Advanced Photocatalytic Synthesis of Chlorocyclopentanone Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex cyclic scaffolds with high efficiency and minimal environmental impact. Patent CN114920640B introduces a groundbreaking photocatalytic strategy for the synthesis of chlorocyclopentanone derivatives, which serve as critical intermediates in the production of bioactive aromatic sesquiterpenes. This technology leverages tetrabutylammonium decatungstate (TBADT) as a potent hydrogen atom transfer catalyst to drive a [3+2] cycloaddition reaction between 3-methylbutyraldehyde and p-methylphenylacetylene chloride. Unlike traditional thermal methods that often require extreme temperatures or stoichiometric metal reagents, this approach operates under mild visible light irradiation at room temperature, achieving 100% atom economy. For R&D directors and procurement managers, this represents a significant shift towards sustainable manufacturing, offering a reliable API intermediate supplier pathway that reduces waste and energy consumption while maintaining rigorous purity standards essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic sesquiterpenes like Laurokamurene B and Cuparene has relied on cumbersome multi-step sequences that pose significant challenges for commercial scale-up of complex polymer additives and fine chemicals. Prior art, such as the methods reported by Srikrishna et al., often necessitates up to ten synthetic steps involving Claisen rearrangements and ring-closing metathesis, resulting in overall yields as low as 22%. These conventional routes frequently employ expensive transition metal catalysts like palladium or rhodium, which introduce heavy metal impurities that are difficult to remove to ppm levels required by regulatory bodies. Furthermore, the use of harsh reagents and high-energy conditions increases the operational risk and cost of goods sold, making the supply chain vulnerable to raw material fluctuations. The need for pre-functionalized substrates further complicates the logistics, extending lead times and reducing the overall agility of the manufacturing process in a competitive global market.
The Novel Approach
The novel photocatalytic methodology described in CN114920640B offers a transformative solution by streamlining the construction of the cyclopentanone core in a single step. By utilizing readily available 3-methylbutyraldehyde as a three-carbon synthon, this process eliminates the need for pre-functionalized starting materials, drastically simplifying the raw material sourcing strategy for procurement teams. The reaction proceeds via a radical mechanism initiated by light, avoiding the use of precious metals entirely and thereby reducing the burden on downstream purification systems. This direct [3+2] cycloaddition not only improves the step economy but also enhances the safety profile of the manufacturing plant by operating at ambient temperatures and pressures. For supply chain heads, this translates to a more resilient production model where cost reduction in electronic chemical manufacturing and pharma intermediates is achieved through process intensification rather than mere negotiation, ensuring a stable supply of high-value intermediates.
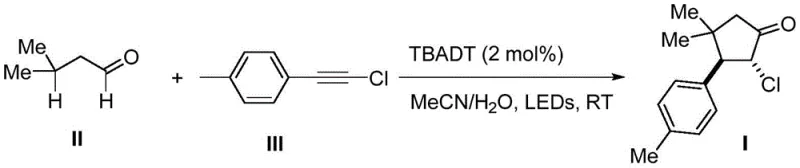
Mechanistic Insights into TBADT-Catalyzed [3+2] Cycloaddition
The core of this technological advancement lies in the unique ability of the decatungstate anion to act as a hydrogen atom transfer (HAT) photocatalyst. Upon irradiation with 390nm LEDs, the excited state of the TBADT catalyst abstracts a hydrogen atom from the formyl group of the 3-methylbutyraldehyde, generating an acyl radical species with high selectivity. This radical intermediate then undergoes a conjugate addition to the electron-deficient alkyne, followed by a crucial 1,5-hydrogen atom transfer that sets the stage for ring closure. The subsequent anti-Baldwin 5-endo-trig cyclization is particularly noteworthy, as it defies traditional thermal rules but is facilitated by the radical nature of the pathway. This mechanistic pathway ensures that the reaction proceeds with high diastereoselectivity, minimizing the formation of unwanted isomers that would otherwise complicate the purification process and reduce the overall yield of the target high-purity OLED material or pharmaceutical intermediate.
Impurity control is inherently built into this catalytic cycle due to the specific reactivity of the radical intermediates generated under photocatalytic conditions. Unlike ionic reactions that may suffer from competing nucleophilic attacks or elimination side reactions, the radical pathway is highly chemoselective for the desired C-C bond formation. The use of a mixed solvent system of acetonitrile and water further aids in solubilizing the ionic catalyst while maintaining the organic substrates in solution, creating a homogeneous reaction environment that promotes consistent kinetics. This level of control over the reaction trajectory means that the resulting chlorocyclopentanone derivative possesses a clean impurity profile, reducing the need for extensive recrystallization or chromatographic purification. For quality assurance teams, this predictability is invaluable, as it ensures batch-to-b consistency and facilitates the registration of the final drug substance with regulatory agencies without extensive impurity qualification studies.
