Advanced Synthesis of Mononuclear Nickel Diphosphine Complexes for Industrial Electrocatalysis
Advanced Synthesis of Mononuclear Nickel Diphosphine Complexes for Industrial Electrocatalysis
The global transition towards sustainable energy systems has intensified the search for efficient, non-precious metal catalysts capable of driving the hydrogen evolution reaction (HER). Addressing this critical technological gap, patent CN108285473B discloses a groundbreaking methodology for the synthesis of novel mononuclear nickel metal compounds containing specialized diphosphine ligands. These complexes, specifically characterized by the formula Ni(S2P{O}OR)[(Ph2P)2NCH(CH3)C6H5], represent a significant leap forward in electrocatalytic material science. Unlike traditional methods that rely on harsh thermal conditions, this innovation utilizes a mild, room-temperature stirring protocol that dramatically simplifies the manufacturing workflow while maintaining exceptional product integrity. For R&D directors and procurement specialists seeking reliable nickel catalyst suppliers, this technology offers a robust pathway to producing high-performance materials essential for next-generation fuel cells and electrolyzers.
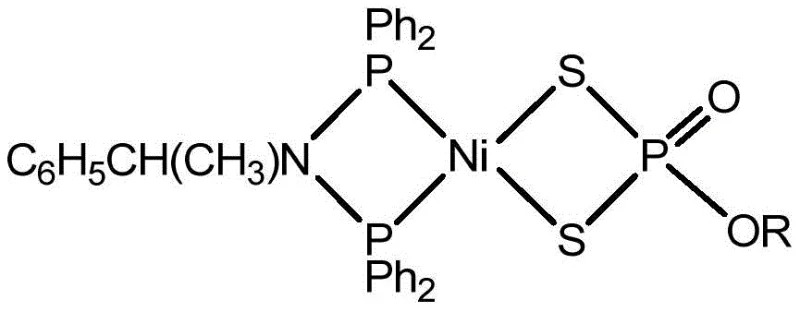
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mononuclear nickel complexes for catalytic applications has been plagued by inefficient protocols that hinder large-scale adoption. Conventional routes typically necessitate prolonged heating under reflux conditions, often requiring elevated temperatures that consume substantial energy resources and increase the carbon footprint of the manufacturing process. Furthermore, these thermal methods frequently lead to the formation of unwanted side products and decomposition intermediates, complicating the downstream purification stages and ultimately depressing the overall yield of the target catalyst. The reliance on rigid diphosphine ligands such as dppe (1,2-bis(diphenylphosphino)ethane) in older methodologies often results in complexes with suboptimal electronic properties, limiting their turnover frequency in proton reduction reactions. Consequently, the industry has faced a persistent bottleneck in sourcing high-purity electrocatalysts at a cost structure that competes with established platinum-based systems, creating a pressing need for process innovation.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in patent CN108285473B introduces a paradigm shift by enabling the formation of the target nickel complex through simple stirring at ambient temperature. This approach leverages the unique reactivity of the chiral bisphosphine ligand (Ph2P)2NCH(CH3)C6H5, which coordinates efficiently with the nickel precursor without the need for thermal activation. By operating at 25-30°C, the process not only slashes energy consumption but also preserves the structural fidelity of the sensitive organometallic bonds, resulting in significantly cleaner reaction profiles. The operational simplicity extends to the workup phase, where standard solvent removal and extraction techniques suffice to isolate the product, bypassing the need for complex crystallization or distillation setups. This streamlined workflow translates directly into cost reduction in electrocatalyst manufacturing, making the commercial scale-up of complex catalysts far more economically viable for industrial partners.
