Scalable Manufacturing of Cytotoxic Indolino Benzodiazepine Dimers for ADC Applications
The pharmaceutical landscape for Antibody-Drug Conjugates (ADCs) is rapidly evolving, driven by the demand for more potent and stable cytotoxic payloads. Patent CN116102615A introduces a significant advancement in the preparation of indolino benzodiazepine derivatives, which serve as critical warheads in next-generation oncology therapeutics. This intellectual property outlines a robust, modular synthetic route that addresses historical challenges associated with dimerization efficiency and impurity profiles. By leveraging a strategic sequence of ether formation, selective nitro reduction, and amide coupling, the disclosed methodology offers a pathway to high-purity intermediates essential for clinical and commercial success. For industry stakeholders, understanding the nuances of this process is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards. The following analysis dissects the technical merits and commercial implications of this novel manufacturing approach.
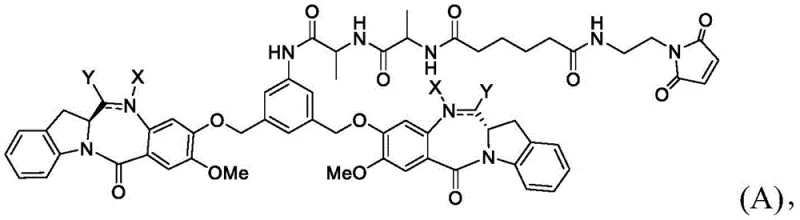
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzodiazepine dimers has been plagued by inefficiencies inherent in late-stage functionalization and harsh reaction conditions. Traditional routes often rely on catalytic hydrogenation using noble metals such as palladium or platinum to reduce nitro precursors to amines, a step that introduces significant risks of heavy metal contamination requiring costly purification protocols. Furthermore, conventional dimerization strategies frequently suffer from poor solubility of intermediates, leading to heterogeneous reaction mixtures that result in inconsistent yields and difficult scale-up parameters. The formation of the central ether linkage, a cornerstone of the dimer structure, often necessitates aggressive alkylating agents that can compromise the stereochemical integrity of the chiral centers within the benzodiazepine rings. These technical bottlenecks not only inflate the cost of goods sold but also extend lead times for high-purity pharmaceutical intermediates, creating vulnerabilities in the supply chain for ADC developers seeking rapid clinical progression.
The Novel Approach
In contrast, the methodology detailed in CN116102615A presents a paradigm shift through its modular design and chemoselective transformations. The process decouples the construction of the dimer core from the attachment of the linker payload, allowing for rigorous quality control at the nitro-dimer stage before proceeding to the final reduction. A standout feature is the substitution of catalytic hydrogenation with an iron-mediated reduction system using ammonium chloride, which operates under mild thermal conditions and eliminates the need for specialized high-pressure equipment. This chemical strategy inherently reduces the burden on downstream purification, as iron salts are easily removed compared to trace noble metals. Additionally, the use of activated ester intermediates for the final coupling step ensures high conversion rates while preserving the sensitive imine or amine functionalities within the benzodiazepine scaffold. This refined approach translates directly into enhanced process robustness and a more predictable manufacturing timeline for complex polymer additives and specialty chemical applications.
Mechanistic Insights into Iron-Mediated Nitro Reduction and Coupling
The core chemical innovation lies in the selective reduction of the nitro group on the dimeric precursor without affecting other reducible functionalities such as the lactam carbonyls or the imine bonds within the benzodiazepine rings. The mechanism involves the transfer of electrons from zero-valent iron to the nitro group in the presence of a proton source like ammonium chloride, proceeding through nitroso and hydroxylamine intermediates to the final aniline derivative. This specific reagent combination is crucial because it maintains a pH environment that prevents the hydrolysis of the acid-sensitive benzodiazepine core, a common failure mode in stronger acidic or basic reduction conditions. The subsequent coupling reaction utilizes advanced activating agents, potentially including phosphonium salts or carbodiimides, to facilitate the amide bond formation between the newly formed aniline and the linker carboxylic acid. This activation strategy minimizes racemization at the alpha-carbon of the amino acid residues in the linker, ensuring the final conjugate retains its intended biological activity and binding affinity.
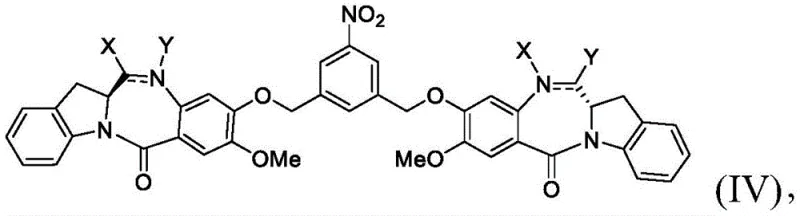
Impurity control is meticulously managed through the modular nature of the synthesis, where the nitro-dimer intermediate serves as a purge point for unreacted monomers and side products before the final commitment steps. The crystallization properties of the nitro-intermediate are superior to the amino-analog, allowing manufacturers to achieve high purity levels prior to reduction. Furthermore, the choice of solvents, such as mixtures of tetrahydrofuran and methanol with water, is optimized to maintain the solubility of the polar dimeric species while facilitating the precipitation of inorganic iron byproducts. This careful balance of solvation and reactivity prevents the formation of over-reduced byproducts or oligomeric impurities that could complicate the toxicological profile of the final drug substance. Such attention to mechanistic detail underscores the feasibility of this route for producing high-purity OLED material grade intermediates or similar high-value fine chemicals where impurity thresholds are exceptionally low.
