as the base instead of the traditionally employed sodium ethoxide. For R&D directors and process engineers, this shift is not merely a reagent swap but a fundamental restructuring of the reaction thermodynamics and byproduct profile. The patent outlines a robust two-step sequence that enhances production efficiency while drastically mitigating the safety risks associated with volatile alcohol byproducts. By aligning the alkoxide base with the ester chain, the process achieves a level of atom economy and operational safety that addresses long-standing pain points in organophosphorus manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the domestic and international synthesis of di(2-ethyl)hexyl 2-ethylhexylphosphonate has relied heavily on sodium ethoxide as the alkaline catalyst. While chemically effective, this traditional approach introduces severe engineering and safety challenges that burden procurement and supply chain teams. The primary issue stems from the generation of ethanol as a stoichiometric byproduct during the salt formation step. Ethanol possesses a high vapor pressure, a low boiling point, and a critically low flash point, creating a hazardous environment that requires explosion-proof equipment and rigorous ventilation systems. Furthermore, the recovery of ethanol from the reaction mixture is energetically expensive and often inefficient, leading to significant solvent loss and increased waste treatment costs. The presence of ethanol also complicates the downstream alkylation step, as residual moisture or alcohol can interfere with the Michaelis-Becker reaction, potentially lowering yields and increasing the complexity of the impurity profile. These factors collectively inflate the cost of goods sold (COGS) and introduce volatility into the supply chain due to regulatory pressures on volatile organic compound (VOC) emissions.
The Novel Approach
The innovative methodology presented in the patent data circumvents these issues by employing sodium 2-ethylhexanoxide, a base that matches the carbon skeleton of the target molecule. This strategic alignment ensures that the byproduct of the initial neutralization step is 2-ethylhexanol rather than ethanol. Unlike ethanol, 2-ethylhexanol has a much higher boiling point and flash point, substantially reducing fire hazards and VOC emissions during the production cycle. Crucially, this byproduct is not waste; it is a valuable raw material that can be directly recycled into the upstream synthesis of the di(2-ethyl)hexyl phosphite starting material. This closed-loop capability simplifies the production process by reducing the number of distinct raw materials required and eliminating the need for complex ethanol recovery distillation columns. The result is a streamlined workflow that improves overall production efficiency and product quality while offering a inherently safer operating environment for plant personnel.
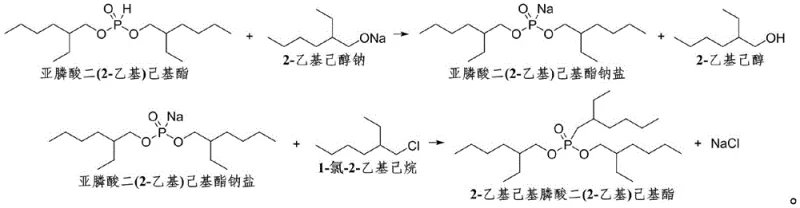
Mechanistic Insights into Sodium 2-ethylhexanoxide Catalyzed Phosphonation
The core of this synthesis relies on a modified Michaelis-Becker reaction mechanism, where the nucleophilicity of the phosphorus center is enhanced through deprotonation. In the first stage, sodium 2-ethylhexanoxide reacts with di(2-ethyl)hexyl phosphite to generate the corresponding sodium salt. The patent specifies that this step must be conducted under reduced pressure, typically between 5kPa and 80kPa, at temperatures ranging from 70°C to 160°C. This vacuum condition is critical for the continuous removal of the generated 2-ethylhexanol, driving the equilibrium towards the formation of the sodium salt. The efficiency of this step is highly sensitive to water content; the data explicitly states that the water content in the sodium alkoxide solution must be maintained at ≤0.2%. Any deviation above this threshold leads to hydrolysis of the phosphite species, drastically reducing the yield of the active nucleophile available for the subsequent alkylation. This mechanistic requirement underscores the need for high-quality, anhydrous reagents to ensure consistent batch-to-batch performance.
In the second stage, the generated sodium salt undergoes nucleophilic substitution with 1-chloro-2-ethylhexane. This alkylation step is performed at elevated temperatures, specifically between 161°C and 250°C, for a duration of 2 to 10 hours. The high thermal energy is necessary to overcome the activation barrier for the displacement of the chloride ion by the bulky phosphinate anion. The molar ratio of the chloro-alkane to the phosphinate salt is optimized between 1.3:1 and 3.0:1 to ensure complete conversion of the limiting reagent while minimizing side reactions such as elimination. The patent examples demonstrate that maintaining these specific thermal and stoichiometric parameters results in yields exceeding 90%, with optimal conditions reaching nearly 97%. This high level of conversion is indicative of a clean reaction profile with minimal formation of phosphonate mono-esters or other degradation products, which is essential for producing high-purity intermediates suitable for sensitive rare earth separation processes.
