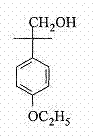
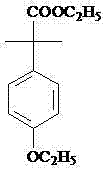
Advanced Synthesis of 2-(4-Ethoxyl Phenyl)-2-Methylpropanol for Scalable Agrochemical Production
The global demand for high-performance pyrethroid insecticides continues to drive innovation in the synthesis of key intermediates, specifically focusing on safety, yield, and scalability. Patent CN103467254A introduces a transformative preparation method for 2-(4-ethoxyl phenyl)-2-methylpropanol, a critical building block for Etofenprox, by utilizing a modified borohydride reduction system. This technical breakthrough addresses the longstanding limitations of traditional reduction methods, offering a pathway that is not only chemically robust but also commercially viable for large-scale manufacturing. As a reliable agrochemical intermediate supplier, understanding the nuances of this patent is essential for R&D teams aiming to optimize their supply chains for next-generation pest control agents. The method leverages a synergistic interaction between alkali metal borohydrides and Lewis acidic metal salts to activate the reduction of esters, a transformation that is typically challenging under mild conditions. This report provides a deep dive into the mechanistic advantages and commercial implications of adopting this technology for the production of high-purity etofenprox intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
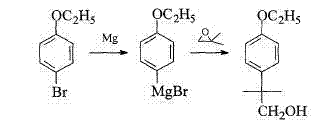
Historically, the synthesis of 2-(4-ethoxyl phenyl)-2-methylpropanol has relied on several operational paths that present significant hurdles for industrial application. One common route involves the use of lithium aluminum hydride (LiAlH4) for the reduction of the corresponding ester; however, this reagent is notoriously dangerous, requiring strictly anhydrous conditions and posing severe safety risks during quenching and scale-up. Another traditional approach utilizes Grignard reactions, such as the reaction of bromoethyl phenyl ether with magnesium followed by epoxide ring opening, which suffers from high synthetic difficulty and stringent requirements for moisture exclusion. Furthermore, alternative routes starting from p-tert-butylphenol involve complex chlorination and hydrolysis steps that generate difficult-to-separate byproducts and require high-pressure reactors, leading to increased operational costs and safety concerns. These conventional methods often result in lower overall yields and generate substantial hazardous waste, making them less attractive for modern, environmentally conscious manufacturing facilities seeking cost reduction in pyrethroid manufacturing.
The Novel Approach
The novel approach detailed in the patent data overcomes these deficiencies by employing a hydroborate and metal-salt mixed system, specifically utilizing potassium borohydride or sodium borohydride in the presence of salts like lithium chloride. This method allows for the reduction of 2-(4-ethoxyl phenyl)-2-methyl ethyl propanoate under significantly milder conditions, typically between 30°C and 85°C, using common alcoholic solvents like ethanol or methanol. Unlike the violent reactivity of lithium aluminum hydride, this system is stable and manageable, allowing for safer handling and simpler workup procedures involving standard acid quenching and solvent recovery. The elimination of hazardous reagents and the simplification of the reaction protocol drastically reduce the barrier to entry for commercial scale-up of complex alcohol intermediates. By shifting to this chemistry, manufacturers can achieve a total reaction yield above 80% with product content reaching 98%, demonstrating a clear superiority in both efficiency and product quality compared to legacy technologies.
Mechanistic Insights into Borohydride-Metal Salt Activated Reduction
The core innovation of this synthesis lies in the activation of the borohydride anion by the metal salt, which enhances its reducing power to effectively convert esters into primary alcohols. Typically, sodium or potassium borohydride alone is insufficient for reducing esters at a practical rate, but the addition of metal salts such as lithium chloride, aluminum chloride, or zinc chloride creates a more potent reducing species in situ. This activation likely involves the coordination of the metal cation with the carbonyl oxygen of the ester substrate, increasing its electrophilicity and facilitating the hydride transfer from the borohydride. The reaction proceeds through a tetrahedral intermediate which eventually collapses to release the alkoxide, followed by protonation during the acidic workup to yield the final alcohol. This mechanism allows the reaction to proceed in protic solvents like ethanol, which would otherwise decompose unactivated borohydride rapidly, thereby enabling the use of cheaper and greener solvent systems. The precise control of the molar ratio between the hydroborate, metal salt, and substrate is critical, with optimal results observed at ratios around 2.0:1.2:1, ensuring complete conversion while minimizing reagent waste.
Impurity control is another critical aspect where this mechanism offers distinct advantages over traditional Grignard or LiAlH4 routes. In Grignard reactions, side reactions such as Wurtz coupling or enolization can lead to complex impurity profiles that are difficult to remove, whereas the borohydride reduction is highly chemoselective for the ester group. The mild reaction conditions prevent the degradation of the sensitive ether linkage on the phenyl ring, which can be susceptible to cleavage under harsh acidic or basic conditions found in other methods. Furthermore, the use of ethanol as a solvent facilitates the dissolution of polar intermediates and byproducts, allowing for a cleaner reaction profile that simplifies downstream purification. The final workup involves a straightforward pH adjustment to neutral, followed by extraction with organic solvents like dichloromethane or ethyl acetate, which effectively separates the product from inorganic salts. This streamlined purification process contributes to the high purity of 98% reported in the patent, ensuring that the intermediate meets the stringent specifications required for the synthesis of active pharmaceutical or agrochemical ingredients.
