Advanced Synthesis of N-Boc-hexahydro-5-oxo-cyclopenta(C)pyrrole for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for critical intermediates, particularly those serving oncology and cardiovascular therapeutic areas. Patent CN103242216A introduces a transformative synthesis method for N-Boc-hexahydro-5-oxo-cyclopenta(C)pyrrole, a pivotal building block used in the development of antitumor agents, antibacterials, FSH receptor agonists, and renin inhibitors. This innovation addresses long-standing inefficiencies in prior art by shifting away from hazardous alkyne chemistry toward a stable, high-yielding route based on tetrahydrophthalimide. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize supply chains and reduce the cost of goods sold (COGS) for complex heterocyclic scaffolds. By leveraging a four-step sequence involving reduction, protection, oxidation, and cyclization, the method achieves total yields exceeding 50%, a dramatic improvement over previous benchmarks. This report analyzes the technical merits and commercial implications of this novel approach, positioning it as a preferred strategy for reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-Boc-hexahydro-5-oxo-cyclopenta(C)pyrrole relied on a convoluted pathway initiating with the reaction of allylamine and 3-propargyl bromide to form propenyl propargyl amine. This intermediate was subsequently protected and subjected to cyclization using cobalt octacarbonyl as a catalyst. This legacy approach suffers from severe economic and technical drawbacks that hinder industrial viability. Firstly, the raw materials, specifically specialized alkynes and bromides, are significantly more expensive and less readily available than bulk commodity chemicals. Secondly, the reliance on cobalt octacarbonyl introduces substantial safety hazards due to its toxicity and sensitivity, alongside the risk of heavy metal contamination in the final API. Furthermore, the conventional route is plagued by low atom economy and poor selectivity; the propensity for raw material polymerization and competing side reactions results in dismal yields often falling below 20%. Such inefficiencies create bottlenecks in the supply chain, driving up costs and extending lead times for downstream drug manufacturers who require consistent, high-volume supplies.
The Novel Approach
In stark contrast, the methodology disclosed in CN103242216A utilizes tetrahydrophthalimide as the starting material, a compound known for its stability, low cost, and widespread availability in the fine chemical market. This new route bypasses the hazardous alkyne chemistry entirely, replacing it with a streamlined sequence of reduction, Boc-protection, oxidative cleavage, and thermal cyclization. The strategic selection of tetrahydrophthalimide allows for a highly controlled transformation where side reactions are minimized, and purification is simplified. By avoiding transition metal catalysts like cobalt, the process inherently produces a cleaner crude profile, reducing the burden on downstream purification units. The visual representation of this superior synthetic pathway highlights the logical progression from a bicyclic imide to the target fused pyrrolidinone system.
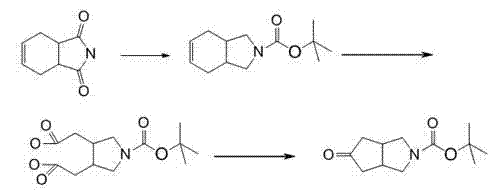
This structural evolution not only enhances safety but also drastically improves the overall mass balance of the production process. The ability to achieve yields in the range of 50% to 60% effectively triples the output per batch compared to traditional methods, offering immediate scalability benefits for commercial operations.
Mechanistic Insights into Tetrahydrophthalimide Reduction and Oxidative Cyclization
The core of this technological breakthrough lies in the precise manipulation of the tetrahydrophthalimide scaffold through a carefully orchestrated series of redox reactions. The first stage involves the reduction of the imide carbonyls using strong hydride sources such as Lithium Aluminium Hydride (LiAlH4) or Red-Al in solvents like tetrahydrofuran (THF) at temperatures between 30°C and 70°C. This step converts the rigid imide structure into a reactive amino-alcohol intermediate, which is immediately trapped with di-tert-butyl dicarbonate (Boc2O) under basic conditions to form N-Boc-2,3,3a,4,7,7a-hexahydroisoindole. This in-situ protection strategy is crucial for preventing over-reaction and stabilizing the nitrogen center for subsequent transformations. The second critical phase is the oxidative cleavage of the cyclohexene double bond. Utilizing oxidants like potassium permanganate or sodium periodate in the presence of phase transfer catalysts (e.g., tetrabutylammonium hydrogen sulfate), the carbon-carbon double bond is severed to generate a dicarbonyl species, referred to in the patent as an oxalic acid derivative. This step proceeds under mild aqueous conditions, demonstrating excellent functional group tolerance.
Following oxidation, the final cyclization step leverages the inherent reactivity of the generated dialdehyde or keto-acid intermediate. Heating the precursor in acetic anhydride with sodium acetate at 110°C to 125°C promotes an intramolecular condensation or aldol-type cyclization, closing the five-membered ring to form the target cyclopenta[c]pyrrole core. The mechanism ensures high regioselectivity, driven by the thermodynamic stability of the newly formed fused ring system. Impurity control is inherently managed by the specificity of the oxidants and the crystallization properties of the intermediates. For instance, the intermediate N-Boc-3,4-tetramethyleneimine oxalic acid precipitates as a white solid upon acidification, allowing for a simple filtration wash that removes soluble organic impurities before the final cyclization. This "purify-as-you-go" approach minimizes the accumulation of byproducts, ensuring the final product meets purity specifications of 95% to 98% without requiring resource-intensive chromatography.
