Advanced Photocatalytic Synthesis of Sulfone-ylated Gamma-Lactones for Commercial Scale-up
Advanced Photocatalytic Synthesis of Sulfone-ylated Gamma-Lactones for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high efficiency and minimal environmental impact. Patent CN113387914B introduces a groundbreaking synthesis method for sulfone-ylated γ-lactone compounds, utilizing a visible light-driven photocatalytic system that operates under exceptionally mild conditions. This innovation represents a significant leap forward for manufacturers aiming to produce reliable pharmaceutical intermediates supplier grade materials without the safety hazards associated with traditional thermal processes. By leveraging trivalent metal iridium salts or divalent ruthenium salts as photocatalysts, this technology enables the direct sulfonylation and lactonization of 2-vinyl aryl carboxylic acids at room temperature. The strategic implementation of this patent allows for the creation of high-purity OLED material precursors and bioactive scaffolds with superior regioselectivity, addressing critical bottlenecks in modern organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sulfone-ylated γ-lactone frameworks has relied heavily on methodologies that pose significant safety and economic challenges for large-scale operations. Traditional approaches often utilize aryl diazonium salts in combination with sulfur dioxide complexes as sulfone donors, which are inherently unstable and carry a high risk of explosion, creating severe safety liabilities for production facilities. Furthermore, alternative pathways developed to mitigate these risks, such as those employing arylsulfinic acid sodium salts, still necessitate multi-step preparation from sulfonyl chlorides, adding unnecessary complexity and cost to the supply chain. Perhaps most critically, prior art involving copper-catalyzed systems frequently requires stoichiometric amounts of expensive noble metal salts like silver carbonate to drive the reaction, which drastically inflates the cost of goods sold and generates heavy metal waste that complicates environmental compliance and disposal protocols.
The Novel Approach
In stark contrast to these legacy techniques, the novel photocatalytic approach described in the patent data utilizes readily available sulfonyl chlorides directly as the sulfone group donor, effectively shortening the synthetic route and eliminating the need for hazardous diazonium intermediates. This method operates under visible blue light radiation at ambient temperatures ranging from 20°C to 25°C, which not only ensures energy efficiency but also preserves the integrity of sensitive functional groups that might degrade under harsh thermal conditions. The use of catalytic amounts of iridium or ruthenium complexes, combined with inexpensive inorganic bases such as sodium carbonate, creates a highly atom-economical process that aligns perfectly with the principles of green chemistry. This shift towards photochemical activation allows for cost reduction in fine chemical manufacturing by removing the dependency on stoichiometric oxidants and expensive silver additives, thereby streamlining the production workflow for complex organic intermediates.
Mechanistic Insights into Photocatalytic Sulfonylation and Lactonization
The core of this technological advancement lies in the sophisticated interplay between the photocatalyst and the visible light source, which initiates a single-electron transfer (SET) process to generate reactive radical species under mild conditions. Upon irradiation with blue LEDs, the iridium or ruthenium catalyst enters an excited state capable of activating the sulfonyl chloride bond, releasing a sulfonyl radical that selectively attacks the vinyl moiety of the 2-vinyl aryl carboxylic acid substrate. This radical addition is followed by an intramolecular cyclization event that forms the γ-lactone ring with exceptional regiocontrol, ensuring that the sulfone group is installed at the precise position required for downstream biological activity or material science applications. The mechanistic pathway avoids high-energy transition states associated with thermal activation, thereby minimizing the formation of undesired by-products and simplifying the purification process for the final API intermediate.
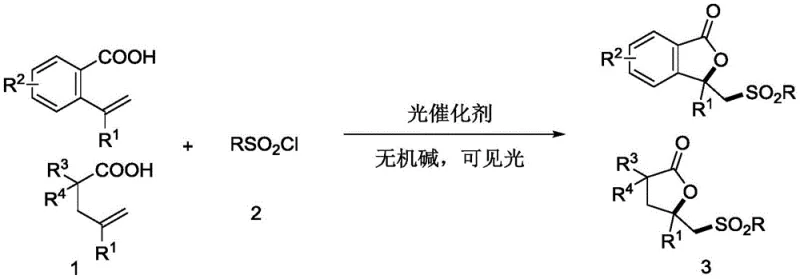
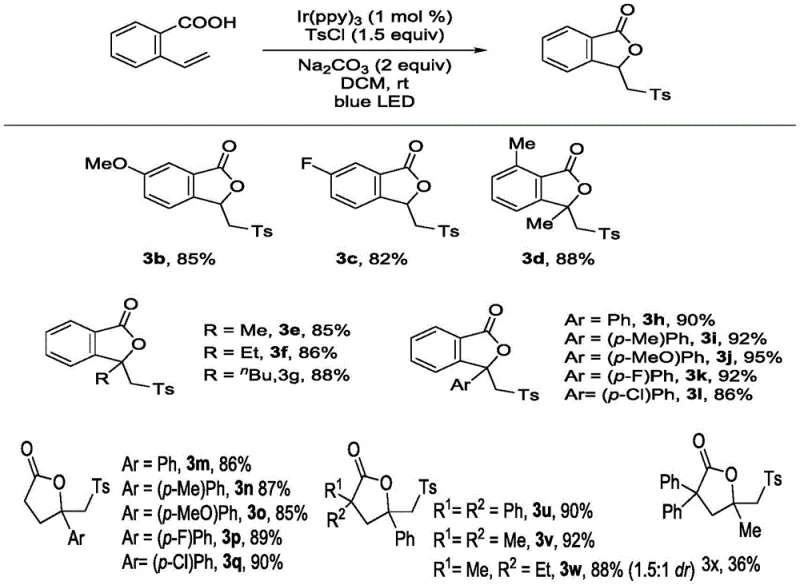
Furthermore, the impurity control mechanism inherent in this photocatalytic system is driven by the high specificity of the radical generation step, which occurs only in the presence of light and the specific catalyst, preventing uncontrolled side reactions common in thermal radical chemistry. The tolerance for a wide range of functional groups, including halogens, alkyl chains, and electron-donating or withdrawing substituents on the aromatic rings, demonstrates the robustness of the catalytic cycle against potential deactivation pathways. This broad substrate compatibility is crucial for R&D teams looking to diversify their chemical libraries without needing to re-optimize reaction conditions for every new analog, thus accelerating the drug discovery timeline. The ability to achieve high yields with minimal impurity profiles directly translates to reduced solvent usage during recrystallization and chromatography, further enhancing the overall sustainability and economic viability of the manufacturing process.
