Advanced DD-pi-AA Porphyrin Synthesis: Scaling High-Efficiency DSSC Materials for Global Markets
Advanced DD-pi-AA Porphyrin Synthesis: Scaling High-Efficiency DSSC Materials for Global Markets
The landscape of dye-sensitized solar cells (DSSC) is undergoing a significant transformation driven by the need for higher photoelectric conversion efficiencies and improved stability. A pivotal advancement in this domain is detailed in patent CN103044951B, which discloses an asymmetric synthetic method for double para-position donor-acceptor type porphyrin molecules. This technology represents a strategic leap from traditional one-dimensional charge transfer systems to a more sophisticated two-dimensional architecture. By chemically modifying the porphyrin core to include dual electron-donating groups at the para-positions, manufacturers can achieve a unique DD-pi-AA structure that significantly enhances light-induced charge transfer capabilities. For R&D directors and procurement specialists in the electronic materials sector, understanding the nuances of this synthesis is critical for securing a competitive edge in the renewable energy supply chain.
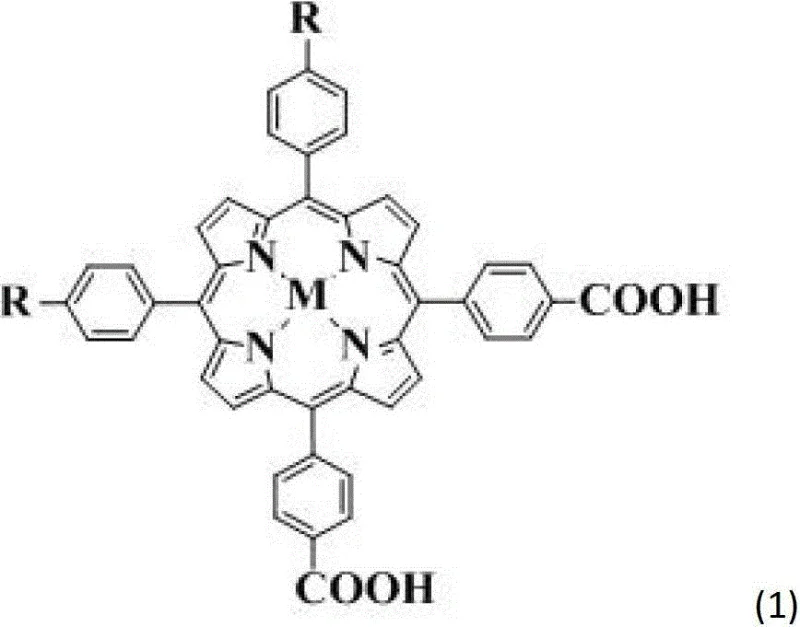
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, porphyrin sensitizers for DSSC applications have predominantly relied on a D-pi-A (Donor-pi-Acceptor) structural motif. While these molecules have demonstrated reasonable success, they suffer from inherent limitations regarding molecular aggregation and restricted charge transfer pathways. In conventional single charge transfer systems, the electron flow is largely unidirectional, which can lead to bottlenecks in electron injection efficiency when scaled up for commercial module production. Furthermore, planar porphyrin structures tend to aggregate on the semiconductor surface, a phenomenon known as pi-pi stacking, which drastically reduces the open-circuit voltage and overall cell efficiency. Traditional synthetic routes often struggle to introduce sufficient steric hindrance to prevent this aggregation without compromising the conjugation system necessary for light absorption.
The Novel Approach
The methodology outlined in the patent data introduces a paradigm shift by extending the molecular design from one-dimensional to two-dimensional charge transfer. By synthesizing double para-position donor-acceptor (DD-pi-AA) porphyrin molecules, the new approach effectively creates a dual-channel system for electron transport. This structural modification not only broadens the absorption spectrum across the solar range but also incorporates bulky donor groups that act as insulating spacers. These spacers physically prevent the porphyrin cores from stacking too closely on the TiO2 surface, thereby mitigating aggregation losses. The result is a dye molecule that maintains high molar extinction coefficients while offering superior stability and charge separation kinetics, addressing the core pain points of previous generation sensitizers.
Mechanistic Insights into Palladium-Catalyzed Asymmetric Synthesis
The core of this technological breakthrough lies in a robust multi-step synthetic sequence that combines the classic Adler condensation with modern transition-metal catalysis. The process begins with the acid-catalyzed condensation of bromobenzaldehyde and methyl formylbenzoate with pyrrole in a propionic acid solvent at elevated temperatures (approximately 140°C). This step constructs the fundamental porphyrin macrocycle with high precision. Following the formation of the free-base porphyrin, the protocol employs a metalation step where metal acetates, such as zinc or iron, are complexed into the central cavity. This metalation is crucial as it stabilizes the excited state of the molecule and tunes the HOMO-LUMO energy levels for optimal alignment with the semiconductor conduction band.
Subsequently, the most critical functionalization occurs via a palladium-catalyzed amination reaction, often referred to as a Buchwald-Hartwig coupling. In this step, the bromo-substituted porphyrin intermediate reacts with bulky arylamines or alkylamines in the presence of a palladium catalyst (e.g., Pd2(dba)3) and a phosphine ligand (e.g., BINAP). This reaction installs the electron-donating groups at the specific para-positions required for the DD-pi-AA architecture. The use of bulky amines, such as bis(4-tert-butylphenyl)amine, serves a dual purpose: it acts as a strong electron donor to push electron density into the porphyrin ring, and its steric bulk provides the necessary physical barrier against molecular aggregation. Finally, the ester groups are hydrolyzed under alkaline conditions to yield the carboxylic acid anchors, which are essential for covalently binding the dye to the metal oxide surface.
