Advanced One-Pot Synthesis of Tribromoneopentanol Acetate for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational safety, particularly for complex intermediates like tribromoneopentanol acetate. A significant breakthrough in this domain is detailed in patent CN110903188A, which discloses a highly efficient method for preparing this critical compound. Unlike traditional approaches that often struggle with low conversion rates or hazardous gas handling, this novel technique leverages an elegant in-situ generation of hydrobromic acid. By utilizing sulfur powder to reduce elemental bromine directly within the reaction matrix, the process circumvents the logistical and safety challenges associated with storing and transporting pressurized HBr gas. This innovation not only streamlines the workflow but also enhances the selectivity towards the desired acetate ester, making it a compelling candidate for reliable pharma intermediate supplier networks aiming to optimize their supply chains.
Furthermore, the versatility of this synthesis extends beyond mere convenience; it addresses the fundamental economic drivers of modern chemical manufacturing. The ability to achieve yields exceeding 90% with product purity greater than 99% represents a substantial leap forward in process efficiency. For R&D teams focused on the synthesis of Montelukast sodium side chains or advanced flame retardants, this level of quality control is paramount. The method effectively transforms a multi-step, hazard-prone operation into a streamlined, one-pot procedure that minimizes waste and maximizes throughput. As we delve deeper into the technical specifics, it becomes clear that this approach offers a sustainable pathway for the commercial scale-up of complex pharmaceutical intermediates, aligning perfectly with the rigorous demands of global regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of brominated neopentyl derivatives has been fraught with significant technical hurdles that impede efficient large-scale production. Most prior art, such as the methods described in earlier patents, focuses primarily on the preparation of tribromoneopentyl alcohol rather than its acetate ester. In these conventional routes, pentaerythritol is typically reacted with gaseous hydrogen bromide, a process that requires specialized corrosion-resistant equipment and stringent safety protocols to manage the toxic and corrosive nature of the gas. Moreover, these methods often suffer from poor selectivity, resulting in a mixture of mono-, di-, and tri-brominated species that are difficult to separate. The subsequent esterification step, if required to obtain the acetate, adds another layer of complexity, involving additional reagents, separation stages, and potential yield losses. Consequently, the overall atom economy is low, and the presence of stubborn impurities often necessitates energy-intensive purification steps like repeated recrystallization or column chromatography.
The Novel Approach
In stark contrast, the methodology outlined in CN110903188A introduces a paradigm shift by integrating the bromination and esterification logic into a cohesive, safer workflow. By employing sulfur powder as a reducing agent, the system generates hydrobromic acid exactly where and when it is needed, directly within the acetic acid solvent medium. This in-situ generation ensures a steady, controlled release of the brominating species, which drastically reduces the formation of unwanted poly-brominated byproducts. The reaction conditions are optimized to favor the formation of the tribromo-acetate directly, bypassing the isolation of the alcohol intermediate entirely. This telescoping of steps not only reduces the total processing time but also minimizes the exposure of operators to hazardous materials. The result is a process that is inherently safer, more cost-effective, and capable of delivering the high-purity specifications required for sensitive pharmaceutical applications without the baggage of legacy inefficiencies.
Mechanistic Insights into Sulfur-Mediated Reductive Bromination
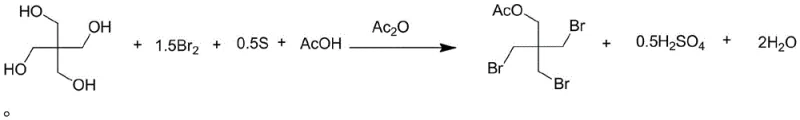
The core of this technological advancement lies in the sophisticated interplay between elemental sulfur and bromine within an acidic medium. When sulfur powder is introduced to the reaction mixture containing bromine and acetic acid, it acts as a potent reducing agent, undergoing oxidation while simultaneously reducing molecular bromine to hydrogen bromide. This redox reaction is exothermic but manageable, providing a continuous source of HBr that reacts immediately with the hydroxyl groups of the pentaerythritol substrate. The mechanism proceeds through a selective substitution pathway where three of the four hydroxyl groups are replaced by bromine atoms, while the fourth undergoes acetylation. The presence of acetic anhydride in the later stages ensures that any remaining hydroxyl functionality or intermediate alcohol species are converted to the stable acetate ester. This dual-functionality of the reaction environment—acting as both a bromination and acetylation vessel—is key to the high selectivity observed.
Controlling the impurity profile is another critical aspect of this mechanism, achieved through precise stoichiometric adjustments. By maintaining the molar ratio of bromine to pentaerythritol between 1.5 and 2.0, and carefully regulating the sulfur-to-bromine ratio, the process suppresses the formation of dibromo and tetrabromo contaminants. The acetic acid solvent plays a dual role here, serving as both the reaction medium and a participant in the equilibrium that drives the esterification forward. The subsequent addition of alkanes like n-heptane or n-hexane induces a phase separation that further purifies the product by precipitating the target molecule while leaving soluble impurities in the mother liquor. This mechanistic elegance ensures that the final crystalline product meets the stringent purity requirements of >99%, making it suitable for direct use in downstream API synthesis without extensive reprocessing.
