Scalable Synthesis of Aminopyrimidine Kinase Inhibitors via Novel Safe Intermediates
Introduction to Advanced Aminopyrimidine Synthesis
The pharmaceutical industry continuously seeks robust manufacturing routes for potent kinase inhibitors, particularly those targeting mutant epidermal growth factor receptors (EGFR) for non-small cell lung cancer treatment. Patent CN111032650A introduces a groundbreaking methodology for synthesizing N-(5-(4-(4-((dimethylamino)methyl)-3-phenyl-1H-pyrazol-1-yl)pyrimidin-2-ylamino)-4-methoxy-2-morpholinophenyl)acrylamide, a critical aminopyrimidine derivative. This innovation addresses severe limitations in prior art by replacing hazardous reagents with safer, scalable alternatives, ensuring high purity and yield essential for clinical applications. The strategic redesign of the synthetic pathway eliminates the need for dangerous sodium hydride and corrosive iron reductions, marking a significant leap forward in process chemistry.
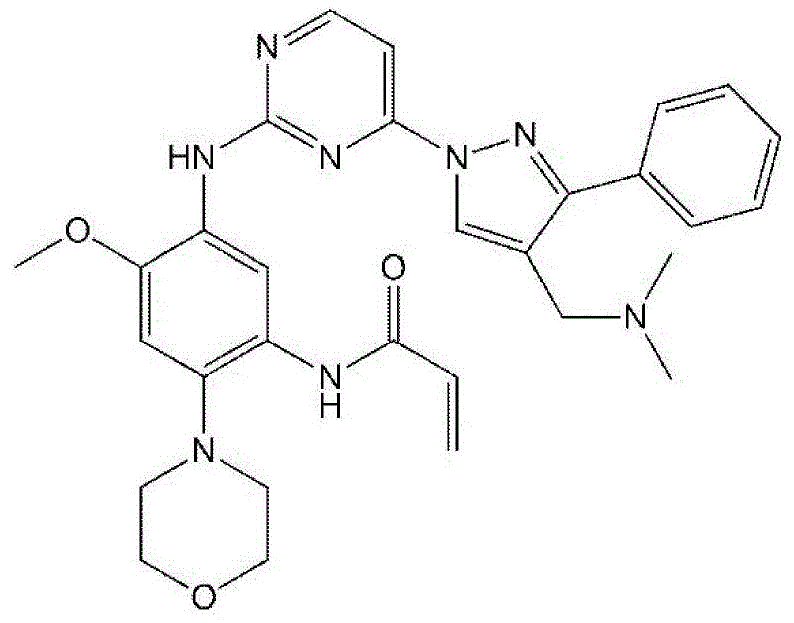
As a reliable pharmaceutical intermediates supplier, understanding the nuances of such structural complexities is vital. The target molecule features a sophisticated architecture combining a morpholine-substituted aniline core, a pyrimidine ring, and a phenyl-pyrazole moiety. Achieving this assembly with high fidelity requires precise control over reaction conditions to prevent degradation of the sensitive acrylamide group. The disclosed technology not only secures the intellectual property around these novel intermediates but also establishes a foundation for cost reduction in oncology drug manufacturing by streamlining purification protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in WO 2016/060443, rely heavily on aggressive reagents that pose substantial risks for industrial mass production. Specifically, the use of sodium hydride (NaH) for nucleophilic substitutions introduces a high probability of fire and explosion, necessitating specialized equipment and rigorous safety protocols that drive up operational costs. Furthermore, the reduction of nitro groups using iron and ammonium chloride creates significant downstream processing challenges, including severe reactor corrosion and the generation of unknown black pitches and degradation products.
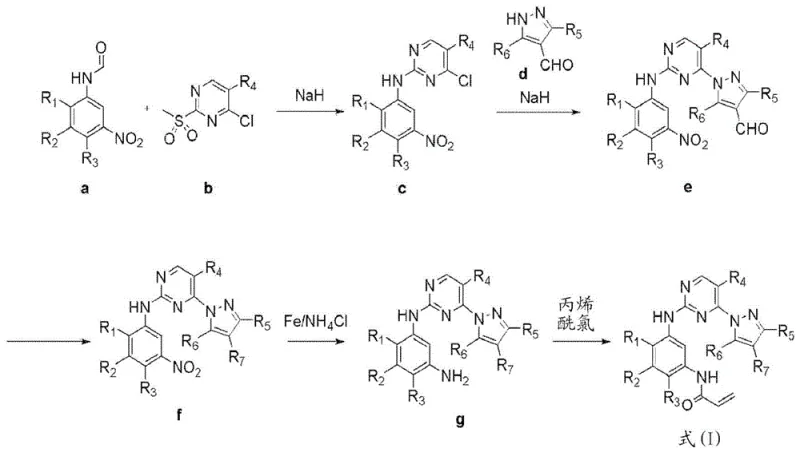
These impurities often mandate the use of column chromatography for purification, a technique that is notoriously difficult to scale and economically unviable for commercial API production. The reliance on acryloyl chloride in the final steps further complicates matters due to its low stability and tendency to generate various degradation products during reaction. Consequently, obtaining the final compound with suitable purity becomes a bottleneck, limiting the ability of supply chain heads to guarantee consistent quality and delivery timelines for high-purity pharmaceutical intermediates.
