Scalable Synthesis of T541-D: A Breakthrough for mTOR Inhibitor Manufacturing
Scalable Synthesis of T541-D: A Breakthrough for mTOR Inhibitor Manufacturing
The pharmaceutical industry constantly seeks robust pathways for synthesizing complex heterocyclic intermediates, particularly those serving as key building blocks for oncology therapeutics. A recent technological advancement detailed in patent CN112409399B introduces a highly efficient preparation method for the drug intermediate compound T541, specifically its boronic ester precursor T541-D. This compound, chemically known as 2-(5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridin-2-yl)propan-2-ol hydrochloride, acts as a critical scaffold for introducing aromatic substituents in the synthesis of potent antitumor agents, including mTORC1 and mTORC2 target inhibitors. The disclosed methodology represents a significant leap forward in process chemistry, addressing long-standing challenges related to yield, purity, and operational safety that have historically plagued the production of such pyridine-based scaffolds.
For R&D directors and process chemists, the implications of this patent are profound. It shifts the paradigm from low-yielding, multi-step sequences requiring rigorous purification to a streamlined, three-step protocol amenable to large-scale manufacturing. By optimizing reaction parameters such as temperature gradients, stoichiometric ratios, and solvent systems, the inventors have achieved a process that not only enhances the chemical fidelity of the final product but also drastically simplifies the downstream processing requirements. This report delves into the technical nuances of this novel route, analyzing its mechanistic underpinnings and evaluating its commercial viability for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in Chinese patent application CN104093398A, relied on synthetic routes that were inherently inefficient for industrial application. As illustrated in the historical context of this chemistry, previous approaches often suffered from poor atom economy and low selectivity during key transformation steps. Specifically, the yield of intermediate compounds analogous to T541-C was reported to be merely around 52%, while the subsequent conversion to the boronic ester derivative (T541-D) plummeted to approximately 27%. Such low yields are economically unsustainable for commercial API manufacturing, leading to excessive waste generation and inflated production costs. Furthermore, these legacy processes often necessitated harsh reaction conditions and complex purification techniques, such as column chromatography, which are impractical for multi-kilogram or ton-scale production.
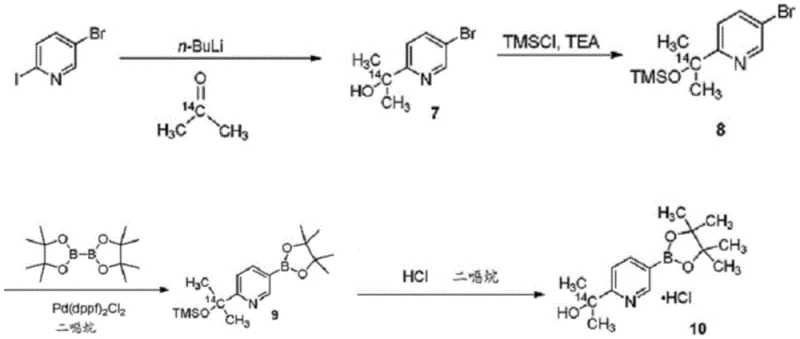
The Novel Approach
In stark contrast, the methodology presented in CN112409399B offers a streamlined three-step synthesis that bypasses these bottlenecks. The new route begins with the selective lithiation of 2,5-dibromopyridine (T541-RM1), followed by silylation and a final palladium-catalyzed borylation. This approach is characterized by its strong controllability and absence of harsh conditions. The process achieves a remarkable improvement in efficiency, with the intermediate T541-C reaching yields of nearly 100% in crude form and the final product T541-D achieving high purity levels suitable for direct use in subsequent coupling reactions. By eliminating the need for intermediate isolation and chromatographic purification, this novel approach significantly reduces the operational footprint and enhances the overall throughput of the manufacturing line.
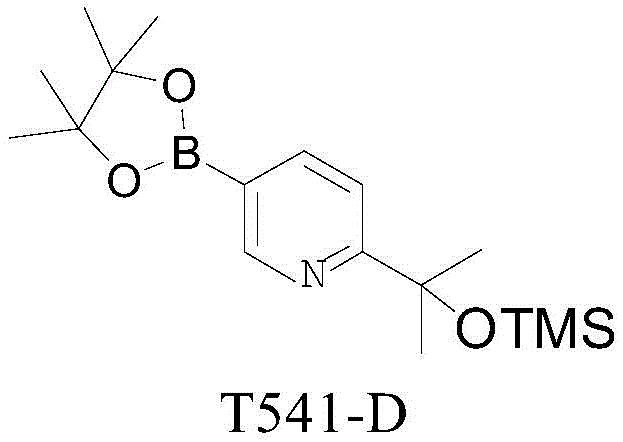
Mechanistic Insights into Lithiation and Pd-Catalyzed Borylation
The success of this synthesis hinges on precise control over two critical chemical transformations: the regioselective lithiation of the pyridine ring and the subsequent cross-coupling borylation. In the first step, the reaction of T541-RM1 with n-butyllithium must be meticulously managed to prevent the formation of regioisomeric impurities. Experimental data indicates that the temperature during the addition of the base is a decisive factor; maintaining the system between -70°C and -55°C maximizes the ratio of the desired product T541-B against byproducts like Z1T541-B and Z2T541-B. Deviations above -50°C lead to a sharp decline in selectivity, underscoring the kinetic sensitivity of the lithiation at the C2 position versus the C5 position of the pyridine ring. The optimized molar ratio of substrate to acetone to base (1.0:1.2:1.05) further ensures complete conversion while minimizing side reactions.
