Scalable Metal-Free Synthesis of 4-Arylquinolines for Pharmaceutical Applications
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and environmentally benign synthetic routes for pharmacologically active scaffolds. A significant breakthrough in this domain is documented in patent CN113527198A, which details a novel method for preparing 4-arylquinoline compounds directly from 2-aminodiaryl methanol. This technology represents a paradigm shift from traditional metal-catalyzed processes to a base-promoted cyclization that utilizes dimethyl sulfoxide (DMSO) and formaldehyde not merely as solvents, but as integral carbon building blocks. For R&D directors and procurement specialists in the pharmaceutical sector, this development offers a compelling alternative for generating high-purity quinoline intermediates. The process operates under an oxygen-containing atmosphere, leveraging the oxidative potential of air or pure oxygen to drive the transformation without the need for stoichiometric oxidants or precious metal catalysts. This approach not only streamlines the synthetic workflow but also aligns with modern green chemistry principles by minimizing toxic waste streams associated with heavy metal removal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinoline nucleus has relied heavily on classical name reactions such as the Skraup synthesis, which often necessitates harsh conditions including strong acids and high temperatures that can degrade sensitive functional groups. Furthermore, more contemporary methods have attempted to address these issues but introduced new complexities. For instance, literature precedents describe the synthesis of 4-aryl-substituted quinolines using anilines, arylethenes, and arylmethyl ketones catalyzed by iodine. 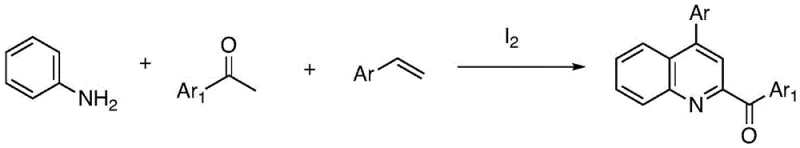 However, a critical drawback of this iodine-mediated pathway is the inadvertent introduction of an aroyl group at the 3-position of the quinoline ring, which limits its utility when a simple 4-aryl substitution pattern is desired. Similarly, other reported methods utilizing ferric iron catalysts to react arylmethyl ketones with anilines and DMSO have shown promise but suffer from inconsistent product yields and the inherent challenges of removing residual iron from the final API intermediate. These conventional routes often involve multiple steps, expensive reagents, and rigorous purification protocols to meet the stringent impurity profiles required by regulatory bodies, thereby inflating the cost of goods and extending lead times for drug development programs.
However, a critical drawback of this iodine-mediated pathway is the inadvertent introduction of an aroyl group at the 3-position of the quinoline ring, which limits its utility when a simple 4-aryl substitution pattern is desired. Similarly, other reported methods utilizing ferric iron catalysts to react arylmethyl ketones with anilines and DMSO have shown promise but suffer from inconsistent product yields and the inherent challenges of removing residual iron from the final API intermediate. These conventional routes often involve multiple steps, expensive reagents, and rigorous purification protocols to meet the stringent impurity profiles required by regulatory bodies, thereby inflating the cost of goods and extending lead times for drug development programs.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113527198A offers a direct, one-pot conversion that bypasses the need for pre-functionalized ketones or alkene coupling partners. The core innovation lies in the use of 2-aminodiaryl methanol as a unified precursor that already contains the necessary aromatic framework. By heating this substrate in a mixed solution of formaldehyde and DMSO under basic conditions, the quinoline ring is constructed with remarkable efficiency. 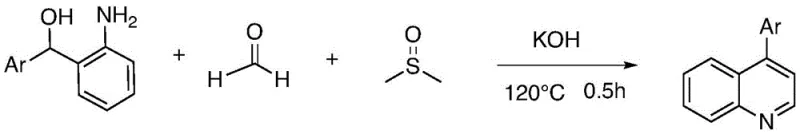 This novel approach utilizes DMSO and formaldehyde as both the reaction medium and the source of the C2 and C3 carbons for the pyridine ring, effectively turning cheap commodity chemicals into valuable structural elements. The reaction proceeds smoothly at temperatures around 120°C and completes in as little as 30 minutes, offering a drastic reduction in processing time compared to multi-step sequences. Moreover, the absence of transition metals means the resulting 4-arylquinoline products are free from heavy metal contamination, a critical quality attribute for pharmaceutical intermediates that simplifies the regulatory filing process and reduces the burden on quality control laboratories.
