Advanced Synthesis of 4,5-Ethylene Carbazole Derivatives for Commercial Optoelectronic Applications
Advanced Synthesis of 4,5-Ethylene Carbazole Derivatives for Commercial Optoelectronic Applications
Introduction to Patent CN101891670A and Optoelectronic Innovation
The landscape of organic optoelectronics is continuously evolving, driven by the demand for materials with superior charge transport and luminescent properties, as exemplified by the technological breakthroughs detailed in patent CN101891670A. This specific intellectual property introduces a robust methodology for synthesizing 4,5-ethylene-2,7-disubstituted carbazole derivatives, which serve as critical building blocks for next-generation organic light-emitting diodes and photovoltaic cells. The core innovation lies in the strategic functionalization of the carbazole scaffold at the 2 and 7 positions, allowing for precise tuning of electronic properties while maintaining the structural rigidity provided by the 4,5-ethylene bridge. 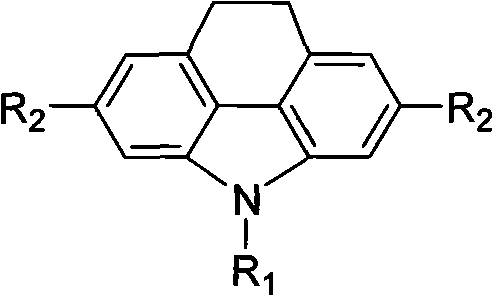 For R&D Directors focusing on material performance, this structural motif offers a unique balance of solubility and conjugation length, essential for achieving high quantum efficiency in device applications. The patent outlines a versatile pathway that accommodates various substituents including halogens and organometallic groups, thereby expanding the chemical space available for material scientists to explore novel conjugated systems.
For R&D Directors focusing on material performance, this structural motif offers a unique balance of solubility and conjugation length, essential for achieving high quantum efficiency in device applications. The patent outlines a versatile pathway that accommodates various substituents including halogens and organometallic groups, thereby expanding the chemical space available for material scientists to explore novel conjugated systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalized carbazoles often suffer from significant drawbacks related to regioselectivity and the harshness of reaction conditions required to introduce substituents at the desired positions. Conventional methods frequently rely on direct halogenation of the carbazole core, which can lead to mixtures of isomers that are difficult and costly to separate, ultimately compromising the purity required for high-performance electronic devices. Furthermore, many existing processes utilize expensive transition metal catalysts in the early stages of synthesis, introducing potential contamination issues that necessitate rigorous and yield-reducing purification steps later in the workflow. These inefficiencies create bottlenecks in the supply chain, increasing lead times for high-purity OLED intermediates and driving up the overall cost of goods sold for manufacturers. The lack of a unified strategy to simultaneously address the 4,5-bridge and the 2,7-substitution patterns has historically limited the structural diversity of available carbazole-based polymers.
The Novel Approach
In contrast, the methodology presented in the patent data proposes a streamlined sequence that begins with the nitration of 2,7-dihalo-9,10-dihydrophenanthrene precursors, effectively bypassing the regioselectivity issues associated with direct carbazole functionalization. This novel approach leverages the inherent reactivity of the dihydrophenanthrene system to install the nitro group selectively, which subsequently serves as the handle for ring closure to form the carbazole nitrogen heterocycle. By deferring the formation of the carbazole core until after the halogen substituents are already in place, the process ensures that the critical 2,7-positions are occupied exactly as designed without the formation of unwanted isomers. This strategic reversal of the synthetic order not only simplifies the purification process but also enhances the overall yield of the target intermediate, providing a more reliable foundation for downstream polymerization reactions. The ability to access diverse derivatives through this common intermediate represents a significant advancement in cost reduction in organic semiconductor manufacturing.
