Advanced Synthesis of 2-Methyl Acetoacetate for High-Purity Pirimicarb Production
The global demand for high-efficiency aphicides continues to drive innovation in the synthesis of key agrochemical intermediates, specifically focusing on the production of pirimicarb. Patent CN111995522A introduces a groundbreaking synthetic method for 2-methyl acetoacetate, a critical precursor in the manufacturing of this selective insecticide. This technical disclosure addresses long-standing challenges in the industry by replacing hazardous alkylation reagents with a safer addition-hydrogenation sequence. For R&D Directors and Procurement Managers seeking a reliable agrochemical intermediate supplier, this patent represents a significant shift towards greener and more efficient manufacturing protocols. The method leverages the reaction between methyl acrylate and acetyl chloride, facilitated by tertiary amines, to construct the carbon skeleton with high precision. This approach not only mitigates the risks associated with traditional methylating agents but also streamlines the purification process, ensuring a consistent supply of high-purity materials essential for downstream formulation. By adopting this novel pathway, manufacturers can achieve substantial improvements in yield and environmental compliance, positioning themselves as leaders in sustainable agrochemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl acetoacetate has relied heavily on the alkylation of methyl acetoacetate using dimethyl sulfate in the presence of strong bases. This conventional route suffers from inherent thermodynamic limitations, as the reaction establishes an equilibrium that is difficult to drive to completion without generating significant impurities. Specifically, the process tends to produce a mixture containing the desired mono-methylated product alongside the over-alkylated byproduct, 2,2-dimethyl-methyl acetoacetate. The separation of these components is notoriously difficult due to their closely matched boiling points, necessitating high-reflux rectification columns that consume excessive energy and result in poor overall recovery rates. Furthermore, the use of dimethyl sulfate introduces severe safety and environmental liabilities, requiring specialized handling equipment and costly waste treatment protocols to manage its high toxicity. These factors collectively inflate the cost of goods sold and create bottlenecks in the supply chain, making the traditional method increasingly untenable for modern, large-scale commercial operations.
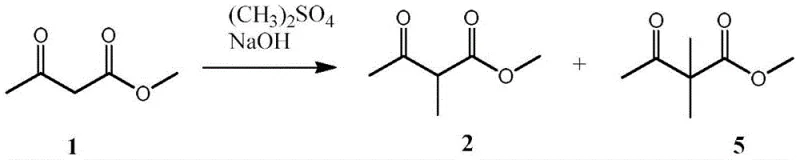
The Novel Approach
In stark contrast to the problematic alkylation routes, the novel approach detailed in the patent utilizes a constructive addition reaction followed by catalytic hydrogenation. This strategy fundamentally bypasses the equilibrium issues of alkylation by building the methyl group onto the acrylic backbone rather than substituting a hydrogen on an existing ketone. The initial step involves the reaction of methyl acrylate with acetyl chloride, mediated by a tertiary amine base and stabilized by a polymerization inhibitor to prevent unwanted chain growth. This addition step is highly selective and avoids the formation of the troublesome 2,2-dimethyl byproduct entirely. The subsequent hydrogenation of the resulting 2-acetyl-methyl acrylate intermediate is a robust and well-understood transformation that proceeds with high conversion rates. By decoupling the carbon skeleton construction from the methylation step, this method offers a cleaner reaction profile, significantly reducing the burden on downstream purification units and enabling a more straightforward path to high-purity final products suitable for sensitive agrochemical applications.
Mechanistic Insights into Acetyl Chloride Addition and Hydrogenation
The core of this synthetic innovation lies in the precise control of the addition reaction between methyl acrylate and acetyl chloride. Mechanistically, the tertiary amine acts as a base to facilitate the formation of an enolate or activated complex that attacks the acetyl chloride, while simultaneously neutralizing the hydrochloric acid byproduct. The inclusion of a polymerization inhibitor, such as hydroquinone or 4-methoxyphenol, is critical to suppress the radical polymerization of the methyl acrylate double bond, ensuring that the reaction proceeds via the desired addition pathway rather than forming polymeric tars. This careful balance of reagents allows for the selective formation of 2-acetyl-methyl acrylate with minimal side reactions. The subsequent hydrogenation step utilizes a heterogeneous catalyst, typically palladium on carbon or platinum on carbon, to reduce the exocyclic double bond. This reduction is highly chemoselective, leaving the ester and ketone functionalities intact while saturating the carbon-carbon double bond to yield the target 2-methyl acetoacetate. The mechanistic clarity of this two-step sequence provides R&D teams with a robust framework for process optimization and scale-up.
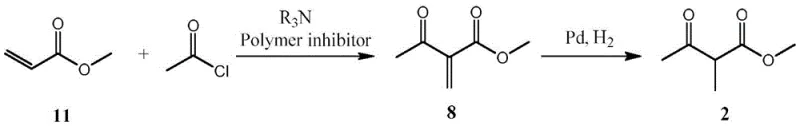
Impurity control is another critical aspect where this new mechanism offers distinct advantages over prior art. In traditional routes, impurities often arise from over-alkylation or the reaction of starting materials with downstream reagents like N,N-dimethylguanidine, leading to complex impurity profiles that are difficult to purge. The new route minimizes these risks by ensuring that the intermediate 2-acetyl-methyl acrylate is formed with high specificity before the reduction step. The hydrogenation catalyst can be finely tuned to prevent over-reduction or hydrogenolysis of the ester group, further enhancing the purity of the final product. By eliminating the formation of the 2,2-dimethyl byproduct at the source, the process reduces the load on distillation columns and minimizes the risk of carryover impurities into the final pirimicarb synthesis. This level of control over the impurity spectrum is essential for meeting the stringent quality specifications required by global agrochemical regulators and ensures consistent batch-to-batch reproducibility in commercial manufacturing.