This novel approach utilizes DMSO and formaldehyde as both the reaction medium and the source of the C2 and C3 carbons for the pyridine ring, effectively turning cheap commodity chemicals into valuable structural elements. The reaction proceeds smoothly at temperatures around 120°C and completes in as little as 30 minutes, offering a drastic reduction in processing time compared to multi-step sequences. Moreover, the absence of transition metals means the resulting 4-arylquinoline products are free from heavy metal contamination, a critical quality attribute for pharmaceutical intermediates that simplifies the regulatory filing process and reduces the burden on quality control laboratories.
Mechanistic Insights into Base-Promoted Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with the oxidation of the starting 2-aminodiaryl methanol to an o-aminobenzophenone intermediate, a step facilitated by the oxygen-containing atmosphere. Simultaneously, the base promotes the reaction between formaldehyde and DMSO to generate a reactive methylsulfinylethene species. ![Detailed reaction mechanism showing [4+2] cycloaddition and elimination steps](/insights/img/4-arylquinoline-synthesis-dmso-pharma-supplier-20260306132220-06.webp) This transient olefin then undergoes a [4+2] cycloaddition with the oxidized aminobenzophenone intermediate, forming a unstable cyclic adduct. This adduct rapidly dehydrates under the alkaline conditions to form a dihydroquinoline derivative, which subsequently eliminates a molecule of methylsulfinic acid upon heating to yield the fully aromatic 4-arylquinoline product. The elegance of this mechanism lies in its atom economy; every component of the reagent mixture plays a specific role in constructing the final heterocycle. GC-MS analysis has confirmed the presence of key intermediates such as the methylsulfinylethene and the cyclic adduct, validating the proposed pathway. This deep mechanistic understanding allows chemists to fine-tune reaction parameters, such as base strength and oxygen flow, to maximize conversion and minimize side reactions.
This transient olefin then undergoes a [4+2] cycloaddition with the oxidized aminobenzophenone intermediate, forming a unstable cyclic adduct. This adduct rapidly dehydrates under the alkaline conditions to form a dihydroquinoline derivative, which subsequently eliminates a molecule of methylsulfinic acid upon heating to yield the fully aromatic 4-arylquinoline product. The elegance of this mechanism lies in its atom economy; every component of the reagent mixture plays a specific role in constructing the final heterocycle. GC-MS analysis has confirmed the presence of key intermediates such as the methylsulfinylethene and the cyclic adduct, validating the proposed pathway. This deep mechanistic understanding allows chemists to fine-tune reaction parameters, such as base strength and oxygen flow, to maximize conversion and minimize side reactions.
From an impurity control perspective, this mechanism offers distinct advantages. Since the reaction does not rely on radical chains initiated by metal catalysts, the formation of complex polymeric byproducts or halogenated impurities is significantly suppressed. The primary byproduct, methylsulfinic acid, is water-soluble and easily removed during the aqueous workup or chromatographic purification. This clean reaction profile ensures that the crude product obtained after concentration is of high purity, reducing the load on downstream purification units. For process chemists, this means fewer crystallization cycles or chromatography passes are needed to achieve the required specification, directly translating to higher overall recovery rates and reduced solvent consumption. The ability to predict and control the formation of the o-aminobenzophenone intermediate also provides a handle for monitoring reaction progress, ensuring consistent batch-to-batch reproducibility which is vital for commercial manufacturing.
How to Synthesize 4-Arylquinoline Efficiently
Implementing this synthesis on a laboratory or pilot scale requires careful attention to the stoichiometry of the base and the ratio of the solvent-reagents. The patent specifies that a molar ratio of 2-aminodiaryl methanol to base, preferably potassium hydroxide, of approximately 1:1 yields optimal results. The solvent system, comprising DMSO and formaldehyde, should be used in large excess to drive the equilibrium towards the product and ensure adequate solubility of the polar intermediates. The detailed standardized synthesis steps, including specific workup procedures and purification gradients, are outlined below to guide your technical team in replicating this high-efficiency route.