Mechanistic Insights into Nitration and Cyclization Pathways
The chemical transformation begins with an electrophilic aromatic substitution where the nitronium ion attacks the electron-rich dihydrophenanthrene ring system under carefully controlled thermal conditions.  The use of a fuming nitric acid and acetic acid mixture allows for precise modulation of the nitrating strength, ensuring that mono-nitration occurs predominantly at the 4-position relative to the existing halogen substituents. Following the isolation of the nitro-intermediate, the cyclization step employs triphenylphosphine in N-methylpyrrolidone at elevated temperatures to effect the reduction of the nitro group and subsequent intramolecular C-N bond formation. This mechanism avoids the use of hydrogen gas and heterogeneous catalysts, which are often problematic in large-scale operations due to safety concerns and catalyst deactivation issues. The result is a clean conversion to the 4,5-ethylene-2,7-dihalocarbazole core, which retains the halogen handles necessary for further cross-coupling reactions without requiring protective group strategies.
The use of a fuming nitric acid and acetic acid mixture allows for precise modulation of the nitrating strength, ensuring that mono-nitration occurs predominantly at the 4-position relative to the existing halogen substituents. Following the isolation of the nitro-intermediate, the cyclization step employs triphenylphosphine in N-methylpyrrolidone at elevated temperatures to effect the reduction of the nitro group and subsequent intramolecular C-N bond formation. This mechanism avoids the use of hydrogen gas and heterogeneous catalysts, which are often problematic in large-scale operations due to safety concerns and catalyst deactivation issues. The result is a clean conversion to the 4,5-ethylene-2,7-dihalocarbazole core, which retains the halogen handles necessary for further cross-coupling reactions without requiring protective group strategies.
Impurity control is inherently built into this mechanism through the crystallization properties of the intermediate nitro compounds and the final carbazole products. The distinct polarity differences between the starting dihydrophenanthrene, the nitro-intermediate, and the final carbazole allow for effective separation via standard silica gel chromatography or recrystallization techniques. This high level of purity is critical for R&D Directors who must ensure that trace impurities do not act as charge traps in the final electronic device, which would degrade performance and longevity. Furthermore, the subsequent N-alkylation step, utilizing sodium hydride and alkyl halides, proceeds with high fidelity to install solubilizing chains that improve the processability of the final polymers. The robustness of this mechanistic pathway ensures that the commercial scale-up of complex conjugated polymers can be achieved with consistent quality batch after batch, minimizing the risk of production failures.
How to Synthesize 4,5-Ethylene-2,7-disubstituted Carbazole Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of anhydrous conditions and inert atmosphere handling during the organometallic functionalization steps. Operators must strictly adhere to the temperature profiles specified for the lithiation and subsequent quenching with trialkyltin chlorides or boronic esters to prevent side reactions that could compromise the integrity of the reactive intermediates. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that have been optimized to maximize recovery and minimize waste generation.
- Preparation of 2,7-dihalo-4-nitro-9,10-dihydrophenanthrene via controlled nitration.
- Cyclization using triphenylphosphine in N-methylpyrrolidone to form the carbazole core.
- N-alkylation and subsequent functionalization with tin or boron groups for polymerization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial cost savings by eliminating the need for precious metal catalysts in the core ring-forming step, which is traditionally one of the most expensive components of fine chemical manufacturing. The reliance on commodity chemicals such as triphenylphosphine and common solvents like dichloromethane and tetrahydrofuran ensures that raw material availability remains high and price volatility is minimized. 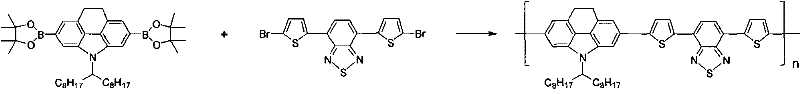 This stability in raw material sourcing translates directly into enhanced supply chain reliability, allowing manufacturers to plan production schedules with greater confidence and reduced risk of disruption. Additionally, the simplicity of the workup procedures, which primarily involve aqueous washes and solvent extraction, reduces the operational complexity and energy consumption associated with purification, further driving down the overall manufacturing costs. These factors combine to create a highly competitive cost structure for the production of high-purity carbazole derivatives.