How to Synthesize 2-Methyl Acetoacetate Efficiently
The implementation of this synthetic route requires careful attention to reaction conditions and reagent ratios to maximize yield and safety. The process begins with the dissolution of methyl acrylate, acetyl chloride, and a polymerization inhibitor in a suitable solvent such as diethyl ether, toluene, or tetrahydrofuran under an inert atmosphere. A tertiary amine, such as triethylamine or DIPEA, is then added dropwise to control the exotherm and drive the addition reaction to completion. Following the reaction, the mixture is quenched with water, and the organic phase is separated, washed, and distilled to isolate the 2-acetyl-methyl acrylate intermediate. This intermediate is then subjected to hydrogenation in an alcohol solvent using a precious metal catalyst. The detailed standardized synthesis steps, including specific molar ratios, temperature ranges, and workup procedures, are outlined in the structured guide below to ensure reproducible results in your laboratory or pilot plant.
- Conduct an addition reaction between methyl acrylate and acetyl chloride in the presence of a tertiary amine base and a polymerization inhibitor within an ether or aromatic solvent.
- Quench the reaction mixture with water, perform liquid separation, and purify the intermediate 2-acetyl-methyl acrylate through washing and distillation.
- Subject the purified intermediate to catalytic hydrogenation using palladium on carbon or platinum on carbon in an alcohol solvent to yield the final 2-methyl acetoacetate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic method offers compelling economic and operational benefits. The elimination of dimethyl sulfate removes a significant cost center associated with hazardous material handling, storage, and disposal, leading to substantial cost savings in manufacturing overhead. Additionally, the improved yield and purity reduce the volume of raw materials required per unit of output, further enhancing cost efficiency. The use of readily available commodity chemicals like methyl acrylate and acetyl chloride ensures a stable supply chain, reducing the risk of disruptions caused by the scarcity of specialized reagents. The simplified purification process also translates to shorter production cycles and increased throughput, allowing manufacturers to respond more agilely to market demand fluctuations. These factors combine to create a more resilient and cost-effective supply chain for high-purity agrochemical intermediates.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive and energy-intensive high-reflux rectification required to separate close-boiling byproducts in traditional methods. By avoiding the formation of the 2,2-dimethyl impurity, the yield loss during purification is drastically minimized, directly lowering the cost per kilogram of the final intermediate. Furthermore, the replacement of toxic dimethyl sulfate with safer reagents reduces the regulatory compliance costs and insurance premiums associated with hazardous chemical processing. These cumulative efficiencies result in a significantly more competitive cost structure for the production of pirimicarb intermediates without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, including methyl acrylate and acetyl chloride, are produced on a massive global scale for various industrial applications, ensuring their ready availability and price stability. Unlike specialized alkylating agents that may be subject to supply constraints or strict regulatory controls, these commodity chemicals can be sourced from multiple suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream agrochemical formulators.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing standard reaction types such as addition and hydrogenation that are easily transferred from laboratory to commercial scale. The absence of highly toxic reagents simplifies the environmental permitting process and reduces the burden on waste treatment facilities. The use of common solvents like ethanol or toluene allows for efficient recovery and recycling, minimizing waste generation and aligning with green chemistry principles. This environmental compatibility not only reduces operational risks but also enhances the corporate sustainability profile of the manufacturer, which is increasingly important for securing contracts with major global agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method. These answers are derived directly from the patent data and are intended to provide clarity on the process advantages and operational requirements. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production. The insights provided here cover aspects of purity, safety, and scalability, offering a comprehensive overview of the technology's value proposition for the agrochemical industry.
Q: How does this new synthesis method improve purity compared to traditional alkylation?
A: Traditional alkylation using dimethyl sulfate creates a difficult-to-separate equilibrium mixture containing significant amounts of 2,2-dimethyl-methyl acetoacetate byproduct. The new addition-hydrogenation route avoids this specific alkylation side reaction entirely, resulting in a much cleaner impurity profile and higher final purity without complex rectification.
Q: What are the safety advantages of avoiding dimethyl sulfate in this process?
A: Dimethyl sulfate is a highly toxic and carcinogenic alkylating agent that requires stringent safety protocols and specialized waste treatment. By replacing it with acetyl chloride and methyl acrylate, the process significantly reduces occupational health hazards and simplifies environmental compliance for large-scale manufacturing facilities.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the reaction conditions utilize standard industrial unit operations such as addition reactors and hydrogenation kettles. The use of readily available raw materials and common solvents like ethanol or toluene ensures that the process is highly scalable and compatible with existing fine chemical infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl Acetoacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the production of high-value agrochemical intermediates. Our team of expert chemists has extensively analyzed the technology disclosed in CN111995522A and is fully prepared to implement this advanced synthesis for your supply chain needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-methyl acetoacetate meets the highest industry standards. By leveraging our CDMO expertise, you can accelerate your time-to-market while maintaining full control over quality and cost.
We invite you to collaborate with us to optimize your supply chain for pirimicarb production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate the tangible benefits of this novel synthetic method. Let us partner with you to drive innovation and efficiency in your agrochemical manufacturing operations, ensuring a reliable supply of high-purity intermediates for your global markets.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