The Novel Approach
The novel process outlined in CN111032650A fundamentally reengineers the synthetic route to prioritize safety and scalability. By introducing a sequence that utilizes catalytic transfer hydrogenation instead of iron reduction, the method effectively solves the problems of reactor corrosion and contamination. This shift allows for the preparation of key amino intermediates under mild conditions, significantly reducing the formation of tarry byproducts. Additionally, the strategy employs a unique acrylation approach where a chloro-propionamide intermediate is formed first, followed by base-mediated elimination, thereby avoiding the direct handling of unstable acryloyl chloride on sensitive substrates.
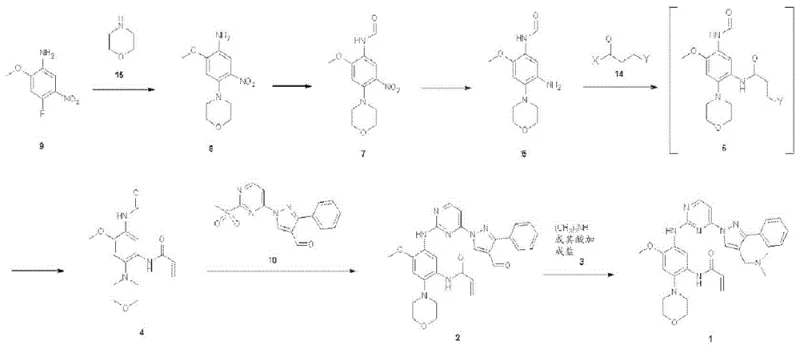
This refined approach enables the use of crystallization for isolation, a unit operation that is far more amenable to commercial scale-up of complex polymer additives and pharmaceutical ingredients than chromatography. The process flow ensures that impurities can be easily removed and controlled at each stage, resulting in a final product with high purity and yield. For procurement managers, this translates to a more reliable supply chain with reduced risk of batch failures, while R&D teams benefit from a cleaner impurity profile that simplifies regulatory filing and quality control assessments.
Mechanistic Insights into Catalytic Transfer Hydrogenation and Elimination
A cornerstone of this improved synthesis is the replacement of traditional metal-acid reduction with catalytic transfer hydrogenation using palladium on carbon and ammonium formate. In this mechanism, ammonium formate serves as the hydrogen donor, decomposing on the palladium surface to generate active hydrogen species in situ. This温和 (mild) reduction environment selectively converts the nitro group of the N-(2-methoxy-4-morpholino-5-nitrophenyl)carboxamide to an amino group without affecting other sensitive functionalities like the methoxy or morpholine groups. The absence of strong acids or metallic iron prevents the acid-catalyzed degradation pathways that typically lead to the formation of intractable black pitches.
Furthermore, the installation of the acrylamide moiety is achieved through a sophisticated two-step sequence involving nucleophilic substitution followed by elimination. Initially, the amino intermediate reacts with 3-chloropropionyl chloride to form a stable chloro-propionamide derivative. Subsequent treatment with a base, such as triethylamine or sodium hydroxide, induces an E2 elimination reaction, removing hydrogen chloride to form the desired double bond. This mechanistic detour provides superior control over the reaction exotherm and minimizes side reactions compared to direct acylation with acryloyl chloride, ensuring the structural integrity of the final aminopyrimidine derivative.
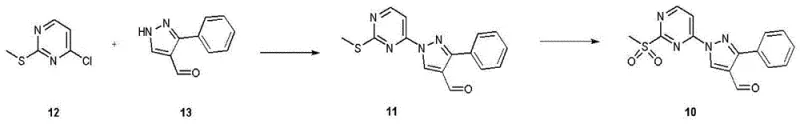
The synthesis of the pyrazole aldehyde coupling partner also demonstrates careful optimization, utilizing oxidation of a methylthio group to a methylsulfonyl group to enhance leaving group ability for the subsequent coupling. This activation strategy ensures high efficiency in the formation of the pyrimidine-pyrazole linkage. By understanding these mechanistic details, technical teams can better appreciate the robustness of the process, which is designed to withstand the rigors of large-scale manufacturing while maintaining stringent purity specifications required for oncology therapeutics.