Mechanistic Insights into Ligand Coordination and Complex Stability
The efficacy of this synthesis lies in the precise electronic and steric interplay between the nickel center and the incoming diphosphine ligand. The reaction proceeds via a ligand exchange mechanism where the bidentate nitrogen-phosphorus ligand displaces weaker coordinating groups on the nickel precursor Ni(S2P{OR}2)2. The presence of the chiral center at the CH(CH3)C6H5 moiety introduces specific stereochemical constraints that stabilize the resulting mononuclear geometry, preventing the formation of inactive polynuclear aggregates that often plague nickel chemistry. The electron-donating capability of the phosphine groups enhances the electron density at the nickel center, a critical factor for facilitating the subsequent electrocatalytic proton reduction steps. This tailored electronic environment ensures that the final complex exhibits superior conductivity and catalytic turnover compared to analogues synthesized via high-temperature routes, providing R&D teams with a material that delivers consistent performance in weak acid media.
Furthermore, the stability of the P=O and P-S bonds within the coordination sphere plays a pivotal role in the longevity of the catalyst under operating conditions. The synthetic route ensures that these functional groups remain intact, avoiding the hydrolysis or oxidation issues common in less controlled environments. The use of chloroform as a solvent provides an ideal medium for solubilizing both the organic ligand and the inorganic nickel salt, fostering a homogeneous reaction environment that maximizes collision frequency between reactants. This mechanistic understanding allows for fine-tuning of the R group (methyl or ethyl) to modulate the solubility and lipophilicity of the final product, offering customization options for specific electrolyte formulations. Such depth of control over the molecular architecture is essential for developing high-purity organometallic complexes that meet the rigorous specifications of advanced energy applications.
How to Synthesize Mononuclear Nickel Diphosphine Complex Efficiently
The practical implementation of this synthesis is designed for seamless integration into existing laboratory and pilot plant infrastructure. The process begins with the precise weighing of the nickel precursor and the chiral ligand, followed by their dissolution in chloroform to form a homogenous red solution. This mixture is then subjected to magnetic stirring at room temperature for a duration of 4 to 8 hours, allowing the coordination equilibrium to shift fully towards the product. Following the reaction period, the solvent is removed under reduced pressure, and the crude residue is extracted with dichloromethane. Final purification is achieved through thin-layer chromatography or column chromatography using a dichloromethane-methanol gradient, yielding the target complex with high purity. The detailed standardized synthesis steps for scaling this reaction are provided in the guide below.
- Dissolve Ni(S2P{OR}2)2 and the chiral bisphosphine ligand (Ph2P)2NCH(CH3)C6H5 in chloroform at a molar ratio of 1: 1 under ambient conditions.
- Stir the reaction mixture at room temperature (25-30°C) for 4 to 8 hours to ensure complete ligand coordination and complex formation.
- Remove the solvent via rotary evaporation, extract the residue with dichloromethane, and purify the final product using silica gel chromatography.
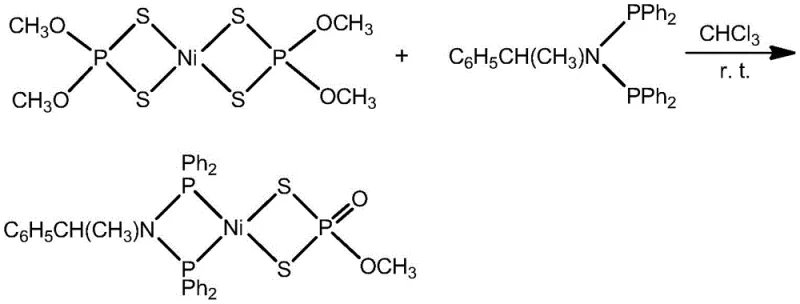
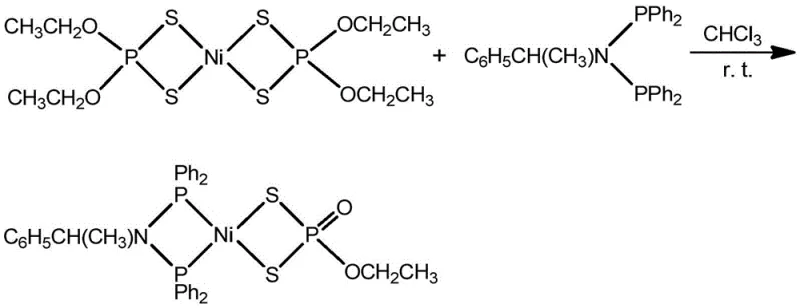
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this room-temperature synthesis route offers compelling strategic benefits that extend beyond mere technical performance. The elimination of heating equipment and the reduction in reaction time significantly lower the utility costs associated with production, directly impacting the bottom line. Moreover, the mild conditions enhance workplace safety by removing the risks associated with high-temperature pressurized vessels, thereby reducing insurance premiums and regulatory compliance burdens. The simplicity of the purification process means that fewer specialized separation columns or distillation towers are required, allowing for a more flexible and modular production setup that can be rapidly deployed to meet fluctuating market demands.