How to Synthesize Tribromoneopentanol Acetate Efficiently
Executing this synthesis requires careful attention to thermal management and reagent addition rates to maintain the delicate balance of the in-situ generation process. The protocol begins with the charging of pentaerythritol, sulfur powder, and acetic acid into a reactor equipped with efficient stirring and temperature control. Liquid bromine is then added dropwise, a step that must be monitored closely to prevent localized overheating, although the sulfur mediation helps dampen violent exotherms. Once the bromination is complete, typically indicated by the consumption of bromine color, the mixture is heated to facilitate the reaction completion before volatile components are recovered. The residue is then treated with an alkane and acetic anhydride to drive the esterification to completion, followed by a quench with water to induce crystallization. For the precise operational parameters and standardized workup procedures, please refer to the detailed guide below.
- Mix pentaerythritol, bromine, sulfur powder, and acetic acid solvent, then heat to initiate the reaction where sulfur reduces bromine to generate hydrogen bromide in situ.
- Upon completion, distill under reduced pressure to recover acetic acid and hydrogen bromide, then add alkane and acetic anhydride to the residue for esterification.
- Add water to the reaction mixture, separate the organic layer, cool to crystallize, and filter to obtain high-purity tribromoneopentanol acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic route offers profound advantages that translate directly into bottom-line value and supply chain resilience. The elimination of external hydrogen bromide gas cylinders removes a major logistical bottleneck and safety liability, simplifying the storage and handling requirements for the manufacturing facility. This reduction in hazard classification can lead to lower insurance premiums and reduced regulatory compliance burdens, which are often hidden costs in chemical production. Furthermore, the use of commodity chemicals like sulfur powder and acetic acid ensures that the raw material supply is robust and less susceptible to the volatility seen with specialized reagents. The ability to recover and recycle acetic acid from the reaction distillate further enhances the economic viability, creating a closed-loop system that minimizes waste disposal costs and environmental impact.
- Cost Reduction in Manufacturing: The integration of bromination and esterification into a streamlined sequence significantly lowers the operational expenditure associated with multiple unit operations. By avoiding the isolation of intermediate alcohols and the need for separate esterification reactors, the process reduces energy consumption, labor hours, and equipment footprint. The high yield reported in the patent data implies that less raw material is wasted on byproducts, directly improving the cost-per-kilogram of the final active ingredient. Additionally, the simplicity of the workup, which relies on standard crystallization techniques rather than complex chromatography, ensures that production costs remain competitive even at smaller batch sizes.
- Enhanced Supply Chain Reliability: Dependence on hazardous gas supplies can often lead to delivery delays and production stoppages due to transport restrictions or supplier shortages. By generating the necessary reagent in-situ from stable solid and liquid precursors, this method decouples production from these external vulnerabilities. The raw materials involved are widely available in the global chemical market, ensuring a consistent feedstock supply that supports continuous manufacturing campaigns. This reliability is crucial for maintaining the continuity of supply for critical pharmaceutical intermediates, where any disruption can have cascading effects on the downstream drug product availability.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents and conditions that are easily managed in standard glass-lined or stainless steel reactors. The wastewater generated is primarily acidic and can be neutralized with standard alkaline solutions before entering conventional sewage treatment systems, avoiding the need for specialized hazardous waste incineration. The high purity of the product reduces the burden on downstream purification processes, thereby lowering the overall environmental footprint of the manufacturing lifecycle. This alignment with green chemistry principles makes the technology attractive for companies aiming to meet increasingly strict sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific beneficial effects and experimental data presented in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists assessing the quality assurances of potential suppliers.
Q: What is the primary advantage of using sulfur powder in this synthesis?
A: Using sulfur powder as a reducing agent allows for the in-situ generation of hydrogen bromide from elemental bromine. This eliminates the need for handling hazardous HBr gas cylinders, significantly improving operational safety and process controllability while reducing side reactions.
Q: How does this method improve product purity compared to conventional routes?
A: Conventional methods often produce tribromoneopentyl alcohol as the main product with the acetate only as a minor byproduct. This novel route optimizes raw material ratios and reaction conditions to directly target the acetate ester, achieving over 99% purity and minimizing dibromo or tetrabromo impurities.
Q: Is the solvent system suitable for industrial scale-up?
A: Yes, the process utilizes acetic acid as both a solvent and a reactant, which can be recovered and recycled via distillation. The subsequent use of common alkanes like n-heptane or n-hexane for crystallization ensures that the solvent system is cost-effective and compatible with large-scale industrial equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tribromoneopentanol Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN110903188A can be realized in a full-scale industrial setting. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical instrumentation. Our facility is equipped to handle the specific safety requirements of bromination chemistry, providing a secure and compliant environment for the manufacture of sensitive intermediates like tribromoneopentanol acetate.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior synthetic route. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us help you secure a stable, high-quality supply of this critical intermediate for your pharmaceutical or specialty chemical applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