How to Synthesize N-(5-(4-(4-((dimethylamino)methyl)-3-phenyl-1H-pyrazol-1-yl)pyrimidin-2-ylamino)-4-methoxy-2-morpholinophenyl)acrylamide Efficiently
The efficient synthesis of this complex kinase inhibitor intermediate relies on a convergent strategy that builds the molecule from stable, readily available starting materials. The process begins with the preparation of the aniline core via formylation and subsequent transfer hydrogenation, followed by the careful installation of the acrylamide tail. Parallel to this, the pyrazole-pyrimidine fragment is constructed and activated as a methylsulfonyl derivative. The final convergence involves coupling these two advanced fragments and performing a reductive amination to install the dimethylaminomethyl group. Detailed standardized synthesis steps see the guide below.
- Perform formylation of 2-methoxy-4-morpholino-5-nitroaniline followed by catalytic transfer hydrogenation using Pd/C and ammonium formate to generate the amino-carboxamide intermediate.
- React the amino intermediate with 3-chloropropionyl chloride to form a chloro-propionamide, followed by base-mediated elimination to install the acrylamide moiety safely.
- Couple the acrylamide intermediate with a methylsulfonyl-pyrimidine aldehyde, followed by reductive amination with dimethylamine to yield the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
The transition to this novel synthetic route offers profound commercial advantages that directly address the pain points of modern pharmaceutical supply chains. By eliminating the use of sodium hydride, the process removes a major safety hazard that often incurs high insurance premiums and requires specialized storage facilities. The removal of iron reduction steps not only extends the lifespan of reactor vessels by preventing corrosion but also drastically simplifies the workup procedure, as there is no need to filter off large volumes of iron sludge. These operational improvements contribute to significant cost savings in aminopyrimidine derivative manufacturing without compromising on quality.
- Cost Reduction in Manufacturing: The ability to purify intermediates and the final product via crystallization rather than column chromatography represents a massive reduction in processing costs. Chromatography is solvent-intensive, time-consuming, and difficult to scale, whereas crystallization is a standard, low-cost unit operation. Furthermore, the avoidance of expensive and hazardous reagents like sodium hydride reduces raw material costs and waste disposal fees. The higher yields reported in the examples, such as the 99.6% yield in the acrylamide formation step, further enhance the overall economic viability of the process.
- Enhanced Supply Chain Reliability: Utilizing stable reagents like 3-chloropropionyl chloride and ammonium formate ensures a more resilient supply chain compared to relying on unstable acryloyl chloride or pyrophoric sodium hydride. The robustness of the catalytic hydrogenation step means that batch-to-batch variability is minimized, leading to more predictable production schedules. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their production cycles with greater confidence and efficiency.
- Scalability and Environmental Compliance: The process is inherently designed for industrial mass production, avoiding the generation of heavy metal waste associated with iron reductions. The use of catalytic amounts of palladium, which can potentially be recovered, aligns better with green chemistry principles and environmental regulations. The simplified purification train reduces the volume of organic solvents required, lowering the environmental footprint of the manufacturing process. This scalability ensures that the technology can seamlessly transition from pilot plant to multi-ton commercial production to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of aminopyrimidine derivatives. These answers are derived directly from the experimental data and technical disclosures within patent CN111032650A, providing clarity on the process advantages and intermediate structures. Understanding these details is essential for technical procurement teams evaluating potential suppliers for this critical oncology intermediate.
Q: Why is the new process safer than the conventional method described in WO 2016/060443?
A: The conventional method relies on sodium hydride, which poses significant fire and explosion risks, and iron/ammonium chloride reduction, which causes reactor corrosion and generates difficult-to-remove black pitches. The new process utilizes catalytic transfer hydrogenation and mild base elimination, significantly enhancing operational safety and equipment longevity.
Q: How does this synthesis route improve purity for pharmaceutical applications?
A: By avoiding the formation of unknown degradation products and black pitches associated with iron reduction, the new method allows for purification via crystallization rather than column chromatography. This ensures higher purity suitable for GMP manufacturing and reduces the risk of metal contamination.
Q: What are the key intermediates protected in patent CN111032650A?
A: The patent protects several novel intermediates including N-(5-(4-(4-formyl-3-phenyl-1H-pyrazol-1-yl)pyrimidin-2-ylamino)-4-methoxy-2-morpholinophenyl)acrylamide (Formula 2), N-(5-carboxamido-4-methoxy-2-morpholinophenyl)acrylamide (Formula 4), and specific halogenated propionamide derivatives (Formula 5).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(5-(4-(4-((dimethylamino)methyl)-3-phenyl-1H-pyrazol-1-yl)pyrimidin-2-ylamino)-4-methoxy-2-morpholinophenyl)acrylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving oncology therapies. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like this aminopyrimidine derivative are manufactured with the highest standards of quality. We operate stringent purity specifications and maintain rigorous QC labs to verify that every batch meets the exacting requirements of global pharmaceutical partners, minimizing the risk of delays in your drug development timeline.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your mission to bring effective cancer treatments to patients worldwide.