The final step involves a Miyaura borylation, a cornerstone reaction in modern medicinal chemistry for installing boron handles for Suzuki-Miyaura couplings. As depicted in the reaction scheme, T541-C reacts with pinacol diboron ((BPIN)2) in the presence of a palladium catalyst, specifically 1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium(II) (Pd(dppf)Cl2), and potassium acetate. This catalytic cycle operates efficiently in 1,4-dioxane at elevated temperatures (80-90°C). The choice of ligand and base is crucial here; the bidentate phosphine ligand stabilizes the palladium center, facilitating the oxidative addition and transmetallation steps required to replace the bromine atom with the pinacol boronate ester. The result is a high-purity product (99.79%) that retains the integrity of the sensitive silyl-protected alcohol moiety, demonstrating the chemoselectivity of the catalytic system.
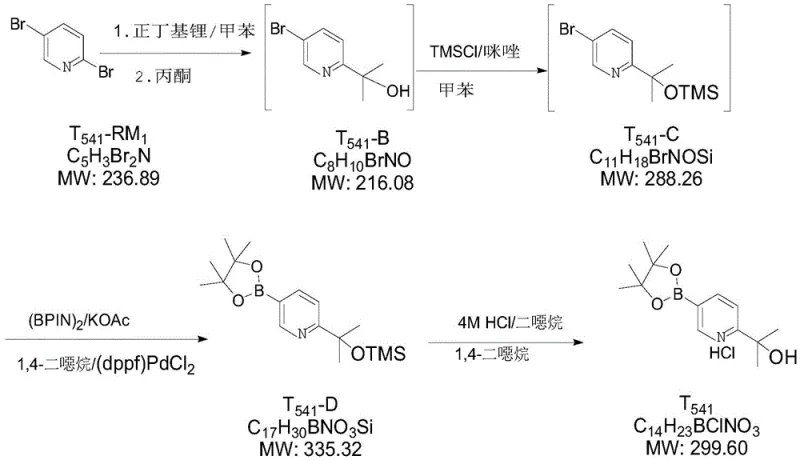
How to Synthesize T541-D Efficiently
The synthesis of T541-D is executed through a sequential three-step protocol that prioritizes operational simplicity and safety. The process begins with the cryogenic lithiation of the dibromopyridine starting material, followed by a quenching and protection sequence, and concludes with the thermal borylation reaction. Each step has been rigorously optimized to balance reaction kinetics with impurity profiles, ensuring that the intermediate streams are of sufficient quality to proceed without extensive purification. For a detailed breakdown of the specific operating parameters, reagent grades, and workup procedures, please refer to the standardized synthesis guide below.
- React 2,5-dibromopyridine (T541-RM1) with n-butyllithium and acetone in toluene at -70 to -55°C to form T541-B.
- Protect the hydroxyl group of T541-B using trimethylchlorosilane and imidazole in toluene to generate T541-C.
- Perform Miyaura borylation on T541-C using pinacol diboron and Pd(dppf)Cl2 in dioxane at 80-90°C to yield T541-D.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere chemical elegance. The primary value driver is the drastic simplification of the manufacturing workflow, which translates directly into reduced operational expenditures and enhanced supply reliability. By designing a process that avoids chromatographic purification and utilizes common, low-toxicity solvents like toluene and dioxane, the barrier to entry for scaling this molecule is significantly lowered. This makes the supply chain more resilient to disruptions, as the raw materials are commodity chemicals readily available from multiple global vendors, reducing dependency on single-source specialty reagents.
- Cost Reduction in Manufacturing: The elimination of complex purification steps such as column chromatography represents a massive saving in both time and material costs. In traditional fine chemical manufacturing, chromatography is a major bottleneck that limits batch size and increases solvent consumption exponentially. By relying on aqueous workups, crystallization, and filtration, this process enables the use of standard reactor trains found in most multipurpose chemical plants. Furthermore, the high yield of the intermediate T541-C means that less starting material is wasted, improving the overall mass balance and reducing the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable production schedule. The tolerance of the process to standard industrial equipment and the use of stable reagents minimize the risk of batch failures due to sensitive handling requirements. For supply chain managers, this reliability is paramount; it ensures consistent lead times and the ability to forecast inventory levels with greater accuracy. The ability to produce high-purity intermediates that can be telescoped into subsequent steps without isolation further compresses the manufacturing timeline, allowing for faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing solvents and conditions that are manageable in large-volume reactors. The avoidance of exotic or highly hazardous reagents simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The high selectivity of the lithiation step reduces the burden on downstream waste streams by minimizing the formation of difficult-to-separate isomers. This environmental efficiency not only lowers disposal costs but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of T541-D. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and advantages of this specific synthetic route. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their own development pipelines.
Q: What is the primary advantage of the new T541-D synthesis route?
A: The new route described in patent CN112409399B eliminates harsh conditions and complex purification steps, achieving a 3-step synthesis with significantly improved selectivity and yield compared to prior art methods.
Q: How is regioselectivity controlled during the lithiation step?
A: Regioselectivity is strictly controlled by maintaining the reaction temperature between -70°C and -55°C during the addition of n-butyllithium, minimizing the formation of unwanted isomers like Z1T541-B and Z2T541-B.
Q: Is chromatographic purification required for the intermediates?
A: No, the process is designed for industrial scalability where intermediates T541-B and T541-C can be used directly in subsequent steps without column chromatography, relying instead on aqueous workups and crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable T541-D Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation oncology therapeutics. Our team of expert process chemists has extensively analyzed the route described in CN112409399B and possesses the technical capability to execute this synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from clinical trials to full-scale commercialization. Our facilities are equipped with state-of-the-art cryogenic reactors and rigorous QC labs capable of meeting stringent purity specifications required for GMP manufacturing.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