How to Synthesize Indolino Benzodiazepine Dimers Efficiently
The practical execution of this synthesis requires precise control over stoichiometry and temperature to maximize yield and safety. The process begins with the preparation of the nitro-dimer scaffold, followed by the critical reduction step and final conjugation. Operators must adhere to strict anhydrous conditions during the coupling phase to prevent hydrolysis of the activated linker species. Detailed standard operating procedures regarding the addition rates of reducing agents and the management of exotherms are essential for safe commercial scale-up of complex pharmaceutical intermediates. The following guide outlines the standardized synthesis steps derived from the patent claims.
- Prepare the nitro-dimer precursor via ether linkage formation using activated alcohol intermediates.
- Execute selective nitro group reduction to amine using iron powder and ammonium chloride in aqueous organic solvent.
- Couple the resulting amino-dimer with the activated linker payload using carbodiimide or phosphonium activators.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial cost savings and supply chain resilience. The elimination of noble metal catalysts removes a volatile cost component from the bill of materials, as prices for palladium and platinum fluctuate wildly based on geopolitical factors. Moreover, the reliance on commodity chemicals like iron powder and ammonium chloride ensures that raw material availability remains stable even during global supply disruptions. The simplified workup procedures reduce the consumption of chromatography resins and solvents, contributing to a lower environmental footprint and reduced waste disposal costs. For supply chain heads, the robustness of the nitro-intermediate allows for strategic stockpiling of semi-finished goods, decoupling the long lead time of dimer synthesis from the final custom coupling steps required by specific ADC programs. This flexibility significantly enhances supply chain reliability and reduces the risk of production delays impacting clinical trial timelines.
- Cost Reduction in Manufacturing: The transition to iron-mediated reduction drastically simplifies the reactor setup by removing the need for high-pressure hydrogenation vessels, which require specialized maintenance and safety certifications. This capital expenditure avoidance allows manufacturing partners to allocate resources towards capacity expansion rather than equipment upgrades. Additionally, the higher selectivity of the reaction minimizes the loss of expensive chiral starting materials, ensuring that the overall mass balance of the process is optimized for economic efficiency. The reduction in purification steps further lowers the operational costs associated with solvent recovery and energy consumption, delivering a leaner manufacturing model that supports competitive pricing strategies for cost reduction in ADC payload manufacturing.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents and avoiding proprietary catalysts that may be subject to export controls or single-source limitations, this process diversifies the supply base. The ability to synthesize the core dimer independently of the specific linker payload means that inventory can be held in a stable nitro-form, ready for rapid conversion upon demand. This decoupling strategy mitigates the risk of obsolescence and allows for a more responsive Just-In-Time delivery model. Consequently, partners can guarantee reducing lead time for high-purity pharmaceutical intermediates, providing a critical advantage in the fast-paced oncology drug development sector where speed to market is paramount.
- Scalability and Environmental Compliance: The aqueous workup associated with the iron reduction generates waste streams that are easier to treat compared to those containing heavy metals, aligning with increasingly stringent environmental regulations. The process demonstrates excellent scalability from kilogram to multi-ton scales without significant re-optimization, as the heat transfer and mixing requirements are less demanding than heterogeneous hydrogenation. This inherent scalability ensures that the technology can support the transition from clinical trials to commercial launch without the need for process redesign. Furthermore, the reduced solvent intensity and safer reagent profile contribute to a greener manufacturing process, enhancing the sustainability credentials of the final therapeutic product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this supply source into your existing development pipeline.
Q: What is the primary advantage of the iron-mediated reduction method described in CN116102615A?
A: The use of iron powder and ammonium chloride avoids expensive noble metal catalysts like palladium, significantly reducing heavy metal impurity risks and lowering raw material costs for large-scale production.
Q: How does this process address the stability issues of benzodiazepine dimers?
A: The modular approach allows for the isolation of stable nitro-intermediates before the final reduction and coupling steps, minimizing decomposition and improving overall yield consistency.
Q: Is this synthesis route suitable for GMP manufacturing of ADC payloads?
A: Yes, the process utilizes standard reagents and avoids hazardous high-pressure hydrogenation, making it highly adaptable for GMP-compliant facilities focusing on potent compound handling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolino Benzodiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality cytotoxic payloads play in the success of modern ADC therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle potent compounds safely, providing a secure environment for the manufacture of sensitive benzodiazepine derivatives.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of this method compared to your current supply chain. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate our capabilities against your technical needs. Let us collaborate to accelerate your drug development timeline with reliable, cost-effective, and high-quality chemical solutions.