How to Synthesize Di(2-ethylhexyl) 2-ethylhexylphosphonate Efficiently
To implement this synthesis effectively, operators must adhere to strict control over reagent quality and thermal profiles. The process begins with the preparation of the sodium salt under vacuum to remove the alcohol byproduct, followed by the addition of the alkyl halide for the coupling reaction. Detailed standard operating procedures regarding specific addition rates, stirring speeds, and quenching protocols are essential for reproducibility. For a comprehensive guide on the exact laboratory-to-pilot scale translation of this method, please refer to the standardized synthesis steps outlined below.
- React sodium 2-ethylhexanoxide solution with di(2-ethyl)hexyl phosphite at 70°C-160°C under reduced pressure (5kPa-80kPa) to form the sodium salt, ensuring water content is ≤0.2%.
- Add 1-chloro-2-ethylhexane to the sodium salt mixture and maintain the reaction temperature between 161°C-250°C for 2-10 hours to complete the alkylation.
- Cool the reaction mixture to room temperature, wash with water to remove inorganic salts, and separate the organic phase to obtain the crude ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic advantages beyond mere chemical yield. The primary benefit lies in the substantial simplification of the raw material portfolio. By eliminating sodium ethoxide and the associated ethanol recovery infrastructure, facilities can reduce their inventory complexity and lower capital expenditure on safety equipment. The ability to recycle the 2-ethylhexanol byproduct back into the feedstock stream creates a circular economy within the plant, significantly reducing the net consumption of raw materials. This internal recycling loop acts as a hedge against market price fluctuations for alcohols, providing greater cost stability over the long term. Furthermore, the reduced safety risk profile lowers insurance premiums and regulatory compliance costs, contributing to a leaner operational budget.
- Cost Reduction in Manufacturing: The elimination of ethanol recovery systems represents a major saving in both energy and equipment maintenance. Since the byproduct 2-ethylhexanol is reusable, the effective cost of the base is lowered, and waste disposal fees are minimized. The simplified workup procedure, which avoids complex distillation trains for low-boiling solvents, reduces utility consumption and labor hours per batch. Additionally, the higher reaction yields observed in the patent data mean less raw material is wasted on side products, directly improving the mass balance and reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Relying on fewer distinct raw materials reduces the risk of supply disruptions. Sodium 2-ethylhexanoxide can often be sourced or generated on-site using the same alcohol supply chain as the phosphite ester, consolidating vendor relationships. The robustness of the reaction conditions, which tolerate a range of molar ratios without catastrophic failure, ensures consistent output even if minor variations in feedstock quality occur. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream clients in the rare earth and agrochemical sectors.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of highly volatile solvents that pose explosion risks in large reactors. The higher flash point of the reaction mixture allows for safer operation in standard stainless steel equipment without the need for specialized nitrogen blanketing to the same extent as ethanol-based processes. From an environmental perspective, the reduction in VOC emissions and the minimization of aqueous waste streams align with increasingly stringent global environmental regulations. This compliance facilitates easier permitting for capacity expansion and ensures long-term operational continuity in regions with tight ecological controls.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process robustness and product quality. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer.
Q: Why is sodium 2-ethylhexanoxide preferred over sodium ethoxide in this synthesis?
A: Using sodium 2-ethylhexanoxide eliminates the formation of ethanol as a byproduct. Ethanol has a low flash point and high vapor pressure, posing significant safety risks and requiring complex recovery systems. The new method produces 2-ethylhexanol, which has a higher boiling point and can be directly reused in the upstream synthesis of the phosphite starting material, simplifying the process and enhancing safety.
Q: What is the critical control parameter for maximizing yield in this reaction?
A: Water content in the sodium 2-ethylhexanoxide solution is critical. The patent data indicates that keeping water content ≤0.2% is essential. Comparative examples show that increasing water content to 0.25% causes a drastic drop in yield from over 93% to approximately 82%, likely due to hydrolysis of the reactive intermediates.
Q: How does this method impact the purity of the final P-507 intermediate?
A: This method reduces the types of raw materials and avoids side reactions associated with high concentrations of imide sodium salts found in traditional methods. By matching the alkoxide base to the ester chain, the process minimizes impurity formation, resulting in a clearer oil phase and higher conversion rates, which simplifies downstream purification for high-purity extractant applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Di(2-ethylhexyl) 2-ethylhexylphosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes for high-value organophosphorus intermediates. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN115028660A are fully realized in industrial practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of di(2-ethylhexyl) 2-ethylhexylphosphonate meets the exacting standards required for rare earth extraction and specialty chemical applications. Our commitment to process excellence means we can deliver this complex intermediate with the consistency and reliability your supply chain demands.
We invite you to collaborate with us to optimize your sourcing strategy for phosphonate esters. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our advanced manufacturing capabilities, we can help you reduce lead time for high-purity extraction agents while ensuring full regulatory compliance. Please contact us today to request specific COA data and route feasibility assessments for your next project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