How to Synthesize Chlorocyclopentanone Derivative Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to specific parameters regarding light intensity and catalyst loading to ensure optimal conversion. The process begins by dissolving the aldehyde and alkyne substrates in a degassed mixture of acetonitrile and water, followed by the addition of the TBADT catalyst under a nitrogen atmosphere to prevent oxygen quenching of the excited state. Detailed standardized synthesis steps see the guide below, which outlines the precise quenching and extraction protocols required to isolate the product with high purity. Adhering to these guidelines ensures that the photochemical efficiency is maximized while minimizing the formation of byproducts that could arise from over-irradiation or improper workup procedures.
- Mix 3-methylbutyraldehyde and p-methylphenylacetylene chloride with TBADT catalyst in MeCN/H2O solvent.
- Irradiate the reaction mixture with 390nm LEDs at room temperature under nitrogen atmosphere for 15 hours.
- Quench with water, extract with ethyl acetate, and purify via silica gel column chromatography to obtain Product I.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this photocatalytic route offers substantial strategic benefits for organizations looking to optimize their supply chain for high-purity API intermediates. The elimination of expensive transition metal catalysts such as palladium or nickel significantly lowers the raw material costs and removes the regulatory hurdle of residual metal testing, which is a common bottleneck in API manufacturing. Additionally, the mild reaction conditions reduce the energy consumption associated with heating and cooling, contributing to a lower carbon footprint and aligning with corporate sustainability goals. This process intensification allows for the potential use of continuous flow reactors, which can further enhance safety and throughput, making it an attractive option for large-scale production where traditional batch processes might be limited by heat transfer or light penetration issues.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts and the reduction in synthetic steps directly translate to lower operational expenditures. By avoiding the need for specialized equipment capable of handling high pressures or temperatures, capital investment is also minimized. The high atom economy ensures that a greater proportion of the raw materials end up in the final product, reducing waste disposal costs and improving the overall material efficiency of the plant. This logical deduction of cost savings is derived from the fundamental chemistry of the process rather than speculative market fluctuations, providing a stable basis for long-term financial planning.
- Enhanced Supply Chain Reliability: The starting materials, 3-methylbutyraldehyde and p-methylphenylacetylene chloride, are commodity chemicals that are readily available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions that often plague specialized reagents used in older synthetic routes. Furthermore, the robustness of the photocatalytic system means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when supply sources change. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The use of LED light sources and ambient temperature conditions makes this process inherently safer and easier to scale than exothermic thermal reactions. The aqueous component in the solvent system reduces the volume of organic waste generated, simplifying wastewater treatment and ensuring compliance with increasingly stringent environmental regulations. The ability to run the reaction in flow chemistry setups further enhances scalability, allowing manufacturers to increase capacity by numbering up reactors rather than building larger vessels, which offers a flexible and capital-efficient path to commercial production volumes.
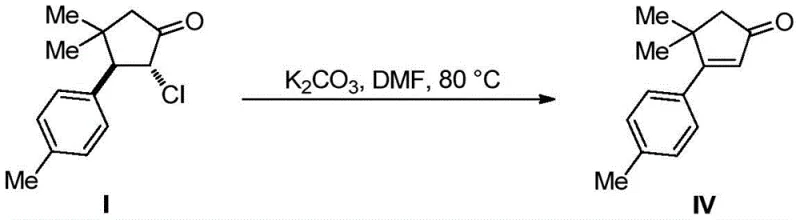
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology in industrial settings. These answers are derived directly from the patent data and practical considerations for scaling photochemical reactions, providing clarity for stakeholders evaluating this route for their own manufacturing portfolios. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: What are the advantages of this photocatalytic method over traditional synthesis?
A: This method achieves 100% atom economy and operates under mild room temperature conditions, avoiding the harsh reagents and multi-step sequences required by conventional Claisen or Stork-Danheiser reactions.
Q: Can this intermediate be used for large-scale sesquiterpene production?
A: Yes, the resulting chlorocyclopentanone derivative serves as a versatile precursor for Laurokamurene B and Cuparene, enabling efficient scale-up with simplified purification processes.
Q: Is the TBADT catalyst recoverable for cost efficiency?
A: The photocatalyst is used in low molar ratios (2 mol%), and the mild reaction conditions minimize catalyst degradation, supporting cost-effective manufacturing protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorocyclopentanone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic route for the production of complex sesquiterpene intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to leveraging this innovative chemistry to deliver cost-effective and sustainable solutions for your most challenging synthetic targets.
We invite you to collaborate with our technical procurement team to explore how this technology can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this photocatalytic method for your projects. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make data-driven decisions that enhance your competitive advantage in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
