Advanced Continuous Manufacturing of High-Purity Alkoxyl Amine Hydrochloride Intermediates
Introduction to Next-Generation Alkoxyl Amine Synthesis
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways for critical intermediates like alkoxyl amine hydrochloride, a versatile reagent used in the synthesis of cephalosporin side chains and various herbicides. A significant technological breakthrough in this domain is detailed in patent CN102911079A, which introduces a novel method for preparing alkoxyl amine hydrochloride using ketoxime ethers via a continuous catalytic distillation process. This innovation addresses long-standing inefficiencies in traditional batch processing by integrating the reaction and separation phases within a single rectification tower. By continuously injecting acetoxime ether or diacetylmonoxime ether alongside aqueous hydrochloric acid into specific zones of the tower, the process achieves high purity and yield without the need for extraneous organic solvents. This approach not only streamlines the manufacturing workflow but also drastically reduces the environmental footprint associated with solvent recovery and waste disposal, marking a pivotal shift towards greener chemical engineering practices for high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkoxyl amine hydrochloride has been plagued by severe safety hazards and environmental burdens inherent to older methodologies. Traditional routes often rely on the reaction of sodium nitrite with sulfur dioxide to generate hydroxylamine-N,N-disulfonate, a process that produces massive quantities of sodium sulfate wastewater and toxic nitrogen oxide gases. Alternative methods utilizing hydroxylamine-O-sulfonic acid require dangerous oleum and complex pressure controls, leading to significant operational risks and difficult waste acid treatment protocols. Furthermore, previous attempts to hydrolyze ketoxime ethers typically involved batch reactions requiring the addition of organic solvents like toluene or hexane to facilitate separation. These batch processes suffer from prolonged reaction times, increased side reactions due to poor heat transfer, and the logistical complexity of solvent recovery, all of which contribute to lower overall yields and higher production costs that are unsustainable for modern large-scale manufacturing requirements.
The Novel Approach
The methodology outlined in CN102911079A represents a paradigm shift by employing a continuous catalytic distillation technique that eliminates the need for additional organic solvents during the critical hydrolysis stage. In this advanced setup, the ketoxime ether and aqueous hydrochloric acid are fed continuously into a rectification tower at precisely calculated theoretical plate positions, ensuring optimal contact and reaction kinetics. The design specifies that the number of theoretical plates from the feed ports to the tower bottom accounts for 30-80% of the total, with a specific relationship between the two feed points (D1 < D2) that enhances separation efficiency. This configuration allows the target alkoxyl amine hydrochloride to be isolated from the tower bottom liquid with exceptional purity, while the byproduct ketone is efficiently removed as an overhead distillate. The integration of reaction and separation not only shortens the process time but also suppresses side reactions, resulting in a robust, scalable, and economically superior production route compared to legacy batch technologies.
Mechanistic Insights into TS-1 Catalyzed Oximation and Reactive Distillation
The complete synthetic route described in the patent begins with the formation of the ketoxime precursor, a step that leverages the unique properties of titanium-silicon molecular sieve catalysts, specifically TS-1. In this initial phase, acetone or butanone reacts with ammonia and hydrogen peroxide in an organic solvent to form acetoxime or diacetylmonoxime. The use of TS-1 is critical here, as it facilitates a highly selective ammoximation reaction under mild conditions (45-85°C), avoiding the heavy metal contamination often associated with homogeneous catalysts. This heterogeneous catalysis ensures that the resulting oxime has a clean impurity profile, which is essential for downstream pharmaceutical applications. The reaction stoichiometry is carefully controlled, with a hydrogen peroxide to ketone molar ratio of 1-1.3, ensuring complete conversion while minimizing excess oxidant waste. This green chemistry approach sets a high-purity foundation for the subsequent etherification and hydrolysis steps.

Following the oximation, the intermediate undergoes an etherification reaction to produce the stable ketoxime ether required for the final hydrolysis. This substitution reaction involves treating the acetoxime or diacetylmonoxime with a halohydrocarbon, such as methyl chloride or ethyl chloride, in the presence of an alkali metal hydroxide like sodium hydroxide. The reaction proceeds efficiently at temperatures between 20-70°C, yielding the corresponding ether with high selectivity. The use of polar aprotic solvents like DMSO or ionic liquids in this step further enhances the reaction rate and solubility of the reagents. Once the ketoxime ether is isolated, it serves as the key feedstock for the core innovation of this patent: the solvent-free hydrolysis via reactive distillation.
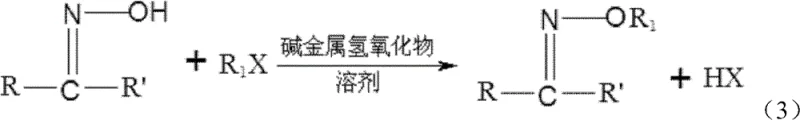
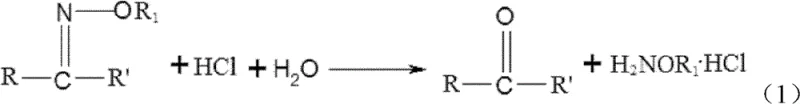
The final and most critical transformation is the hydrolysis of the ketoxime ether into alkoxyl amine hydrochloride, which occurs directly within the rectification tower. As depicted in the reaction scheme, the ketoxime ether reacts with aqueous hydrochloric acid and water to cleave the C=N bond, releasing the alkoxyl amine salt and regenerating the parent ketone. The brilliance of this process lies in the simultaneous reaction and separation; as the ketone byproduct forms, it is immediately vaporized and removed from the reaction zone as an overhead distillate, driving the equilibrium towards the product side according to Le Chatelier's principle. The tower bottom temperature is maintained between 70-110°C, while the top temperature is kept lower at 50-65°C, ensuring that the thermally sensitive alkoxyl amine hydrochloride remains in the liquid phase at the bottom while the volatile ketone exits the top. This precise thermal management prevents decomposition and maximizes yield.