How to Synthesize 2-(4-Ethoxyl Phenyl)-2-Methylpropanol Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reducing mixture and the control of reaction temperature to maximize yield and safety. The process begins with dissolving the ester substrate in a suitable alcoholic solvent, followed by the sequential or simultaneous addition of the borohydride and metal salt components under controlled thermal conditions. Detailed standard operating procedures regarding the specific addition rates, stirring speeds, and quenching protocols are essential to ensure reproducibility and safety on a manufacturing scale. The following guide outlines the critical steps derived from the patent examples to assist technical teams in evaluating the feasibility of this pathway for their production lines.
- Dissolve the ester substrate in an alcoholic solvent such as ethanol or methanol, optionally mixed with ethers like THF, and warm the mixture to a temperature range of 30-85°C.
- Add a mixed reducing system comprising potassium borohydride or sodium borohydride alongside a metal salt catalyst such as lithium chloride or aluminum chloride in specific molar ratios.
- Maintain the reaction under insulation for 3-8 hours, followed by acid quenching, solvent recovery, organic extraction, and vacuum concentration to isolate the high-purity alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this borohydride-based reduction route presents a compelling value proposition centered around cost stability and operational reliability. By replacing expensive and hazardous reagents like lithium aluminum hydride with commodity chemicals such as potassium borohydride and lithium chloride, the raw material costs are significantly reduced without compromising reaction efficiency. The ability to use ethanol, a widely available and inexpensive solvent, further drives down the variable costs associated with solvent purchase and recovery, contributing to substantial cost savings in the overall manufacturing budget. Moreover, the simplified workup procedure reduces the consumption of utilities and labor hours, enhancing the overall economic viability of the process for high-volume production. These factors combine to create a more resilient supply chain that is less susceptible to price volatility in specialty chemical markets.
- Cost Reduction in Manufacturing: The elimination of high-cost reducing agents and the shift to ambient pressure operations drastically lower the capital and operational expenditures required for production. The removal of expensive heavy metal catalysts or complex organometallic reagents means that the process does not require specialized equipment for handling pyrophoric materials, thereby reducing maintenance and safety compliance costs. Additionally, the high yield of over 80% ensures that raw material utilization is optimized, minimizing waste disposal fees and maximizing the output per batch. This economic efficiency allows manufacturers to offer more competitive pricing for the final agrochemical intermediate while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including potassium borohydride and lithium chloride, are commodity chemicals with robust global supply chains, reducing the risk of production stoppages due to material shortages. Unlike specialized catalysts that may have long lead times or single-source dependencies, these materials can be sourced from multiple vendors, ensuring continuity of supply even during market disruptions. The mild reaction conditions also reduce the wear and tear on reactor vessels and ancillary equipment, leading to higher asset availability and reduced downtime for maintenance. This reliability is crucial for meeting the just-in-time delivery requirements of downstream pesticide formulators and maintaining strong customer relationships.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent examples which describe procedures easily adaptable from laboratory to pilot and commercial scales. The use of less hazardous chemicals and the generation of fewer toxic byproducts align with increasingly strict environmental regulations, reducing the burden on waste treatment facilities and lowering the risk of regulatory penalties. The ability to recover and recycle the alcoholic solvent further enhances the sustainability profile of the manufacturing process, appealing to environmentally conscious stakeholders. This combination of scalability and compliance ensures that the production facility can grow with market demand without facing significant regulatory or technical bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, based on the specific data points and advantages outlined in the patent documentation. These answers are designed to provide clarity for decision-makers evaluating the transition to this new manufacturing protocol. Understanding these details is key to assessing the risk and reward profile of adopting this technology for your specific production needs.
Q: Why is the borohydride-metal salt system preferred over lithium aluminum hydride for this intermediate?
A: The borohydride-metal salt system offers significantly improved safety profiles and operational simplicity compared to lithium aluminum hydride, which poses severe handling risks and is unsuitable for large-scale industrial production due to its violent reactivity with protic solvents.
Q: What is the expected purity and yield for the 2-(4-ethoxyl phenyl)-2-methylpropanol produced via this method?
A: According to patent data, this optimized reduction pathway consistently achieves a total reaction yield exceeding 80%, with the final product content reaching up to 98%, ensuring high quality for downstream pesticide synthesis.
Q: How does this synthesis route impact the environmental compliance of agrochemical manufacturing?
A: By eliminating the need for hazardous reagents and simplifying the workup process to standard solvent recovery and extraction, this method reduces toxic waste generation and aligns with stricter environmental regulations governing modern chemical production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Ethoxyl Phenyl)-2-Methylpropanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final agrochemical products. Our team of expert chemists has extensively analyzed the pathway described in CN103467254A and possesses the technical capability to execute this reduction with precision and consistency. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without sacrificing quality. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of 2-(4-ethoxyl phenyl)-2-methylpropanol meets the highest industry standards for impurity profiles and physical properties.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and logistical constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your production of Etofenprox and related pyrethroids. Together, we can build a more sustainable and profitable future for the agrochemical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