How to Synthesize N-Boc-hexahydro-5-oxo-cyclopenta(C)pyrrole Efficiently
Implementing this synthesis requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent to maximize yield and safety. The process begins with the dissolution of tetrahydrophthalimide in dry THF, followed by the controlled addition of the reducing agent to manage exotherms. After the reduction and subsequent Boc-protection, the intermediate is isolated via distillation. The oxidation step is performed in water, leveraging the solubility differences to facilitate product isolation via pH swing. Finally, the cyclization in acetic anhydride must be monitored to prevent degradation, followed by a workup involving ethyl acetate extraction and recrystallization from mixed solvents like ethyl acetate and petroleum ether.
- Perform reduction of tetrahydrophthalimide using LiAlH4 or Red-Al followed by Boc-protection with di-tert-butyl dicarbonate to form N-Boc-2,3,3a,4,7,7a-hexahydroisoindole.
- Execute oxidative cleavage of the intermediate using potassium permanganate or sodium periodate with a phase transfer catalyst to generate the dicarbonyl precursor.
- Complete the synthesis via thermal cyclization in acetic anhydride with sodium acetate, followed by pH adjustment and recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers profound strategic advantages beyond mere technical elegance. The shift from specialized alkynes to tetrahydrophthalimide fundamentally alters the cost structure of the intermediate. Tetrahydrophthalimide is a bulk commodity chemical produced at massive scales for various industries, ensuring a stable, multi-vendor supply base that mitigates the risk of raw material shortages. In contrast, the alkyne precursors used in older methods are often custom-synthesized specialty chemicals with volatile pricing and limited availability. By switching to this novel route, manufacturers can achieve substantial cost savings simply through raw material arbitrage and improved yield efficiency. The elimination of cobalt octacarbonyl further reduces costs by removing the need for expensive heavy metal scavengers and rigorous residual metal testing, which are mandatory for pharmaceutical compliance.
- Cost Reduction in Manufacturing: The tripling of overall yield from less than 20% to over 50% means that for every kilogram of final product, significantly less raw material and solvent volume is required. This drastic improvement in mass efficiency directly lowers the variable cost per unit. Furthermore, the avoidance of precious or toxic metal catalysts eliminates a major cost center associated with catalyst recovery and waste disposal. The simplified purification protocol, relying on crystallization rather than chromatography, reduces solvent consumption and processing time, leading to lower utility and labor costs per batch. These factors combine to create a highly competitive cost position for this intermediate in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is critical for pharmaceutical production, and this route enhances reliability by utilizing robust, non-hazardous reagents. The starting material, tetrahydrophthalimide, is not subject to the same regulatory restrictions or transportation hazards as pyrophoric reducing agents or toxic cobalt complexes. This simplifies logistics and warehousing requirements. Additionally, the high yield and reproducibility of the process reduce the likelihood of batch failures, ensuring consistent delivery schedules to downstream API manufacturers. The ability to source key inputs from multiple global suppliers prevents single-source bottlenecks, providing procurement teams with greater negotiating power and supply security.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is far superior. The removal of cobalt reduces the toxicity of the waste stream, simplifying effluent treatment and lowering disposal fees. The use of water as a solvent for the oxidation step aligns with green chemistry principles, reducing the reliance on volatile organic compounds (VOCs). The mild reaction conditions (mostly below 125°C) reduce energy consumption compared to high-pressure or cryogenic alternatives. These attributes make the process easier to scale from pilot plant to multi-ton commercial production without encountering the safety barriers often associated with organometallic chemistry, facilitating faster technology transfer and regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific experimental data and comparative analysis provided in the patent documentation. Understanding these details is essential for evaluating the feasibility of adopting this route for your specific production needs.
Q: What are the primary advantages of the tetrahydrophthalimide route over conventional alkyne methods?
A: The tetrahydrophthalimide route avoids expensive and toxic cobalt octacarbonyl catalysts and unstable alkyne raw materials. It significantly improves total yield from less than 20% to over 50%, reduces side reactions like polymerization, and utilizes low-cost, commercially available starting materials.
Q: What purity levels can be achieved with this synthesis method?
A: According to patent data, the final product achieves a purity range of 95% to 98% after standard recrystallization processes, meeting stringent requirements for pharmaceutical intermediate applications without needing complex chromatographic purification.
Q: Is this process scalable for industrial production?
A: Yes, the process uses mild reaction conditions (30-125°C) and common solvents like THF, water, and ethyl acetate. The elimination of sensitive organometallic catalysts and the use of robust oxidation steps make it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Boc-hexahydro-5-oxo-cyclopenta(C)pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the pathway described in CN103242216A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient synthesis to market. We are equipped with state-of-the-art reactors capable of handling the specific thermal and safety profiles of the reduction and oxidation steps, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs utilize advanced analytical techniques to verify identity and purity, guaranteeing that the N-Boc-hexahydro-5-oxo-cyclopenta(C)pyrrole we supply is free from the heavy metal contaminants associated with older synthetic routes.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this cost-effective technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of this vital pharmaceutical intermediate, enabling you to focus on what matters most: delivering innovative therapies to patients worldwide.