This stability in raw material sourcing translates directly into enhanced supply chain reliability, allowing manufacturers to plan production schedules with greater confidence and reduced risk of disruption. Additionally, the simplicity of the workup procedures, which primarily involve aqueous washes and solvent extraction, reduces the operational complexity and energy consumption associated with purification, further driving down the overall manufacturing costs. These factors combine to create a highly competitive cost structure for the production of high-purity carbazole derivatives.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the cyclization step removes a significant cost driver and simplifies the removal of metal residues, which is a costly and time-consuming process in traditional methods. By utilizing organic reagents like triphenylphosphine for the reduction and cyclization, the process avoids the need for specialized equipment required for high-pressure hydrogenation or handling pyrophoric catalysts. This simplification of the reaction infrastructure allows for broader manufacturing capabilities and reduces the capital expenditure required to bring new capacity online. Consequently, the overall cost of goods is significantly reduced, making these advanced materials more accessible for widespread commercial application in the optoelectronics industry.
- Enhanced Supply Chain Reliability: The starting materials, specifically the 2,7-dihalo-9,10-dihydrophenanthrene derivatives, are synthesized from readily available feedstocks, ensuring a stable and continuous supply chain for the production of these critical intermediates. The robustness of the synthetic pathway means that production is less susceptible to fluctuations in the availability of specialized reagents, which often plague more exotic synthetic routes. This reliability is crucial for Supply Chain Heads who need to guarantee delivery timelines to downstream device manufacturers without the risk of unexpected delays. The ability to source materials consistently supports long-term planning and strengthens the partnership between chemical suppliers and technology developers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions and solvents that are compatible with standard large-scale chemical manufacturing equipment and safety protocols. The waste streams generated are primarily organic solvents and aqueous salts, which can be managed through established recovery and treatment systems, ensuring compliance with environmental regulations. The high atom economy of the cyclization step minimizes the generation of chemical waste, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process. This focus on sustainability not only meets regulatory requirements but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbazole derivatives, based on the specific details provided in the patent documentation. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating these materials into their product development pipelines.
Q: What are the key advantages of this nitration-cyclization route?
A: This route avoids harsh metal catalysts in the core formation step, significantly simplifying purification and reducing heavy metal contamination risks in the final optoelectronic material.
Q: Can this intermediate be used for large-scale polymer production?
A: Yes, the presence of reactive halogen or boron/tin groups at the 2,7-positions allows for efficient Suzuki or Stille coupling, facilitating the commercial scale-up of complex conjugated polymers.
Q: How is impurity control managed during the nitration step?
A: Strict temperature control during the dropwise addition of fuming nitric acid ensures selective mono-nitration, preventing over-nitration and ensuring high purity specifications for the downstream cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Ethylene-2,7-disubstituted carbazole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver high-quality intermediates for the global market. Our technical team is adept at optimizing the synthesis of complex molecules like 4,5-ethylene-2,7-disubstituted carbazole, ensuring that stringent purity specifications are met through our rigorous QC labs and advanced analytical capabilities. We understand the critical nature of material consistency in the optoelectronic industry and are committed to providing a supply partner that can grow with your demand. Our facility is equipped to handle the specific safety and handling requirements of organometallic intermediates, ensuring a secure and reliable source for your R&D and production needs.
We invite you to engage with our technical procurement team to discuss how we can support your specific material requirements and help you achieve your development goals. Request a Customized Cost-Saving Analysis to understand how our optimized processes can benefit your bottom line, and ask for specific COA data and route feasibility assessments to verify our capabilities. We are ready to collaborate on custom synthesis projects that require the high level of expertise and flexibility that only a dedicated CDMO partner can provide. Let us help you accelerate your time to market with reliable supply and superior quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