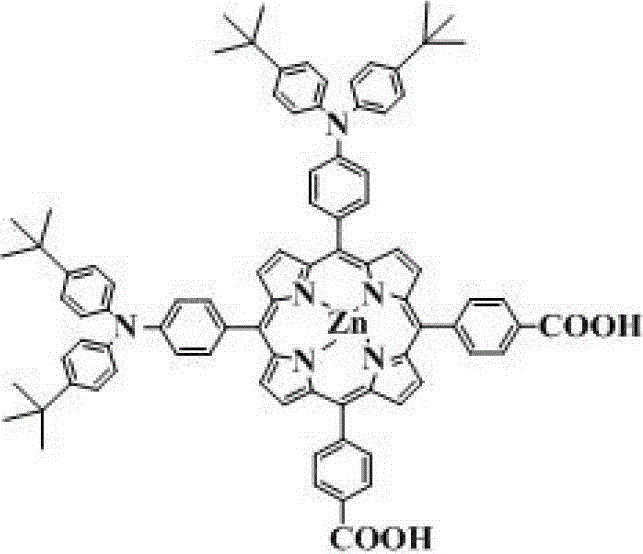
How to Synthesize Double Para-Position Porphyrin Dyes Efficiently
The synthesis of these advanced photovoltaic materials requires precise control over reaction conditions to ensure high purity and yield. The patented procedure offers a standardized workflow that balances reaction efficiency with product quality, making it suitable for both laboratory optimization and pilot-scale production. The process leverages widely available starting materials and established purification techniques, reducing the barrier to entry for manufacturers looking to adopt this technology. For a detailed breakdown of the specific stoichiometric ratios, temperature profiles, and workup procedures required to replicate this synthesis, please refer to the standardized guide below.
- Condense bromobenzaldehyde and methyl formylbenzoate with pyrrole in propionic acid at 140°C to form the porphyrin macrocycle precursor.
- Complex the porphyrin precursor with metal acetates (such as Zinc or Iron) in a methanol/dichloromethane mixture to stabilize the core structure.
- Perform a palladium-catalyzed amination reaction using bulky arylamines to install the dual electron-donating groups at the para-positions.
- Hydrolyze the ester groups under alkaline conditions to reveal the carboxylic acid anchors required for semiconductor binding.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DD-pi-AA porphyrin synthesis route offers tangible strategic benefits beyond mere performance metrics. The reliance on the Adler condensation method ensures that the foundational porphyrin skeleton is generated using commodity chemicals like pyrrole and substituted benzaldehydes, which are readily available in the global market. This reduces the risk of supply chain disruptions associated with exotic or proprietary precursors. Furthermore, the modular nature of the palladium-catalyzed amination step allows for the easy substitution of different amine donors without overhauling the entire synthetic process, providing flexibility in raw material sourcing based on cost and availability fluctuations.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for complex, multi-step protection and deprotection strategies often seen in fine-tuned organic synthesis. By utilizing direct condensation and efficient coupling reactions, the overall step count is minimized, which directly correlates to reduced labor costs and lower solvent consumption. The high yields reported in the experimental data for the metalation and coupling steps further contribute to cost efficiency by maximizing the output per batch. Additionally, the use of standard column chromatography for purification avoids the need for expensive preparative HPLC equipment, significantly lowering capital expenditure requirements for production facilities.
- Enhanced Supply Chain Reliability: The robustness of the chemistry involved ensures consistent batch-to-batch reproducibility, a critical factor for long-term supply contracts. The starting materials, including various metal acetates and substituted anilines, are produced by multiple global suppliers, preventing single-source dependency. This diversification of the supply base mitigates the risk of price spikes or shortages. Moreover, the stability of the intermediate porphyrin species allows for potential stockpiling of key precursors, enabling manufacturers to respond rapidly to surges in demand from the solar energy sector without lengthy lead times.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram or ton-scale commercial production. The solvents used, such as dichloromethane, toluene, and methanol, are standard industrial solvents with well-established recovery and recycling protocols, facilitating compliance with environmental regulations. The elimination of heavy metal catalysts in the final product through rigorous purification ensures that the resulting dye meets the stringent purity specifications required for electronic applications, reducing the burden on downstream waste treatment processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this porphyrin synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and performance expectations for potential partners and licensees.
Q: What is the primary advantage of the DD-pi-AA structure over traditional D-pi-A porphyrins?
A: The double para-position donor-acceptor (DD-pi-AA) structure facilitates a two-dimensional charge transfer pathway, theoretically offering stronger light-induced charge transfer capabilities and reduced molecular aggregation compared to traditional one-dimensional D-pi-A systems.
Q: Which metals are compatible with this porphyrin synthesis protocol?
A: The protocol supports the incorporation of various metal ions including Copper (Cu), Cobalt (Co), Iron (Fe), Nickel (Ni), Zinc (Zn), and Magnesium (Mg), allowing for tunable electronic properties.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes the classic Adler condensation followed by robust palladium-catalyzed coupling, avoiding overly sensitive reagents and relying on standard purification techniques like column chromatography, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Porphyrin Dye Supplier
As the global demand for high-efficiency renewable energy materials continues to surge, the ability to reliably manufacture complex organic sensitizers becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in organometallic chemistry and process optimization to deliver superior photovoltaic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of porphyrin dye meets the exacting standards required for high-performance DSSC modules.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods sold. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific production volumes. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our manufacturing capabilities can accelerate your product development timeline and enhance your market competitiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
