Advanced Purification Technology for Aripiprazole Intermediates Enabling Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies to ensure the highest purity standards for active pharmaceutical ingredients and their precursors. Patent CN101768113A introduces a groundbreaking purification technique for 7-(4-chlorobutoxy)-3,4-dihydro-2(1H)-quinolinone, commonly referred to as 7-CBQ, which serves as a critical intermediate in the synthesis of the atypical antipsychotic agent Aripiprazole. This innovation addresses a persistent challenge in the manufacturing sector where traditional purification methods often struggle to remove specific dimeric impurities known as BQB without sacrificing overall yield. By leveraging a specialized adsorption mechanism using aluminum oxide, this technology enables producers to achieve impurity levels below 0.5 percent while maintaining operational simplicity. For R&D directors and procurement specialists, understanding this technical leap is essential for optimizing supply chains and ensuring the consistent quality required for regulatory compliance in global markets.
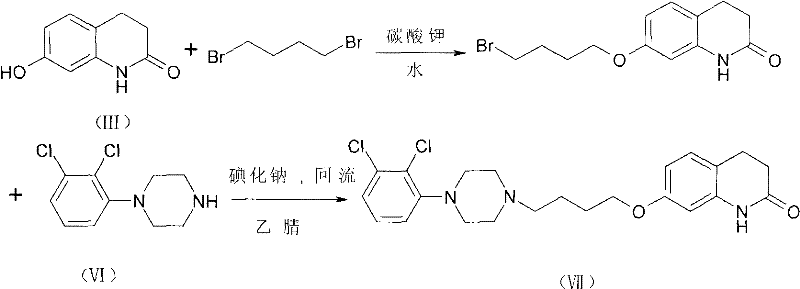
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of 7-CBQ has been fraught with significant technical and economic inefficiencies that hinder large-scale production capabilities. Prior art methods, such as those described in US20060079689A1, often rely on recrystallization techniques that fail to adequately reduce the BQB impurity, leaving levels around 1.5 percent which are unacceptable for final drug substance quality. Alternative approaches involving silica gel treatment have demonstrated poor economic viability, with documented yields dropping drastically to approximately 49.2 percent due to excessive product adsorption and solvent consumption. Furthermore, the reliance on column chromatography, while effective for small-scale laboratory purification, presents a severe bottleneck for industrial scale-up due to high solvent usage, prolonged processing times, and complex equipment requirements. These limitations collectively increase the cost of goods sold and introduce supply chain vulnerabilities that procurement managers must carefully mitigate when sourcing reliable pharmaceutical intermediate supplier partners.
The Novel Approach
The methodology disclosed in CN101768113A represents a paradigm shift by replacing inefficient silica-based adsorption with a highly selective aluminum oxide treatment protocol. This novel approach utilizes a specific weight ratio of aluminum oxide to the starting material, typically ranging from 1:1 to 6:1, to effectively capture the BQB impurity while allowing the target 7-CBQ molecule to pass through into the filtrate. Experimental data from the patent indicates that this method consistently achieves yields exceeding 70 percent, with specific embodiments demonstrating yields up to 92.95 percent while maintaining HPLC purity above 97 percent. The process eliminates the need for complex column chromatography, substituting it with a straightforward stirring and filtration step that is easily adaptable to standard industrial reactors. This simplification not only enhances cost reduction in pharmaceutical intermediate manufacturing but also drastically reduces the environmental footprint by minimizing solvent waste, aligning with modern green chemistry principles and regulatory expectations for sustainable production.
Mechanistic Insights into Aluminum Oxide Adsorption Purification
The efficacy of this purification strategy lies in the differential adsorption properties of aluminum oxide compared to traditional silica gel when interacting with the specific chemical structures involved. The target molecule, 7-CBQ, and the impurity, BQB, possess distinct polarities and structural conformations that influence their interaction with the adsorbent surface. Aluminum oxide, acting as a polar adsorbent, demonstrates a higher affinity for the dimeric BQB impurity structure, effectively retaining it on the solid phase while the monomeric 7-CBQ remains in the solution phase. This selectivity is critically dependent on thermal conditions, as the patent highlights that lower temperatures result in poor adsorption efficiency, whereas temperatures between 60 and 70 degrees Celsius optimize the interaction kinetics. Understanding this mechanistic nuance is vital for process chemists aiming to replicate these results, as it underscores the importance of precise thermal control to balance impurity removal with product recovery rates.