- Mix 2-aminodiaryl methanol with potassium hydroxide in a solvent system of DMSO and formaldehyde.
- Heat the reaction mixture to 120°C under an oxygen or air atmosphere for approximately 30 minutes.
- Cool the reaction, concentrate the mixture, and purify the crude product via silica gel column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift away from specialized, high-cost catalysts to ubiquitous inorganic bases fundamentally alters the cost structure of the manufacturing process. By eliminating the dependency on iodine or iron complexes, the supply chain becomes more resilient to fluctuations in the prices of specialty chemicals. Furthermore, the use of DMSO and formaldehyde, which are produced on a massive industrial scale, ensures a stable and continuous supply of raw materials, mitigating the risk of production stoppages due to reagent shortages. This stability is crucial for maintaining the continuity of supply for long-term pharmaceutical projects where reliability is paramount.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the simplification of the bill of materials. Removing expensive transition metal catalysts not only lowers the direct material cost but also eliminates the downstream costs associated with metal scavenging and validation testing for heavy metal residues. The one-pot nature of the reaction reduces labor hours and energy consumption by consolidating multiple synthetic steps into a single thermal operation. Additionally, the high yields reported for a wide range of substrates mean that less starting material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final API intermediate. These cumulative savings contribute to a significantly more competitive pricing structure for the finished product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like formaldehyde and DMSO, coupled with simple inorganic bases, creates a robust supply chain that is less susceptible to geopolitical or logistical disruptions often seen with niche reagents. The short reaction time of approximately 30 minutes allows for rapid turnover of reactor vessels, increasing the throughput capacity of existing manufacturing infrastructure without the need for capital investment in new equipment. This agility enables suppliers to respond quickly to changes in demand, ensuring that clients receive their orders within tighter lead times. The simplicity of the process also facilitates technology transfer between different manufacturing sites, providing redundancy and flexibility in the global supply network.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of hazardous reagents and the use of high-boiling, stable solvents. The reaction conditions are mild enough to be managed with standard stainless steel reactors, avoiding the need for specialized glass-lined or Hastelloy equipment required for corrosive acid-catalyzed processes. From an environmental standpoint, the reduction in waste generation, particularly heavy metal sludge, simplifies wastewater treatment and disposal, helping manufacturers meet increasingly stringent environmental regulations. The use of oxygen from air as the terminal oxidant further enhances the green credentials of the process, making it an attractive option for companies committed to sustainable manufacturing practices and carbon footprint reduction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the specific capabilities and constraints detailed in the patent literature. These insights are intended to clarify the operational parameters and quality expectations for stakeholders evaluating this technology for integration into their supply chains. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: What are the carbon sources for the quinoline ring in this novel synthesis?
A: In this specific pathway, the carbon atom at the 2-position of the quinoline ring is derived from DMSO, while the carbon atom at the 3-position is provided by formaldehyde. The remaining structural framework originates from the 2-aminodiaryl methanol starting material.
Q: Does this method require expensive transition metal catalysts?
A: No, this process eliminates the need for expensive transition metals like iodine or iron complexes. It utilizes common inorganic or organic bases, such as potassium hydroxide, which significantly reduces raw material costs and simplifies downstream purification by avoiding heavy metal residues.
Q: What is the typical reaction time and temperature for this conversion?
A: The reaction operates under relatively mild thermal conditions, typically requiring heating to 120°C. The conversion is rapid, often completing within 30 minutes under an oxygen-containing atmosphere, which enhances throughput compared to traditional multi-step syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Arylquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the methodology described in CN113527198A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities and rigorous QC labs capable of meeting stringent purity specifications for complex heterocyclic intermediates. Our commitment to quality ensures that every batch of 4-arylquinoline delivered meets the highest standards required for pharmaceutical applications, providing you with a reliable foundation for your drug development pipeline.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free route can optimize your budget. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom derivatives. Let us be your partner in transforming innovative chemistry into commercial success.