How to Synthesize Sulfone-ylated Gamma-Lactones Efficiently
The synthesis of these valuable lactone derivatives follows a streamlined protocol designed for reproducibility and ease of handling in both laboratory and pilot plant settings. The process begins with the precise mixing of the 2-vinyl aryl carboxylic acid starting material with the chosen sulfonyl chloride reagent in an anhydrous organic solvent such as dichloromethane or acetonitrile, ensuring that moisture-sensitive steps are minimized. Following the addition of the inorganic base and the photocatalyst, the reaction vessel is subjected to continuous irradiation from blue LED sources while maintaining vigorous stirring to ensure uniform light penetration and mass transfer throughout the reaction mixture. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 2-vinyl aryl carboxylic acid, sulfonyl chloride, inorganic base, and photocatalyst in an organic solvent.
- Irradiate the mixture with visible blue light at room temperature while stirring to initiate the photocatalytic cycle.
- Purify the resulting sulfone-ylated gamma-lactone compound using silica gel column chromatography after reaction completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers substantial strategic benefits that extend far beyond simple yield improvements, fundamentally altering the cost structure of producing these specialized intermediates. By eliminating the requirement for explosive diazonium salts and expensive silver carbonate promoters, the raw material costs are significantly reduced, allowing for more competitive pricing models in the global market for high-purity specialty chemicals. The operation at room temperature removes the need for energy-intensive heating or cryogenic cooling systems, leading to drastic simplifications in reactor design and utility consumption which directly impacts the operational expenditure of the manufacturing facility. Additionally, the use of commodity chemicals like sulfonyl chlorides and sodium carbonate ensures a stable and resilient supply chain, reducing the risk of production delays caused by the scarcity of exotic reagents often found in traditional noble metal catalysis.
- Cost Reduction in Manufacturing: The elimination of stoichiometric silver salts and hazardous diazonium precursors results in a leaner bill of materials, significantly lowering the direct cost of production per kilogram of finished product. This process optimization allows manufacturers to reallocate resources from waste management and safety containment towards capacity expansion and quality assurance initiatives. The high atom economy of the reaction means that less raw material is wasted as by-product, maximizing the value extracted from every pound of input chemical and improving the overall gross margin for the production line.
- Enhanced Supply Chain Reliability: Utilizing widely available sulfonyl chlorides and inorganic bases mitigates the risk of supply disruptions associated with specialized or regulated reagents, ensuring consistent production schedules for critical pharmaceutical intermediates. The robustness of the photocatalytic system against variations in substrate electronics means that sourcing flexibility is increased, allowing procurement teams to qualify multiple vendors for starting materials without compromising final product quality. This stability is essential for maintaining long-term contracts with downstream clients who require guaranteed continuity of supply for their own manufacturing pipelines.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal oxidants simplify the scale-up process from gram to ton scale, facilitating the commercial scale-up of complex polymer additives and drug intermediates with minimal engineering hurdles. The reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, reducing the liability and cost associated with effluent treatment and disposal. This eco-friendly profile enhances the corporate sustainability metrics of the manufacturing entity, making it a preferred partner for multinational corporations with strict carbon footprint and green chemistry mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method, providing clarity for stakeholders evaluating its integration into their existing workflows. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of adopting this technology for specific project requirements and regulatory filings.
Q: What are the safety advantages of this photocatalytic method over traditional diazonium salt routes?
A: This method eliminates the use of explosive aryl diazonium salts, significantly enhancing operational safety and environmental compliance during manufacturing.
Q: Does this synthesis require expensive noble metal scavengers like silver carbonate?
A: No, unlike previous copper-catalyzed methods requiring silver carbonate, this protocol uses earth-abundant bases and recyclable photocatalysts, reducing material costs.
Q: Is the reaction scalable for industrial production of pharmaceutical intermediates?
A: Yes, the mild room temperature conditions and use of visible light LEDs make the process highly adaptable for large-scale commercial production with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfone-ylated Gamma-Lactone Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our technical team is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of sulfone-ylated gamma-lactones produced under this innovative protocol. We understand the critical nature of supply chain consistency for our partners and are committed to delivering high-quality intermediates that meet the demanding standards of the global pharmaceutical and agrochemical industries.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. By engaging with us, you can access specific COA data and route feasibility assessments that demonstrate how this photocatalytic method can optimize your manufacturing economics. Let us collaborate to leverage this cutting-edge technology for your next project, ensuring a competitive edge through superior process chemistry and reliable supply chain execution.