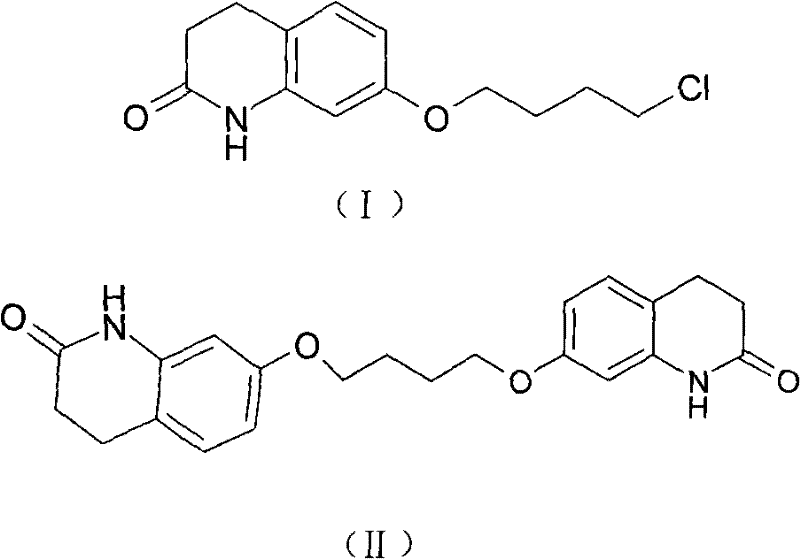
Controlling the impurity profile is paramount for ensuring the safety and efficacy of the final Aripiprazole product, as residual BQB can persist through subsequent synthetic steps. The aluminum oxide treatment effectively reduces the BQB content to less than 0.5 percent, a threshold that is difficult to achieve with recrystallization alone. This deep purification mechanism ensures that the downstream coupling reaction with 1-(2,3-dichlorophenyl)piperazine proceeds with minimal side reactions, thereby safeguarding the quality of the final API. For quality assurance teams, this means a more robust impurity profile that simplifies the validation process and reduces the risk of batch rejection. The ability to consistently produce high-purity intermediates without resorting to labor-intensive chromatographic separation provides a significant competitive advantage in the fast-paced generic pharmaceutical market where speed to market and quality consistency are key differentiators.
How to Synthesize 7-CBQ Efficiently
Implementing this purification technology requires a systematic approach to reaction conditions and workup procedures to maximize the benefits outlined in the patent data. The process begins with the crude synthesis of 7-CBQ, followed by a critical purification stage where the choice of solvent and adsorbent ratio determines the success of the operation. Detailed standard operating procedures regarding specific reagent grades, stirring speeds, and filtration techniques are essential for transferring this laboratory-scale success to commercial production environments. The following guide outlines the fundamental steps required to execute this purification protocol effectively, ensuring that technical teams can replicate the high yields and purity levels reported in the intellectual property documentation.
- Synthesize crude 7-CBQ by reacting 7-HQ with 1-bromo-4-chlorobutane in the presence of potassium carbonate.
- Dissolve the crude product in toluene and treat with aluminum oxide at a controlled temperature range of 60 to 70 degrees Celsius.
- Filter the mixture to remove the adsorbent and recrystallize the filtrate using n-hexane to obtain high-purity 7-CBQ.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this alumina-based purification method offers substantial strategic benefits for organizations managing the supply of complex pharmaceutical intermediates. The elimination of column chromatography translates directly into reduced operational expenditures, as it removes the need for expensive silica gel columns and the vast quantities of solvents associated with elution processes. This simplification of the manufacturing workflow enhances supply chain reliability by reducing the number of unit operations, thereby minimizing the potential for human error and equipment failure during production. For procurement managers, this means a more stable supply of high-purity 7-CBQ with reduced lead times, as the streamlined process allows for faster batch turnover and increased production capacity without significant capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The transition from silica gel to aluminum oxide treatment significantly lowers material costs while improving overall process efficiency. By avoiding the substantial product losses associated with silica-based purification, manufacturers can achieve higher effective yields, which directly impacts the cost per kilogram of the final intermediate. Additionally, the reduction in solvent consumption for chromatography lowers waste disposal costs and aligns with environmental compliance standards, further contributing to long-term financial sustainability. This economic efficiency allows suppliers to offer more competitive pricing structures without compromising on the stringent quality specifications required by regulatory bodies.
- Enhanced Supply Chain Reliability: The robustness of the aluminum oxide purification method ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply chains for downstream API manufacturers. The simplicity of the filtration and recrystallization steps reduces the risk of process deviations that often lead to batch failures in more complex purification schemes. This reliability is particularly valuable for reducing lead time for high-purity pharmaceutical intermediates, as it enables suppliers to respond more agilely to fluctuations in market demand. Procurement teams can rely on this stability to optimize inventory levels and secure long-term supply agreements with confidence.
- Scalability and Environmental Compliance: The method is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing standard equipment that is readily available in most chemical manufacturing facilities. The absence of complex chromatographic columns simplifies the engineering requirements for scaling from pilot plant to multi-ton production, facilitating a smoother technology transfer process. Furthermore, the reduced solvent usage and waste generation support corporate sustainability goals and ensure compliance with increasingly strict environmental regulations. This alignment with green chemistry principles enhances the corporate image of manufacturers and meets the growing demand for environmentally responsible supply chains in the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this purification technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios and assess the potential impact on their supply chain strategies.
Q: How does the alumina treatment compare to silica gel for removing BQB impurity?
A: According to patent CN101768113A, alumina treatment achieves significantly higher yields (over 70%) compared to silica gel methods which may result in yields as low as 49.2%, while effectively reducing the BQB impurity to below 0.5%.
Q: What is the critical temperature range for the purification process?
A: The purification process requires precise temperature control between 60 and 70 degrees Celsius when using toluene as the solvent to ensure optimal adsorption of impurities without compromising the yield of the target intermediate.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the method eliminates the need for complex column chromatography, using simple filtration and recrystallization steps that are highly scalable and cost-effective for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-CBQ Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development and commercialization of pharmaceutical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the alumina-based purification of 7-CBQ can be seamlessly transferred to industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of chemical manufacturing, delivering reliable solutions that support our partners' regulatory filings and market launch timelines.
We invite you to collaborate with us to optimize your supply chain for Aripiprazole intermediates and other complex molecules. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our advanced purification technologies can enhance your operational efficiency. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that you have all the necessary information to make informed sourcing decisions. By partnering with us, you gain access to a wealth of chemical expertise and a dedicated supply chain infrastructure designed to support your long-term growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