How to Synthesize Methoxyamine Hydrochloride Efficiently
The synthesis of methoxyamine hydrochloride via this patented route offers a streamlined alternative to traditional batch methods, leveraging continuous processing to enhance throughput and consistency. The process integrates three distinct chemical transformations—oximation, etherification, and hydrolytic cleavage—into a cohesive manufacturing strategy that minimizes unit operations. By utilizing a titanium-silicon molecular sieve for the initial oxidation and a reactive distillation column for the final hydrolysis, manufacturers can achieve yields exceeding 93% with significantly reduced solvent consumption. The following guide outlines the standardized operational parameters derived from the patent examples, providing a roadmap for implementing this high-efficiency pathway in a commercial setting. Detailed standard operating procedures for equipment setup and safety protocols should be consulted prior to execution.
- Perform ketone oximidation using acetone or butanone with ammonia and hydrogen peroxide over a titanium-silicon molecular sieve catalyst to obtain acetoxime.
- React the resulting acetoxime with a halohydrocarbon in the presence of an alkali metal hydroxide to synthesize the corresponding ketoxime ether.
- Continuously inject the ketoxime ether and aqueous hydrochloric acid into a rectification tower at specific theoretical plate positions to isolate the alkoxyl amine hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN102911079A presents compelling opportunities for cost optimization and risk mitigation. The elimination of organic solvents in the final hydrolysis step removes the substantial capital and operational expenditures associated with solvent storage, recovery, and disposal. Furthermore, the continuous nature of the reactive distillation process allows for a steady, uninterrupted output of product, contrasting sharply with the stop-start cycles of batch processing that often lead to bottlenecks and inventory volatility. This shift to continuous manufacturing enhances supply chain reliability, ensuring that downstream customers receive consistent quality and quantity without the delays typical of batch campaign scheduling. The ability to recycle the byproduct ketone directly back into the front end of the process further insulates the operation from raw material price fluctuations, creating a more resilient and cost-effective supply model.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing train. By removing the requirement for additional organic solvents like toluene or hexane during the hydrolysis phase, the facility saves on both the purchase of these chemicals and the energy-intensive distillation required to recover them. Additionally, the high yield achieved through the reactive distillation mechanism means that less raw material is wasted to side products or incomplete conversion, directly lowering the cost of goods sold. The integration of reaction and separation into a single unit operation also reduces the footprint of the plant and the associated maintenance costs, contributing to substantial long-term operational savings without compromising on product quality or purity specifications.
- Enhanced Supply Chain Reliability: Transitioning from batch to continuous processing fundamentally improves the predictability of production schedules. In a continuous rectification tower setup, once steady state is achieved, the output of alkoxyl amine hydrochloride is constant, allowing for more accurate forecasting and inventory management. This reliability is crucial for pharmaceutical supply chains where interruptions can have cascading effects on drug production timelines. Moreover, the process utilizes commercially available and stable feedstocks such as acetone, ammonia, and hydrochloric acid, reducing the dependency on specialized or hazardous reagents that might face supply constraints. The robustness of the TS-1 catalyst also ensures long run-times without frequent regeneration or replacement, further stabilizing the production schedule and minimizing unplanned downtime.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this technology offers a clear path to sustainable scale-up. The avoidance of toxic reagents like sulfur dioxide and oleum, which are common in legacy methods, significantly reduces the burden on waste treatment facilities and lowers the risk of regulatory non-compliance. The closed-loop recycling of the ketone byproduct minimizes volatile organic compound (VOC) emissions, aligning with increasingly stringent global environmental standards. Scalability is inherently supported by the column-based design, where capacity can be increased by adjusting column diameter and height rather than multiplying the number of batch reactors. This modular scalability allows manufacturers to respond flexibly to market demand surges while maintaining a low environmental impact profile, making it an attractive option for companies aiming to meet corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this catalytic distillation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and advantages of the method. Understanding these details is essential for technical teams evaluating the potential for technology transfer or process optimization within their existing manufacturing infrastructure. The answers reflect the specific conditions and outcomes observed in the patented examples, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of using catalytic distillation for alkoxyl amine hydrochloride production?
A: The primary advantages include the elimination of additional organic solvents during the hydrolysis step, the ability to operate continuously rather than in batches, and the integration of reaction and separation which significantly improves yield and purity while reducing energy consumption.
Q: How does the TS-1 catalyst contribute to the environmental profile of this synthesis?
A: The titanium-silicon molecular sieve (TS-1) catalyst enables the use of hydrogen peroxide as a clean oxidant, producing water as the only byproduct during the oximation step, thereby avoiding the toxic waste streams associated with traditional sodium nitrite or hydroxylamine-O-sulfonic acid methods.
Q: Can the byproduct ketone be recycled in this process?
A: Yes, the acetone or butanone generated during the hydrolysis step is distilled off as an overhead product, concentrated, and can be directly recycled back into the initial oximation step, creating a closed-loop system that minimizes raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkoxyl Amine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic distillation technologies in the production of high-value pharmaceutical intermediates like alkoxyl amine hydrochloride. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that for R&D directors and procurement leaders, consistency and compliance are paramount, and our infrastructure is designed to deliver exactly that, bridging the gap between cutting-edge patent chemistry and reliable commercial supply.
We invite you to collaborate with us to explore how this solvent-free, continuous manufacturing route can optimize your supply chain and reduce costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a supplier, but a strategic ally dedicated to advancing your chemical manufacturing capabilities through innovation and operational excellence.