- Cost Reduction in Manufacturing: The transition from thermal reflux to ambient stirring fundamentally alters the cost structure of catalyst production. By removing the need for continuous energy input to maintain high temperatures, the process achieves substantial energy savings that accumulate significantly over large batch sizes. Additionally, the high yield range of 60-75% minimizes raw material waste, ensuring that expensive phosphine ligands and nickel salts are utilized with maximum efficiency. The simplified workup procedure reduces the consumption of auxiliary solvents and stationary phases for purification, further driving down the variable costs per kilogram of the final product. These cumulative efficiencies position this method as a highly competitive option for cost-sensitive industrial applications.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as chloroform, dichloromethane, and commercially accessible nickel precursors ensures a robust and resilient supply chain. Unlike processes that depend on exotic reagents or custom-synthesized intermediates with long lead times, this route utilizes commodity chemicals that can be sourced from multiple vendors globally. The short reaction time of 4 to 8 hours allows for rapid turnaround of production batches, enabling manufacturers to respond quickly to urgent orders or sudden spikes in demand. This agility is crucial for maintaining continuity in the supply of critical electrocatalytic materials to downstream energy sector clients.
- Scalability and Environmental Compliance: The benign nature of the reaction conditions facilitates straightforward scale-up from gram-scale laboratory experiments to multi-ton commercial production without the need for complex engineering redesigns. The absence of high-temperature exotherms reduces the risk of thermal runaway, making the process inherently safer for large reactor vessels. Furthermore, the use of standard organic solvents allows for established recycling and recovery protocols, minimizing the environmental impact of solvent waste. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly stringent environmental regulations governing chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their operations, we have compiled answers to common inquiries regarding the synthesis and application of these nickel complexes. The following insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial planning. These questions address key concerns regarding scalability, purity, and performance metrics that are critical for decision-making in R&D and procurement departments.
Q: What are the advantages of room-temperature synthesis for nickel catalysts?
A: Room-temperature synthesis eliminates the need for energy-intensive heating reflux, significantly reducing operational costs and minimizing thermal degradation byproducts, leading to higher purity yields between 60-75%.
Q: Can this nickel complex be scaled for industrial hydrogen production?
A: Yes, the mild reaction conditions and simple workup procedure involving standard solvent extraction make this process highly amenable to commercial scale-up from kilogram to multi-ton production levels.
Q: What is the specific application of this mononuclear nickel compound?
A: This compound serves as an efficient electrocatalyst for proton reduction to generate hydrogen gas, offering a cost-effective alternative to precious metal catalysts like platinum in renewable energy systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mononuclear Nickel Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this room-temperature synthesis route for the broader electrocatalysis industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for high-performance catalytic applications. We are committed to delivering consistent quality and reliability, leveraging our deep expertise in organometallic chemistry to support your energy innovation goals.
We invite you to collaborate with us to optimize your supply chain for next-generation hydrogen technologies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can accelerate your project timelines while reducing overall production costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
